Abstract
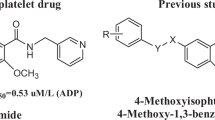
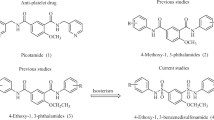
Twelve N-substituted anthranilamide esters (1–5, 8, 9, 12, 13, and 15–17) were synthesized and evaluated for their ability to inhibit the in vitro aggregation by washed human platelets induced by adenosine 5′-diphosphate (10 μM). The antiplatelet activity of dl-n-butyl 5-hydroxy-N-(2-phenoxypropionyl)anthranilate (9, IC50 = 10.5 μM) was most active among the tested compounds and ethyl ester 8 (IC50 = 11.2 μM) showed the second most activity. dl-Ethyl and dl-n-butyl 5-(p-toluenesulfonyloxy)-N-(2-phenoxypropionyl)anthranilate (12, IC50 = 13.1 μM and 13, IC50 = 14.0 μM), dl-methyl N-(2-phenoxybutyryl)anthranilate (2, IC50 = 12.7 μM), dl-N-(2-phenoxypropionyl)anthranilic acid (5, IC50 = 13.7 μM) displayed lower antiplatelet activity than 8 and 9. Compound 5 was more active than methyl ester prodrug 1. n-Butyl 5-hydroxy-N-(4′-acetoxybenzoyl)anthranilate (15, IC50 = 28.3 μM) showed moderate activity. Compounds 1 (IC50 = 42.8 μM), 4 (IC50 = 56.7 μM), 16 (IC50 = 51.0 μM), and 17 (IC50 = 49.8 μM) exhibited low antiplatelet activity. Methyl N-phenoxyacetylanthranilate (3, IC50 = 78.0 μM) showed the lowest antiplatelet activity. The compounds with branched alkyl chain (2 and 5) were more active than compounds with straight chain (3 and 4). The apparent permeability coefficient (Papp, cm/s) values of compounds 2 and 9 were determined as 45.34 ± 4.67 and 33.17 ± 5.15 × 10−6 cm/s by Caco-2 cell permeability assay.




Similar content being viewed by others
References
Angiolillo, D.J., D.L. Bhatt, P.A. Gurbel, and L.A. Jennings. 2009. Advances in antiplatelet therapy: Agents in clinical development. American Journal of Cardiology 103: 40A–51A.
Anninos, H., G. Andrikopoulos, S. Pastromas, D. Sakellarich, G. Theodorakis, and P. Vardas. 2009. Triflusal: An old drug in modern antiplatelet therapy. Review of its action, use, safety and effectiveness. Hellenic Journal of Cardiology 50: 199–207.
Antithrombotic triallist’s collaboration. 2002. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 324: 71–86.
Artursson, P., and J. Karlsson. 1991. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochemical and Biophysical Research Communication 175: 880–885.
Artursson, P., K. Palm, and K. Luthman. 1996. Caco-2 monolayers in experimental and theoretical predictions of drug transport. Advanced Drug Delivery Reviews 22: 67–84.
Bennett, J.S. 2001. Novel platelet inhibitors. Annual Review of Medicine 52: 161–184.
Berger, P.B. 1999. The thienopyridines in coronary artery disease. Current Cardiology Reports 1: 192–198.
Bhatt, D.L., and E.J. Topol. 2003. Scientific and therapeutic advances in antiplatelet therapy. Nature Reviews Drug Discovery 2: 15–28.
Born, G.V.R. 1962. Aggregation of blood platelets by adenosine diphosphate (ADP) and its reversal. Nature 194: 927–929.
Borne, R.F., R.L. Peden, I.W. Waters, M. Weiner, R. Jordan, and E.A. Coats. 1974. Anti-inflammatory activity of para-substituted N-benzenesulfonyl derivatives of anthranilic acid. Journal of Pharmaceutical Sciences 63: 615–617.
Caldwell, G.W., D.M. Richie, and J.A. Masucci. 2001. The new pre-preclinical paradigm: Compound optimization in early and late phase drug discovery. Current Topics in Medicinal Chemistry 1: 353–366.
Daniel, J.L., C. Dangelmaier, J. Jin, B. Ashby, J.B. Smith, and S.P. Kunapuli. 1998. Molecular basis for ADP induced platelet activation, I: Evidence for three distinct ADP receptors on human platelets. Journal of Biological Chemistry 273: 2024–2029.
Davi, G., and C. PatRono. 2007. Platelet activation and atherothrombosis. New England Journal of Medicine 357: 2482–2494.
Eisert, W.G. 2001. How to get from antiplatelet to antithrombotic treatment. American Journal of Therapeutics 8: 443–449.
Gan, L.-S., and D. Thakker. 1997. Applications of the Caco-2 model in the design and development of orally active drug elucidation of biochemical and physical barriers posed by the intestinal epithelium. Advanced Drug Delivery Reviews 23: 77–98.
He, X., M. Sugawara, Y. Takekuma, and K. Hiyazaki. 2004. Absorption of ester prodrugs in Caco-2 and rat intestine models. Antimicrobial Agents and Chemotherapy 48: 2604–2609.
Ha-Duong, N.T., S. Dijols, A.-C. Macherey, J.A. Goldstein, P.M. Dansette, and D. Mansey. 2001. Ticlopidine as a selective mechanism-based inhibitor of human cytochrome P450 2C19. Biochemistry 40: 12112–12122.
Ho, P.M., T.M. Maddox, L. Wang, S.D. Fihn, R.L. Jesse, E.D. Peterson, and J.S. Rumsfeld. 2009. Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibitors following acute coronary syndrome. Journal of the American Medical Association 301: 937–944.
Hsieh, P.W., S.Z. Chiang, C.C. Wu, Y.C. Lo, Y.T. Shih, and Y.C. Wu. 2008. Synthesis and anti-platelet evaluation of 2-benzoylaminobenzoate analogs. Bioorganic & Medicinal Chemistry 16: 5803–5814.
Hsieh, P.W., T.L. Hwang, C.C. Wu, S.Z. Chiang, C.L. Wu, and Y.C. Wu. 2007. The evaluation and structure-activity relationships of 2-benzoylaminobenzoic esters and their analogues as anti-inflammatory and anti-platelet aggregation agents. Bioorganic & Medicinal Chemistry Letters 17: 1812–1817.
Iwasa, Y., T. Iwasa, K. Matsui, T. Yoshimura, N. Tanaka, and K. Miyazaki. 1986. Anti-platelet action of an anti-allergic agent, N-(3′,4′-dimethoxycinnamoyl)anthranilic acid (Tranilast). European Journal of Pharmacology 120: 231–234.
Jackson, S.P., and S.M. Schoenwaelder. 2003. Antiplatelet therapy in search of the ‘magic bullet’. Nature Reviews Drug Discovery 2: 775–789.
Kunapuli, S.D. 1998. Multiple P2 receptor subtypes on platelets: A new interpretation of their function. Trends in Pharmacological Sciences 19: 391–394.
Lim, J.K., W.S. Woo, K.R. Lee, and E. Ma. 1994. Synthesis of melandrin derivatives. Yakhak Hoeji 38: 281–285.
Liu, F.-C., C.-H. Liao, Y.-E. Chang, J.-T. Liou, and Y.-J. Day. 2009. A new insight of anti-platelet effects of sirtinol in platelets aggregation via cyclic AMP phosphodiesterse. Biochemical Pharmacology 77: 1364–1373.
Losena, M.V., and B.M. Bolotin. 1972. Reaction of anthranilic acid and methoxy- and nitro-substituted anthranilic acid with p-toluenesulfonyl chloride in pyridine. Chemistry of Heterocyclic Compounds 8: 1212–1215.
Maekowska, M., S. Oberle, C.-P. Juzwin, M. Hsu, M. Gryszkiewicz, and A.J. Streeter. 2001. Optimizing Caco-2 cell monolayers to increase throughput in drug intestinal absorption analysis. Journal of Pharmacological and Toxicological Methods 46: 51–55.
Manikeri, S.R., C.N. Raghu, V.N. Dadlkar, and U.K. Sheth. 1980. Effect of Tromaril on platelet and blood coagulation. Indian Journal of Medical Research 71: 438–445.
Michelson, A.D. 2010. Antiplatelet therapies for the treatment of cardiovascular disease. Nature Reviews Drug Discovery 9: 154–169.
Schrör, K. 1995. Antiplatelet drugs: A comparative reviews. Drugs 50: 7–28.
Turner, N.A., J.L. Moake, and L.V. McIntire. 2001. Blockade of adenosine diphosphate receptors P2Y(12) and P2Y(1) is required to inhibit platelet aggregation in whole blood under flow. Blood 98: 3340–3345.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2010-0013516).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, S., Shin, B.S. & Ma, E. Synthesis and Caco-2 cell permeability of N-substituted anthranilamide esters as ADP inhibitor in platelets. Arch. Pharm. Res. 38, 1147–1156 (2015). https://doi.org/10.1007/s12272-014-0353-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-014-0353-1