Abstract
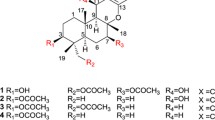
Two new meroterpenoids, arisugacins I (1) and J (2), together with five known meroterpenoids including arisugacin B (3), arisugacin F (4), arisugacin G (5), territrem B (6) and territrem C (7) were isolated from an endophytic fungus Penicillium sp. SXH-65. Their structures were determined by extensive spectroscopic experiments and comparison with literature data. Their cytotoxicities were evaluated against Hela, HL-60 and K562 cell lines, and only 3 and 4 exhibited weak cytotoxicities against Hela, HL-60 and K562 cell lines with IC50 values ranging from 24 to 60 μM.



Similar content being viewed by others
References
Dowd, P.F., F.C. Peng, J.W. Chen, and K.H. Ling. 1992. Toxicity and anticholinesterase activity of the fungal metabolites territrems to the corn earworm, Helicoverpa zea. Entomology Experimental Application 65: 57–64.
Itoh, T., K. Tokunaga, Y. Matsuda, I. Fujii, I. Abe, Y. Ebizuka, and T. Kushiro. 2010. Reconstitution of a fungal meroterpenoid biosynthesis reveals the involvement of a novel family of terpene cyclases. Nature Chemistry 2: 858–864.
Kogel, K.-H., P. Franken, and R. Huckelhoven. 2006. Endophyte or parasite: What decides. Current Opinion in Plant Biology 9: 358–363.
Kuno, F., K. Shiomi, K. Otoguro, T. Sunazuka, and S. Omura. 1996. Arisugacin A and B, novel and selective acetylcholinesterase inhibitors from Penicillium sp. FO-4259 II. Structure elucidation. Journal of Antibiotics 49: 748–751.
Ling, K.H., H.-H. Liou, C.-M. Yang, and C.-K. Yang. 1984. Isolation, chemical structure, acute toxicity, and some physicochemical properties of territrem C from Aspergillus terreus. Applied and Environment Microbiology 47: 98–100.
Omura, S., F. Kuno, K. Otoguro, T. Sunazuka, K. Shiomi, R. Masuma, and Y. Iwai. 1995. Arisugacin, a novel and selective inhibitor of acetylcholinesterase from Penicillium sp. FO-4259. Journal of Antibiotics 48: 745–746.
Otoguro, K., K. Shiomi, Y. Yamaguchi, N. Arai, T. Sunazuka, R. Masuma, Y. Iwai, and S. Omura. 2000. Arisugacins C and D, novel acetylcholinesterase inhibitors and their novel metabolites produced by Penicillium sp. FO-4259-11. Journal of Antibiotics 53: 50–57.
Peng, Fu-Chuo. 1997. Structure and anti-acetylcholinesterase activity of 4α-(hydroxymethyl)-4α-demethylterritrem B. Journal of Natural Products 60: 842–843.
Saikkonen, K., P. Wali, M. Helande, and S.H. Faeth. 2004. Evolution of endophyte: Plant symbioses. Trends in Plant Science 9: 275–280.
Schiff, P.B., and S.B. Horowitz. 1980. Taxol stabilizes microtubules in mouse fibroblast cells. Proceedings of the National Academy of Sciences of the United States of America 77: 1561–1565.
Schula, B., and C. Boyl. 2005. The endophytic continuum. Mycological Research 109: 661–686.
Strobel, G., B. Daisy, U. Castillo, and J. Harper. 2004. Natural product from endophytic microorganisms. Journal of Natural Products 67: 257–268.
Strobel, G.A., and B. Daisy. 2003. Bioprospecting for microbial endophytes and their natural products. Microbiology and Molecular Biology Reviews 67: 491–502.
Strobel, G.A., E. Ford, J. Worapong, J.K. Harper, A.M. Artif, D.M. Grant, P.C.W. Fung, and K. Chan. 2002. Ispoestacin, an isobenzofuranone from Pestalotiopsis microspora, possessing antifungal and antioxidant activities. Phytochemistry 60: 179–183.
Strobel, G.A., R.V. Miller, C. Miller, M. Condron, and D.B. Teplow. 1999. Cryptocandin, a potent antimycotic from endophytic fungus Cryptosporiopsis cf. quercina. Microbiology 145: 1919–1926.
Suffness, M. 1995. Taxol, science and applications. Boca Raton, FL: CRC Press.
Yang, S.X., J.M. Gao, H. Laatsch, J.M. Tian, and G. Pescitelli. 2012. Absolute configuration of fusarone, a new azaphilone from the endophytic fungus Fusarium sp. isolated from melia azedarach, and of related azaphilones. Chirality 24: 621–627.
Zhou, L.N., H.Q. Gao, ShX Cai, T.J. Zhu, Q.Q. Gu, and D.H. Li. 2011. Two new cyclic pentapeptides from the marine-derived fungus Aspergillus versicolor. Helvetica Chimica Acta 94(6): 1065–1070.
Acknowledgments
This work was supported by the Chinese National Science Fund (No. 41176120), Promotive Research Fund for Excellent Young and Middle-aged Scientists of Shandong Province (No. BS2010HZ027), Specialized Research Fund for the Doctoral Program of Higher Education (No. 20100132120026), the Public Projects of State Oceanic Administration (No. 2010418022-3), and the state key laboratory of Bio-organic and Natural Products Chemistry (No. SKLBNPC12331).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, X., Kong, X., Gao, H. et al. Two new meroterpenoids produced by the endophytic fungus Penicillium sp. SXH-65. Arch. Pharm. Res. 37, 978–982 (2014). https://doi.org/10.1007/s12272-013-0268-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0268-2