Abstract
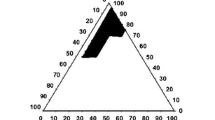
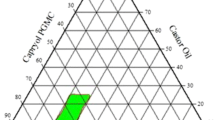
The aim of this study was to develop and characterize a self-microemulsifying drug delivery system (SMEDDS) of Brucea javanica oil (BJO) and transform the liquid formulation into solid granules. Solubility studies of BJO and pseudo-ternary phase diagrams were used to identify the most efficient self-emulsification region. A methyl thiazolyl tetrazolium (MTT) assay was performed to identify cell apoptosis. Antitumor activity studies were also employed to evaluate the BJO SMEDDS. The optimized BJO SMEDDS in liquid and granule formulations rapidly formed fine oil-in-water microemulsions with particle sizes <50 nm. Additionally, the MTT assay demonstrated that BJO SMEDDS had a significant effect on cancer cells, and antitumor activity studies showed remarkable inhibition of S180 tumors. The BJO SMEDDS, optimized to have good characteristics, was successfully transformed into solid granules by adsorbing onto crospovidone. The studies of the release of the BJO SMEDDS of liquid and granules in vitro suggested that the release of BJO was enhanced by the SMEDDS. These studies revealed that the new self-microemulsifying systems of liquid and granule forms might be promising strategies for the oral delivery of the poorly water-soluble drug BJO.





Similar content being viewed by others
References
Agarwal, V., A. Siddiqui, H. Ali, and S. Nazzal. 2009. Dissolution and powder flow characterization of solid self-emulsified drug delivery system (SEDDS). International Journal of Pharmaceutics 366: 44–52.
Balakrishnan, P., B.J. Lee, D.H. Oh, J.O. Kim, Y.I. Lee, D.D. Kim, J.P. Jee, Y.B. Lee, J.S. Woo, C.S. Yong, and H.G. Choi. 2009. Enhanced oral bioavailability of Coenzyme Q10 by self-emulsifying drug delivery systems. International Journal of Pharmaceutics 374: 66–72.
Cai, B., G. Chen, L. Wen, X.Y. Wu, G.Q. Liu, and X.Q. Huang. 2011. Determination of oleic acid and linoleic acid in Brucea javanica oil microcapsules by GC. Lishizhen Medicine and Materia Medica Research 22: 1057–1058.
Chang, R.K., K.S. Raghavan, and M.A. Hussain. 1998. A study on gelatin capsule brittleness: moisture transfer between the capsule shell and its content. Journal of Pharmaceutical Sciences 87: 556–558.
Charman, S.A., W.N. Charman, M.C. Rogge, T.D. Wilson, F.J. Dutko, and C.W. Pouton. 1992. Self-emulsifying drug delivery systems: formulation and biopharmaceutic evaluation of an investigational lipophilic compound. Pharmaceutical Research 9: 87–93.
Chen, G., X. Zhang, G. Lu, G.Q. Liu, J. Zeng, Y. Li, and S.Y. Wei. 2012a. O/W partition coefficient of Brucea javanica oil and absorption characteristics of its emulsion in rat intestine in situ. China Journal of Traditional Chinese Medicine and Pharmacy 27: 1669–1772.
Chen, G., X. Zhang, G. Lu, A.L. Shao, G.Q. Liu, and B. Cai. 2012b. Preparation of Brucea javanica oil microcapsule in situ gel system and its in vitro release mechanism. Chinese Traditional and Herbal Drugs 7: 1306–1309.
Constantinides, P.P. 1995. Lipid microemulsions for improving drug dissolution and oral absorption: physical and biopharmaceutical aspects. Pharmaceutical Research 12: 1561–1572.
Cui, Y., Z. Wu, X. Liu, R. Ni, X. Zhu, L. Ma, and J. Liu. 2010. Preparation, safety, pharmacokinetics, and pharmacodynamics of liposomes containing Brucea javanica oil. AAPS PharmSciTech 11: 878–884.
Gershanik, T., and S. Benita. 2000. Self-dispersing lipid formulations for improving oral absorption of lipophilic drugs. European Journal of Pharmaceutics and Biopharmaceutics 50: 179–188.
Gursoy, R.N., and S. Benita. 2004. Self-emulsifying drug delivery systems (SEDDS) for improved oral delivery of lipophilic drugs. Biomedicine and Pharmacotherapy 58: 173–182.
Huang, J.Z., X.G. Chen, and M.H. Gao. 2012. Determination of oleic acid fructus bruceae oil oral emulsion by gas chromatography. Traditional Chinese Drug Research and Clinical Pharmacology 23: 197–199.
Kang, B.K., J.S. Lee, S.K. Chon, S.Y. Jeong, S.H. Yuk, G. Khang, H.B. Lee, and S.H. Cho. 2004. Development of self-microemulsifying drug delivery systems (SMEDDS) for oral bioavailability enhancement of simvastatin in beagle dogs. International Journal of Pharmaceutics 274: 65–73.
Kim, I.H., S. Takashima, Y. Hitotsuyanagi, T. Hasuda, and K. Takeya. 2004. New quassinoids, javanicolides C and D and javanicosides B-F, from seeds of Brucea javanica. Journal of Natural Products 67: 863–868.
Lawrence, M.J., and G.D. Rees. 2000. Microemulsion-based media as novel drug delivery systems. Advanced Drug Delivery Reviews 45: 89–121.
Lei, Y., Y. Lu, J. Qi, S. Nie, F. Hu, W. Pan, and W. Wu. 2011. Solid self-nanoemulsifying cyclosporin A pellets prepared by fluid-bed coating: preparation, characterization and in vitro redispersibility. International Journal of Nanomedicine 6: 795–805.
Liu, J.H., J.J. Qin, H.Z. Jin, X.J. Hu, M. Chen, Y.H. Shen, S.K. Yan, and W.D. Zhang. 2009a. A new triterpenoid from Brucea javanica. Archives of Pharmacal Research 32: 661–666.
Liu, Y., G.D. Li, and C.C. Liu. 2009b. Recipe for self-emulsifying preparation of Brucea javanica seed oil. Academic Journal of Second Military Medical University 30: 1069–1073.
Mei, X., F.M. Etzler, and Z. Wang. 2006. Use of texture analysis to study hydrophilic solvent effects on the mechanical properties of hard gelatin capsules. International Journal of Pharmaceutics 324: 128–135.
Narang, A.S., D. Delmarre, and D. Gao. 2007. Stable drug encapsulation in micelles and microemulsions. International Journal of Pharmaceutics 345: 9–25.
Nazzal, S., and Y. Wang. 2001. Characterization of soft gelatin capsules by thermal analysis. International Journal of Pharmaceutics 230: 35–45.
Pan, L., Y.W. Chin, H.B. Chai, T.N. Ninh, D.D. Soejarto, and A.D. Kinghorn. 2009. Bioactivity-guided isolation of cytotoxic constituents of Brucea javanica collected in Vietnam. Bioorganic and Medicinal Chemistry 17: 2219–2224.
Peng, X.H., M.Q. Cui, Z.M. Ci, C.M. Fu, and S.H. Zhang. 2011. Development of self-emulsifying drug delivery system of Brucea javanica seed oil. Chinese Traditional Patent Medicine 33: 1520–1524.
Pouton, C.W. 2000. Lipid formulations for oral administration of drugs: non-emulsifying, self-emulsifying and ‘self-microemulsifying’ drug delivery systems. European Journal of Pharmaceutical Sciences 11(Suppl 2): S93–S98.
Shi, X.P., and H.R. Zhao. 2010. Development of Brucea javanica oil. Northwest Pharmaceutical Journal 25: 240–242.
Singh, B., S. Bandopadhyay, R. Kapil, R. Singh, and O. Katare. 2009. Self-emulsifying drug delivery systems (SEDDS): formulation development, characterization, and applications. Critical Reviews in Therapeutic Drug Carrier Systems 26: 427–521.
Singh, B., L. Khurana, S. Bandyopadhyay, R. Kapil, and O.O. Katare. 2011. Development of optimized self-nano-emulsifying drug delivery systems (SNEDDS) of carvedilol with enhanced bioavailability potential. Drug Delivery 18: 599–612.
Spernath, A., A. Aserin, and N. Garti. 2006. Fully dilutable microemulsions embedded with phospholipids and stabilized by short-chain organic acids and polyols. Journal of Colloid and Interface Science 299: 900–909.
Wang, Z., J. Sun, Y. Wang, X. Liu, Y. Liu, Q. Fu, P. Meng, and Z. He. 2010. Solid self-emulsifying nitrendipine pellets: preparation and in vitro/in vivo evaluation. International Journal of Pharmaceutics 383: 1–6.
Wu, W., Y. Wang, and L. Que. 2006. Enhanced bioavailability of silymarin by self-microemulsifying drug delivery system. European Journal of Pharmaceutics and Biopharmaceutics 63: 288–294.
Xu, L., H. Chen, H. Xu, and X. Yang. 2008. Anti-tumour and immuno-modulation effects of triptolide-loaded polymeric micelles. European Journal of Pharmaceutics and Biopharmaceutics 70: 741–748.
Yi, T., J. Wan, H. Xu, and X. Yang. 2008. Controlled poorly soluble drug release from solid self-microemulsifying formulations with high viscosity hydroxypropylmethylcellulose. European Journal of Pharmaceutical Sciences 34: 274–280.
Ying, W., Ying, L., Jian, W., and Qi, S. 2012. Characterisation and evaluation of self-microemulsifying drug delivery system of Brucea javanica oil. Micro and Nano Letters, IET, 7: 256–261.
Yu, Y.L., Y. Lu, X. Tang, and F.D. Cui. 2008. Formulation, preparation and evaluation of an intravenous emulsion containing Brucea javanica oil and coix seed oil for anti-tumor application. Biological and Pharmaceutical Bulletin 31: 673–680.
Zhang, H., J.Y. Yang, F. Zhou, L.H. Wang, W. Zhang, S. Sha, and C.F. Wu. 2011. Seed oil of Brucea javanica induces apoptotic death of acute myeloid leukemia cells via both the death receptors and the mitochondrial-related pathways. Evidence Based Complementary and Alternative Medicine 2011: 965016.
Acknowledgments
This work was supported by the Key Project of ‘13115’ Technology Innovation Project of Shaanxi Province (Grant No. 2009ZDKG-82) and the Science and Technology Planning Project of Guangdong Province (Grant No. 2011B031700016).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Shao, A., Chen, G., Jiang, N. et al. Development and evaluation of self-microemulsifying liquid and granule formulations of Brucea javanica oil. Arch. Pharm. Res. 36, 993–1003 (2013). https://doi.org/10.1007/s12272-013-0113-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0113-7