Abstract
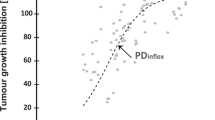
Over-expression of protein kinase CK2 is highly linked to the survival of cancer cells and the poor prognosis of patients with cancers. CX-4945, a potent and selective orally bioavailable ATP-competitive inhibitor of CK2, inhibits the oncogenic cellular events such as proliferation and angiogenesis, and the increase of tumor growth in mouse xenograft model. In this study, the pharmacokinetic information about CX-4945 was provided; at 10 μM, CX-4945 with high stability in human and rat liver microsome exhibited low percentage of inhibition (<10 %) in CYP450 isoforms (1A2, 2C19, 3A4), but considerable inhibition (~70 %) in CYP450 2C9 and 2D6. In hERG potassium channel inhibition assay, CX-4945 exhibited relatively low inhibition rate. Additionally, CX-4945 showed high MDCK cell permeability (>10 × 10−6 cm/s) and above 98 % of plasma protein binding in the rat. After intravenous administration, Vss (1.39 l/kg) and extremely low CL (0.08 l/kg/h) were observed. Moreover, orally administrated CX-4945 showed high bioavailability (>70 %) and these data might be related to the MDCK cell permeability results.



Similar content being viewed by others
References
Battistutta, R., E. De Moliner, S. Sarno, G. Zanotti, and L.A. Pinna. 2001. Structural features underlying selective inhibition of protein kinase CK2 by ATP site-directed tetrabromo-2-benzotriazole. Protein Science 10: 2200–2206.
Belardinelli, L., C. Antzelevitch, and M.A. Vos. 2003. Assessing predictors of drug-induced torsade de pointes. Trends in Pharmacological Sciences 24: 619–625.
Buerra, B., and O.G. Issinger. 2008. Protein kinase CK2 in human diseases. Current Medicinal Chemistry 15: 1870–1886.
Drygin, D., C.B. Ho, M. Omori, J. Bliesath, C. Proffitt, R. Rice, A. Siddiqui-Jain, S. O’Brien, C. Padgett, J.K. Lim, K. Anderes, W.G. Rice, and D. Ryckman. 2011. Protein kinase CK2 modulates IL-6 expression in inflammatory breast cancer. Biochemical and Biophysical Research Communications 415: 163–167.
Duncan, J.S., and D.W. Litchfield. 2008. Too much of a good thing: the role of protein kinase CK2 in tumorigenesis and prospects for therapeutic inhibition of CK2. Biochimica et Biophysica Acta 1784: 33–47.
Hung, M.S., Z. Xu, Y.C. Lin, J.H. Mao, C.T. Yang, P.J. Chang, D.M. Jablons, and L. You. 2009. Identification of hematein as a novel inhibitor of protein kinase CK2 from a natural product library. BMC Cancer 9: 135.
Kim, J., and S.H. Kim. 2012. Druggability of the CK2 inhibitor CX-4945 as an anticancer drug and beyond. Archives of Pharmacal Research 35: 1293–1296.
Landesman-Bollag, E., R. Romieu-Mourez, D.H. Song, G.E. Sonenshein, R.D. Cardiff, and D.C. Seldin. 2001. Protein kinase CK2 in mammary gland tumorigenesis. Oncogene 20: 3247–3257.
Laramas, M., D. Pasquier, O. Filhol, F. Ringeisen, J.L. Descotes, and C. Cochet. 2007. Nuclear localization of protein kinase CK2 catalytic subunit (CK2alpha) is associated with poor prognostic factors in human prostate cancer. European Journal of Cancer 43: 928–934.
Litchfield, D.W. 2003. Protein kinase CK2: Structure, regulation and role in cellular decisions of life and death. Biochemical Journal 369: 1–15.
Ljubimov, A.V., S. Caballero, A.M. Aoki, L.A. Pinna, M.B. Grant, and R. Castellon. 2004. Involvement of protein kinase CK2 in angiogenesis and retinal neovascularization. Investigative Ophthalmology & Visual Science 45: 4583–4591.
Lopez-Ramos, M., R. Prudent, V. Moucadel, C.F. Sautel, C. Barette, L. Lafanechere, L. Mouaawad, D. Grierson, F. Schmidt, J.C. Florent, P. Filippakopoulos, A.N. Bullock, S. Knapp, J.B. Reiser, and C. Cochet. 2010. New potent dual inhibitors of CK2 and Pim kinases: Discovery and structural insights. FASEB Journal 24: 3171–3185.
Mottet, D., S.P. Ruys, C. Demazy, M. Raes, and C. Michiels. 2005. Role for casein kinase 2 in the regulation of HIF-1 activity. International Journal of Cancer 117: 764–774.
O-charoenrat, P., V. Rusch, and S.G. Talbot. 2004. Casein kinase II alpha subunit and C1-inhibitor are independent predictors of outcome in patients with squamous cell carcinoma of the lung. Clinical Cancer Research 10: 5792–5803.
Pagono, M.A., J. Bain, Z. Kazimierczuk, S. Sarno, M.Di. Ruzzene, G. Maira, M. Elliott, A. Orzeszko, G. Cozza, F. Meggio, and L.A. Pinna. 2008. The selectivity of inhibitors of protein kinase CK2: An update. Biochemical Journal 415: 353–365.
Parhar, J., J. Morse, and B. Salh. 2007. The role of protein kinase CK2 in intestinal epithelial cell inflammatory signaling. International Journal of Colorectal Disease 22: 601–609.
Park, J.S., M.S. Kim, J.S. Song, S.H. Choi, B.H. Lee, J. Woo, J.H. Ahn, M.A. Bae, and S.H. Ahn. 2011. Dose-independent pharmacokinetics of a new peroxisome proliferator-activated receptor-γ agonist, KR-62980, in Sprague-Dawley rats and ICR mice. Archives of Pharmacal Research 34: 2051–2058.
Pierre, F., P.C. Chua, S.E. O’Brien, A. Siddiqui-Jain, P. Bourbon, M. Haddach, J. Michaux, J. Nagasawa, M.K. Schwaebe, E. Stefan, A. Vialettes, J.P. Whitten, T.K. Chen, L. Darjania, R. Stansfield, K. Anderes, J. Bliesath, D. Drygin, C. Ho, M. Omori, C. Proffitt, N. Streiner, K. Trent, W.G. Rice, and D.M. Ryckman. 2011. Discovery and SAR of 5-(3-chlorophenylamino)benzo[c][2,6]-naphthyridine-8-carboxylic acid (CX-4945), the first clinical stage inhibitor of protein kinase CK2 for the treatment of cancer. Journal of Medicinal Chemistry 5: 635–654.
Pistorius, K., G. Seitz, K. Remberger, and O.G. Issinger. 1991. Differential CKII activities in human colorectal mucosa, adenomas and carcinomas. Onkologie 14: 256–260.
Redfern, W.S., L. Carlsson, A.S. Davis, W.G. Lynch, I. Mackenzie, and S. Palethoroe. 2003. Relationship between preclinical cardiac electrophysiology, clinical QT interval prolongation and torsade de pointes for a broad range of drugs: Evidence for a provisional safety margin in drug development. Cardiovascular Research 58: 32–45.
Rendic, S., and F.J. Di Carlo. 1992. Human cytochrome P450 enzymes: A status report summarizing their reactions, substrates, inducers, and inhibitors. Drug Metabolism Reviews 29: 413–580.
Rodrigues, A.D. 1999. Integrated P450 reaction phenotyping: Attempting to bridge the gap between cDNA-expressed cytochromes P450 and native human liver microsomes. Biochemical Pharmacology 57: 465–480.
Ruiz-Garcia, A., M. Bermejo, A. Moss, and V.G. Casabo. 2008. Pharmacokinetics in drug discovery. Journal of Pharmaceutical Sciences 97: 654–690.
Ruzzene, M., and L.A. Pinna. 2010. Addition to protein kinase CK2: A common denominator of diverse cancer cells? Biochimica et Biophysica Acta 1804: 499–504.
Sandholt, I.S., B.B. Olsen, B. Guerra, and O.G. Issinger. 2009. Resorufin: A lead for a new protein kinase CK2 inhibitor. Anti-Cancer Drugs 20: 238–248.
Sarno, S., and L.A. Pinna. 2008. Protein kinase CK2 as a druggable target. Molecular BioSystems 4: 889–894.
Seldin, D.D., and P. Leder. 1995. Casein kinase II alpha transgene-induced murine lymphoma: Relation to theileriosis in cattle. Science 267: 894–897.
Siddiqui-Jain, A., D. Drygin, N. Streiner, P. Chua, F. Pierre, S.E. O’Brien, J. Blesath, M. Omori, N. Huser, C. Ho, C. Proffitt, M.K. Schwaebe, D.M. Ryckman, W.G. Rice, and K. Andres. 2010. CX-4945, an orally bioavailable selective inhibitor of protein kinase CK2, inhibits prosurvival and angiogenic signaling and exhibits antitumor efficacy. Cancer Research 70: 10288–10298.
Slaton, J.W., G.M. Unger, D.T. Sloper, A.T. Davis, and K. Ahmed. 2004. Introduction of apoptosis by antisense CK2 in human prostate cancer xenograft model. Molecular Cancer Research 2: 712–721.
Song, J.S., J.W. Chae, K.R. Lee, B.H. Lee, E.J. Choi, S.H. Ahn, K.I. Kwon, and M.A. Bae. 2011a. Pharmacokinetic characterization of decursinol derived from Angelica gigas Nakai in rats. Xenobiotica 41: 895–902.
Song, J.S., H.J. Rho, J.S. Park, M.S. Kim, B.H. Lee, J.W. Seo, D.J. Jeon, H.G. Cheon, S.H. Ahn, K.I. Kwon, and M.A. Bae. 2011b. Preclinical pharmacokinetics of PDE-310, a novel PDE4 inhibitor. Drug Metabolism and Pharmacokinetics 26: 192–200.
Stalter, G., S. Siemer, E. Becht, M. Ziegler, K. Remberger, and O.G. Issinger. 1994. Asymmetric expression of protein kinase CK2 subunits in human kidney tumors. Biochemical and Biophysical Research Communications 202: 141–147.
Thummel, K.E., and G. Wilkinson. 1998. In vitro and in vivo drug interactions involving human CYP3A. Annual Review of Pharmacology and Toxicology 38: 370–381.
van Golen, K.L., Z.F. Wu, X.T. Qiao, L. Bao, and S.D. Merajver. 2000. RhoC GTPase overexpression modulates induction of angiogenic factors in breast cells. Neoplasia 2: 418–425.
Wang, G., G. Unger, K.A. Ahmad, J.W. Slaton, and K. Ahmed. 2005. Downregulation of CK2 induces apoptosis in cancer cells—A potential approach to cancer therapy. Molecular and Cellular Biochemistry 274: 77–84.
Acknowledgments
This work was supported by KRICT’s project, SI-1304 funded by the Ministry of Knowledge Economy, Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Son, Y.H., Song, J.S., Kim, S.H. et al. Pharmacokinetic characterization of CK2 inhibitor CX-4945. Arch. Pharm. Res. 36, 840–845 (2013). https://doi.org/10.1007/s12272-013-0103-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0103-9