Abstract
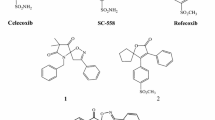
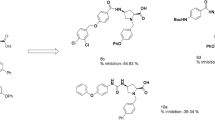
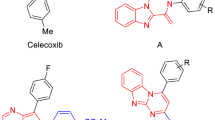
The synthesis of several 4-phenyl-5-pyridin-4-yl-2,3-dihydro-3H-1,2,4-triazole-3-thiones possessing N-2 Mannich bases or S-alkyl substituents, is reported. Several of them exhibited a low nanomolar COX enzyme inhibition activity. Most of the compounds showed inhibition of edema was similar to that evoked by celocoxib in animal model. Molecular docking studies of the compounds into the binding sites of COX-1 and COX-2 allowed us to shed light on the binding mode of these novel COX inhibitors.







Similar content being viewed by others
References
AbdelMoallem, S., F. Hadizadeh, and I. Yavar. 2011. New N-aryl-4-(methysulfony)aminobenzenesulfonamides as selective COX-2 inhibitors. Journal of Biological Sciences 11: 496–499.
Ali, A.M., G.E. Saber, N.M. Mahfouz, M.A. EI-Gendy, A.A. Radwan, and M.A. Hamid. 2007. Synthesis and three-dimensional qualitative structure selectivity relationship of 3,5-disubstituted-2,4-thiazolidinedione derivatives as COX-2 inhibitors. Archives of Pharmacal Research 30: 1186–1204.
Bayrak, H., A. Demirbas, S.A. Karaoglu, and N. Demirbas. 2009. Synthesis of some new 1,2,4-triazoles, their Mannich and Schiff bases and evaluation of their antimicrobial activities. European Journal of Medicinal Chemistry 44: 1057–1066.
Catella-Lawson, F., and L.J. Crofford. 2001. Cyclooxygenase inhibition and thrombogenicity. American Journal of Medicine 110: 28S–32S.
Clark, R.D. 2008. A ligand’s-eye view of protein binding. Journal of Computer-Aided Molecular Design 22: 507–521.
Dannhardt, G., and S. Laufer. 2000. Structural approaches to explain the selectivity of COX-2 inhibitors: is there a common pharmacophore? Current Medicinal Chemistry 7: 1101–1112.
DeGaetano, G., M.B. Donati, and C. Cerletti. 2003. Prevention of thrombosis and vascular inflammation: benefits and limitations of selective or combined COX-1, COX-2 and 5-LOX inhibitors. Trends in Pharmacological Sciences 24: 245–252.
DeWitt, D.L., E.A. el-Harith, S.A. Kraemer, M.J. Andrews, E.F. Yao, R.L. Armstrong, and W.L. Smith. 1990. The aspirin and heme-binding sites of ovine and murine prostaglandin endoperoxide synthases. Journal of Biological Chemistry 265: 5192–5198.
Greig, G.M., D.A. Francis, J.P. Falgueyret, M. Ouellet, M.D. Percival, P. Roy, C. Bayly, J.A. Mancini, and G.P. O’neill. 1997. The interaction of arginine 106 of human prostaglandin G/H synthase-2 with inhibitors is not a universal component of inhibition mediated by nonsteroidal anti-inflammatory drugs. Molecular Pharmacology 52: 829–838.
Jain, A.N. 2003. Surflex: fully automatic flexible molecular docking using a molecular similarity-based search engine. Journal of Medicinal Chemistry 46: 499–511.
Kulmacz, R.J., and W.E.M. Lands. 1983. Requirements for hydroperoxide by the cyclooxygenase and peroxidase activities of prostaglandin H synthase. Prostaglandins 25: 531–540.
Kulmacz, R.J. 1987. Prostaglandin G2 levels during reaction of prostaglandin H synthase with arachidonic acid. Prostaglandins 34: 225–240.
Kurumbail, R.G., A.M. Stevens, J.K. Gierse, J.J. McDonald, R.A. Stegeman, J.Y. Pak, D. Gildehaus, J.M. Miyashiro, T.D. Penning, K. Seibert, P.C. Isakson, and W.C. Stallings. 1996. Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 384: 644–648.
Labanauskas, L., V. Kalcas, E. Udrenaite, P. Gaidelis, A. Brukstus, and U. Dauksas. 2001. Synthesis of 3-(3,4-dimethoxyphenyl)-1H-1,2,4-triazole-5-thiol and 2-amino-5-(3,4-dimethoxyphenyl)-1,3,4-thiadiazole derivatives exhibiting anti-inflammatory activity. Pharmazie 56: 617–619.
Lecomte, M., O. Laneuville, C. Ji, D.L. DeWitt, and W.L. Smith. 1994. Acetylation of human prostaglandin endoperoxide synthase-2 (cyclooxygenase-2) by aspirin. Journal of Biological Chemistry 269: 13207–13215.
Limongell, V., M. Bonomi, L. Marinelli, F.L. Gervasio, A. Cavalli, E. Vovellino, and M. Parrinello. 2010. Molecular basis of cyclooxygenase enzymes (COXs) selective inhibition. PNAS 107: 5411–5416.
Loll, P.J., D. Picot, and R.M. Garavito. 1995. The structural basis of aspirin activity inferred from the crystal structure of inactivated prostaglandin H2 synthase. Natural Structural Biology 2: 637–643.
Luong, C., A. Miller, J. Barnett, J. Chow, C. Ramesha, and M.F. Browner. 1996. Flexibility of the NSAID binding site in the structure of human cyclooxygenase-2. Natural Structural Biology 3: 927–933.
Mancini, J.A., D. Riendeau, J.P. Falgueyret, P.J. Vickers, and G.P. O’Neill. 1995. Arginine 120 of prostaglandin G/H synthase-1 is required for the inhibition by nonsteroidal anti-inflammatory drugs containing a carboxylic acid moiety. Journal of Biological Chemistry 270: 29372–29377.
Meade, E.A., W.L. Smith, and D.L. DeWitt. 1993. Differential inhibition of prostaglandin endoperoxide synthase (cyclooxygenase) isozymes by aspirin and other non-steroidal anti-inflammatory drugs. Journal of Biological Chemistry 268: 6610–6614.
Moreau, A., P.N.P. Rao, and E.E. Knaus. 2006. Synthesis and biological evaluation of acyclic triaryl (Z)-olefins possessing a 3,5-di-tert-butyl-4-hydroxyphenyl pharmacophore: dual inhibitors of cyclooxygenases and lipoxygenases. Bioorganic & Medicinal Chemistry 14: 5340–5350.
Mukherjee, D., S.E. Nissen, and E.J. Topol. 2001. Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA 286: 954–959.
Muthas, D., Y.A. Sabnis, M. Lundborg, and A. Karlén. 2008. Is it possible to increase hit rates in structure-based virtual screening by pharmacophore filtering? An investigation of the advantages and pitfalls of post-filtering. Journal of Molecular Graphics and Modelling 26: 1237–1251.
Navidpour, L., H. Shafaroodi, K. Abdi, M. Amini, M.H. Ghahremani, A.R. Dehpour, and A. Shafiee. 2006. Design, synthesis, and biological evaluation of substituted 3-alkylthio-4,5-diaryl-4H-1,2,4-triazoles as selective COX-2 inhibitors. Bioorganic & Medicinal Chemistry 14: 2507–2517.
Penning, T.D., J.J. Tally, S.R. Bertenshaw, J.S. Carter, P.W. Collins, S. Docter, M.J. Graneto, L.F. Lee, J.W. Malecha, J.M. Miyashiro, D.J. Rogers, S.S. Rogier, G.D. Yu, E.G. Anderson, J.N. Burton, S.A. Cogburn, C.M. Gregory, R.S. Koboldt, W.E. Perkins, K. Seibert, A.W. Veenhuizen, Y.Y. Zhang, and P.C. Isakson. 1997. Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benze nesulfonamide (SC-58635, celecoxib). Journal of Medicinal Chemistry 40: 1347–1365.
Picot, D., P.J. Loll, and R.M. Garavito. 1994. The X-ray crystal structure of the membrane protein prostaglandin H2 synthase-1. Nature 367: 243–249.
Plount-Price, M.L., and W.L. Jorgensen. 2000. Analysis of binding affinities for celecoxib analogues with COX-1 and COX-2 from combined docking and Monte Carlo simulations and insight into the COX-2/COX-1 selectivity. Journal of the American Chemical Society 122: 9455–9466.
Pomarnacka, E., and A. Kornicka. 2001. Synthesis and in vitro anticancer and anti-HIV evaluation of new 2-mercaptobenzenesulfonamides. Il Farmaco 56: 571–577.
Praveen, R.P.N., A. Mohsen, L. Huiying, G.H. Amgad, and E.K. Edward. 2003. Design, synthesis and biological evaluation of 6-substituted-3-(4-methanesulfonylphenyl)-4-phenylpyran-2-ones: a novel class of diarylheterocyclic selective cyclooxygenase-2 inhibitors. Journal of Medicinal Chemistry 46: 4872–4882.
Ranatunge, R.R., R.A. Earl, D.S. Garvey, D.R. Janero, L.G. Letts, A.M. Martino, M.G. Murty, S.K. Richardson, D.J. Schwalb, D.V. Young, and I.S. Zemtseva. 2004. 3-(2-Methoxytetrahydrofuran-2-yl)pyrazoles: a novel class of potent, selective cyclooxygenase-2 (COX-2) inhibitors. Bioorganic & Medicinal Chemistry Letters 14: 6049–6052.
Riendeau, D., M.D. Percival, C. Brideau, C.S. Dube, D. Ethier, J.P. Falgueyret, R.W. Friesen, R. Gordon, G. Greig, J. Guay, Y. Girard, P. Prasit, R. Zamboni, I.W. Rodger, M. Gresser, A.W. Ford-Hutchinson, R.N. Young, and C.C. Chan. 2001. Etoricoxib (MK-0663): preclinical profile and comparison with other agents that selectively inhibit cyclooxygenase-2. Journal of Pharmacology and Experimental Therapeutics 296: 558–566.
Rome, L.H., and W.E. Lands. 1975. Structural requirements for time-dependent inhibition of prostaglandin biosynthesis by anti-inflammatory drugs. Proceedings of the National Academy of Sciences of the United States of America 72: 4863–4865.
Rouzer, C.A., and L.J. Marnett. 2003. Mechanism of free radical oxygenation of polyunsaturated fatty acids by cyclooxygenases. Chemical Reviews 103: 2239–2304.
Ruppert, J., W. Welch, and A.N. Jain. 1997. Automatic identification and representation of protein binding sites for molecular docking. Protein Science 6: 524–533.
Sano, H., T. Noguchi, A. Tanatani, Y. Hashimoto, and H. Miyachi. 2005. Design and synthesis of subtype-selective cyclooxygenase (COX) inhibitors derived from thalidomide. Bioorganic & Medicinal Chemistry 13: 3079–3091.
Silverstein, F.E., G. Faich, J.L. Goldstein, L.S. Simon, T. Pincus, A. Whelton, R. Makuch, G. Eisen, N.M. Agrawal, W.F. Stenson, A.M. Burr, W.W. Zhao, J.D. Kent, J.B. Lefkowith, K.M. Verburg, and G.S. Geis. 2000. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS Study: a randomized controlled trial. JAMA 284: 1247–1255.
Smith, W.L., and D.L. DeWitt. 1996. Prostaglandin endoperoxide H synthases-1 and -2. Advances in Immunology 62: 167–215.
Srimal, R.C., and B.N. Dhawan. 1974. Biological, toxicological and clinical evaluation. Indian Journal of Pharmacology 3: 4–13.
Sui, Z., J. Guan, M.P. Ferro, K. McCoy, M.P. Wachter, W.W. Murray, M. Singer, M. Steber, D.M. Ritchie, and D.C. Argentieri. 2000. 1,3-Diarylcycloalkanopyrazoles and diphenyl hydrazides as selective inhibitors of cyclooxygenase-2. Bioorganic & Medicinal Chemistry Letters 10: 601–614.
SYBYL Molecular Modelling Software. 2006. Tripos Inc., St. Louis, http://www.tripos.com. Accessed July 2010.
Talley, J.J., D.L. Brown, J.S. Carter, M.J. Graneto, C.M. Koboldt, J.L. Masferrer, W.E. Perkins, R.S. Rogers, A.F. Shaffer, Y.Y. Zhang, B.S. Zweifel, and K. Seibert. 2000. 4-[5-Methyl-3-phenylisoxazol-4-yl]-benzenesulfonamide, valdecoxib: a potent and selective inhibitor of COX-2. Journal of Medicinal Chemistry 43: 775–777.
Tanaka, Y., S.L. xWard, and W.L. Smith. 1987. Immunohistochemical and kinetic evidence for two different prostaglandin H-prostaglandin E isomerases in sheep vesicular gland microsomes. Journal of Biological Chemistry 262: 1374–1381.
Tozkoparan, B., E. Kupeli, E. Yesilada, S. Isik, M. Ozalp, and M. Ertan. 2005. Synthesis and evaluation of analgesic/anti-inflammatory and antimicrobial activities of 3-substituted-1,2,4-triazole-5-thiones. Arzneimittelforschung 55: 533–540.
Vera, K., Z. Lenka, W. Karel, K. Jamila, and M. Ute. 2004. Synthesis and antimycobacterial activity of 1,2,4-triazole 3-benzylsulfanyl derivatives. Il Farmaco 59: 279–288.
Winter, C.A., E.A. Risley, and G.W. Nuss. 1962. Carrageenin-induced edema in hind paw of the rat as an assay for antiinflammatory drugs. Proceedings of the Society for Experimental Biology and Medicine 111: 544–547.
Wolfe, M.M., D.R. Lichtenstein, and G. Singh. 1999. Gastrointestinal toxicity of nonsteroidal antiinflammatory drugs. New England Journal of Medicine 340: 1888–1899.
Wujec, M., and P. Paneth. 2008. Mechanism of 4-methyl-1,2,4-triazol-3-thione reaction with formaldehyde. Journal of Physical Organic Chemistry 21: 345–348.
Wujec, M., M. Pitucha, M. Dobosz, U. Kosikowska, and A. Malm. 2003. Synthesis and biological action of 1-substituted-3-R-4-phenyl-delta 2–1,2,4-triazoline-5-thion. Acta Poloniae Pharmaceutica 60: 451–456.
Zarghi, A., F.S. Javid, R. Ghodsi, O.G. Dadrass, B. Daraei, and M. Hedayati. 2011. Design, synthesis and biological evaluation of new 5,5-diarylhydantoin derivatives as selective cyclooxygenase-2 inhibitors. Scientia Pharmaceutica 79: 449–460.
Acknowledgments
The authors are grateful to the sponsorship of the Research Centre, College of Pharmacy and the Deanship of Scientific Research, King Saud University, Riyadh 11451, Saudi Arabia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radwan, A.A., elTahir, K.E.H. Synthesis and in-silico studies of some diaryltriazole derivatives as potential cyclooxygenase inhibitors. Arch. Pharm. Res. 36, 553–563 (2013). https://doi.org/10.1007/s12272-013-0078-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0078-6