Abstract
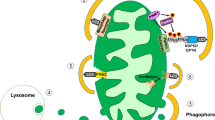
Autophagy plays a crucial role in cellular homeostasis through the degradation and recycling of organelles such as mitochondria or endoplasmic reticulum (ER) that are closely related to the pathogenesis of diabetes. In pancreatic β-cells producing insulin, autophagy helps maintain β-cell mass, structure and function. In mice with β-cell-specific deletion of Atg7 (autophagy-related 7), a critical autophagy gene, reduction of β-cell mass and pancreatic insulin content were observed together with impaired insulin secretory function. Because of such structural and functional defects, β-cell-specific Atg7-null mice showed hypoinsulinemia and hyperglycemia. However, those mice never developed diabetes. Obesity and lipids are physiological ER stressors that can precipitate β-cell dysfunction and insulin resistance. Recent studies showed that β-cell-specific Atg7-null mice, when bred with ob/ob mice, developed severe diabetes, suggesting that autophagy-deficient β-cells can handle basal metabolic stress but have problems dealing with increased metabolic stress. Thus, autophagy deficiency in β-cells could be a factor in the progression from obesity to diabetes due to an inappropriate response to obesity-induced ER stress. Autophagy also appears to play a role in the hypothalamic control of energy expenditure, appetite and body weight. Thus, autophagy is important to body and nutrient metabolism in many ways, and its dysregulation could contribute to the pathogenesis of metabolic disorders and diabetes.





Similar content being viewed by others
References
Back, S.H., D. Scheuner, J. Han, B. Song, M. Ribick, J. Wang, R.D. Gildersleeve, S. Pennathur, and R.J. Kaufman. 2009. Translation attenuation through eIF2alpha phosphorylation prevents oxidative stress and maintains the differentiated state in beta cells. Cell Metabolism 10: 13–26.
Bernales, S., K.L. Mcdonald, and P. Walter. 2006. Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biology 4: e423.
Coupe, B., Y. Ishii, M.O. Dietrich, M. Komatsu, T.L. Horvath, and S.G. Bouret. 2012. Loss of autophagy in pro-opiomelanocortin neurons perturbs axon growth and causes metabolic dysregulation. Cell Metabolism 15: 1–9.
Ebato, C., T. Uchida, M. Arakawa, M. Komatsu, T. Ueno, K. Komiya, K. Azuma, T. Hirose, K. Tanaka, E. Kominami, R. Kawamori, Y. Fujitani, and H. Watada. 2008. Autophagy is important in islet homeostasis and compensatory increase of beta cell mass in response to high-fat diet. Cell Metabolism 8: 325–332.
Hamasaki, M., and T. Yoshimor. 2010. Where do they come from? Insights from autophagosome formation. FEBS Letters 584: 1296–1301.
Jung, H.S., K.W. Chung, J.W. Kim, J. Kim, M. Komatsu, K. Tanaka, Y.H. Nguyen, T.M. Kang, K.H. Yoon, J.W. Kim, Y.T. Jeong, M.S. Han, M.K. Lee, K.W. Kim, J. Shin, and M.S. Lee. 2008. Loss of autophagy diminishes pancreatic beta cell mass and function with resultant hyperglycemia. Cell Metabolism 8: 318–324.
Kaushik, S., E. Arias, H. Kwon, N.M. Lopez, D. Athonvarangkull, S. Sahu, G.J. Schwartz, J.E. Pessin, and R. Singh. 2012. Loss of autophagy in hypothalamic POMC neurons impairs lipolysis. EMBO Reports 13: 258–265.
Kaushik, S., J.A. Rodriguez-Navarro, E. Arias, R. Kiffin, S. Sahu, G.J. Schwartz, A.M. Cuervo, and R. Singh. 2011. Autophagy in hypothalamic AgRP neurons regulates food intake and energy balance. Cell Metabolism 14: 173–183.
Kirkin, V., D.G. Mcewan, I. Novak, and I. Dikic. 2009. A role for ubiquitin in selective autophagy. Molecular Cell 34: 259–269.
Klionsky, D.J., and S.D. Emr. 2000. Autophagy as a regulated pathway of cellular degradation. Science 290: 1717–1721.
Komatsu, M., S. Waguri, M. Koike, Y.S. Sou, T. Ueno, T. Hara, N. Mizushima, J. Iwata, J. Ezaki, S. Murata, J. Hamazaki, Y. Nishito, S. Iemura, T. Natsume, T. Yanagawa, J. Uwayama, E. Warabi, H. Yoshida, T. Ishii, A. Kobayashi, M. Yamamoto, Z. Yue, Y. Uchiyama, E. Kominami, and K. Tanaka. 2007. Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell 131: 1149–1163.
Komatsu, M., S. Waguri, T. Ueno, J. Iwata, S. Murata, I. Tanida, J. Ezaki, N. Mizushima, Y. Ohsumi, Y. Uchiyama, E. Kominami, K. Tanaka, and T. Chiba. 2005. Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice. Journal of Cell Biology 169: 425–434.
Kroemer, G., G. Marino, and B. Levine. 2010. Autophagy and the integrated stress response. Molecular Cell 40: 280–293.
Levine, B., and G. Kroemer. 2008. Autophagy in the pathogenesis of disease. Cell 132: 27–42.
Lum, J.J., D.E. Bauer, M. Kong, M.H. Harris, C. Li, T. Lindsten, and C.B. Thompson. 2005. Growth factor regulation of autophagy and cell survival in the absence of apoptosis. Cell 120: 237–248.
Meng, Q., and D. Cai. 2011. Defective hypothalamic autophagy directs the central pathogenesis of obesity via the IkappaB kinase beta (IKKbeta)/NF-kappaB pathway. Journal of Biological Chemistry 286: 32324–32332.
Merksamer, P.I., A. Trusina, and F.R. Papa. 2008. Real-time redox measurements during endoplasmic reticulum stress reveal interlinked protein folding functions. Cell 135: 933–947.
Mizushima, N., and M. Komatsu. 2011. Autophagy: renovation of cells and tissues. Cell 147: 728–741.
Nakatogawa, H., K. Oh-Oka, and Y. Ohsumi. 2008. Lipidation of Atg8. Autophagy 4: 911–913.
Ozcan, U., Q. Cao, E. Yilmaz, A.H. Lee, N.N. Iwakoshi, E. Ozdelen, G. Tuncman, C. Gorgun, L.H. Glimcher, and G.S. Hotamisligil. 2004. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306: 457–461.
Park, S.W., Y. Zhou, J. Lee, A. Lu, C. Sun, J. Chung, K. Ueki, and U. Ozcan. 2010. The regulatory subunits of PI3K, p85a and p85b, interact with XBP-1 and increase its nuclear translocation. Nature Medicine 16: 429–437.
Quan, W., K.Y. Hur, Y. Lim, S.H. Oh, J.-C. Lee, H.C. Kim, G.-H. Kim, S.-H. Kim, H.L. Kim, M.-K. Lee, K.-W. Kim, J. Kim, M. Komatsu, and M.-S. Lee. 2012a. Autophagy deficiency in beta cells leads to compromised unfolded protein response and progression from obesity to diabetes in mice. Diabetologia 55: 392–403.
Quan, W., H.-K. Kim, E.-Y. Moon, S.S. Kim, C.S. Choi, M. Komatsu, Y.T. Jeong, M.-K. Lee, K.-W. Kim, M.-S. Kim, and M.-S. Lee. 2012b. Role of hypothalamic proopiomelanocortin neuron autophagy in the control of appetite and leptin response. Endocrinology 153: 1817–1826.
Scheuner, D., D. Vander Mierde, B. Song, D. Flamez, J.W. Creemers, K. Tsukamoto, M. Ribick, F.C. Schuit, and R.J. Kaufman. 2005. Control of mRNA translation preserves endoplasmic reticulum function in beta cells and maintains glucose homeostasis. Nature Medicine 11: 757–764.
Winnay, J.N., J. Boucher, M.A. Mori, K. Ueki, and C.R. Kahn. 2010. A regulatory subunit of phosphoinositide 3-kinase increases the nuclear accumulation of X-box-binding protein-1 to modulate the unfolded protein response. Nature Medicine 16: 438–445.
Acknowledgments
This study was supported by the Samsung Biomedical Research Institute Grant (SP1-B2-051-2) and the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Korea (A080967). Lee M-S is a recipient of a Global Research Laboratory Grant (K21004000003-10A0500-00310) and Bio & Medical Technology Development Program (20110019335) of the National Research Foundation of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Quan, W., Jung, H.S. & Lee, MS. Role of autophagy in the progression from obesity to diabetes and in the control of energy balance. Arch. Pharm. Res. 36, 223–229 (2013). https://doi.org/10.1007/s12272-013-0024-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0024-7