Abstract
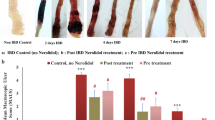
Ulcerative colitis (UC) is characterized by oxidative and nitrosative stress and neutrophil infiltration. In the present study, we aimed to investigate the therapeutic effect of ginsenoside Rd (GRd) in rats with 2,4,6-trinitrobenzenesulfonic acid (TNBS)-induced recurrent UC. After UC was twice-induced by intracolonic injection of TNBS, rats were intragastrically administered different doses of GRd per day for 7 days. The colonic lesions and inflammation were evaluated both histologically and biochemically. Compared with the TNBS group, GRd treatment facilitated recovery of pathologic changes in the colon after induction of recurrent UC, as evidenced by a significant reduction of colonic weight/length ratio and macroscopic and microscopic damage scores (p < 0.01). The myeloperoxidase and inducible nitric oxide synthase activities with malonyldialdehyde and nitric oxide levels in colonic tissues were significantly decreased in the GRd group compared with those in the TNBS group (p < 0.01). GRd treatment was associated with remarkably increased superoxide dismutase and glutathione peroxidase activities. Results showed a valuable effect of GRd against TNBS-induced recurrent UC by inhibiting neutrophil infiltration and promoting the antioxidant capacity of the damaged colonic tissue.
Similar content being viewed by others
References
Baynes, J. W. and Thorpe, S. R., Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes, 48, 1–9 (1999).
Beck, P. L., Xavier, R., Wong, J., Ezedi, I., Mashimo, H., Mizoguchi, A., Mizoguchi, E., Bhan, A. K., and Podolsky, D. K., Paradoxical roles of different nitric oxide synthase isoforms in colonic injury. Am. J. Physiol. Gastrointest. Liver Physiol., 286, G137–G147 (2004).
Bradley, P. P., Priebat, D. A., Christensen, R. D., and Rothstein, G., Measurement of cutaneous inflammation estimation of neutrophil content with an enzyme marker. J. Invest. Dermatol., 78, 206–209 (1982).
Cetinkaya, A., Bulbuloglu, E., Kantarceken, B., Ciralik, H., Kurutas, E. B., Buyukbese, M. A., and Gumusalan, Y., Effects of 1-carnitine on oxidant/antioxidant status in acetic acid-induced colitis. Dig. Dis. Sci., 51, 488–494 (2006).
Corsi, L., Effects of the novel non-peptidyl low molecular weight radical scavenger IAC in different models of inflammation: a new perspective in anti-inflammatory therapy. Curr. Med. Chem., 17, 3918–3924 (2010).
Di Sabatino, A., Rovedatti, L., Kaur, R., Spencer, J. P., Brown, J. T., Morisset, V. D., Biancheri, P., Leakey, N. A., Wilde, J. I., Scott, L., Corazza, G. R., Lee, K., Sengupta, N., Knowles, C. H., Gunthorpe, M. J., McLean, P. G., MacDonald, T. T., and Kruidenier, L., Targeting gut T cell Ca2+ release-activated Ca2+ channels inhibits T cell cytokine production and T-box transcription factor T-bet in inflammatory bowel disease. J. Immunol., 183, 3454–3462 (2009).
Elson, C. O., Cong, Y., Brandwein, S., Weaver, C. T., McCabe, R. P., Mähler, M., Sundberg, J. P., and Leiter, E. H., Experimental models to study molecular mechanisms underlying intestinal inflammation. Ann. NY Acad. Sci., 859, 85–95 (1998).
Fantini, M. C., Monteleone, G., and Macdonald, T. T., New players in the cytokine orchestra of inflammatory bowel disease. Inflamm. Bowel Dis., 13, 1419–1423. (2007).
Feske, S., Calcium signalling in lymphocyte activation and disease. Nat. Rev. Immunol., 7, 690–702 (2007).
Gálvez, J., Coelho, G., Crespo, M. E., Cruz, T., Rodríguez-Cabezas, M. E., Concha, A., Gonzalez, M., and Zarzuelo, A., Intestinal anti-inflammatory activity of morin on chronic experimental colitis in the rat. Aliment. Pharmacol. Ther., 15, 2027–2039 (2001).
Giris, M., Erbil, Y., Dogru-Abbasoglu, S., Yanik, B.T., Alis, H., Olgaç, V., and Toker, G. A., The effect of heme oxygenase-1 induction by glutamine on TNBS-induced colitis. Int. J. Colorectal Dis., 22, 591–599 (2007).
Giris, M., Depboylu, B., Dogru-Abbasoglu, S., Erbil, Y., Olgaç, V., Alis, H., Aykaç-Toker, G., and Uysal, M., Effect of taurine on oxidative stress and apoptosis-related protein expression in trinitrobenzene sulphonic acid-induced colitis. Clin. Exp. Immunol., 152, 102–110 (2008).
Gordon, J. N., Di Sabatino, A., and Macdonald, T. T., The pathophysiologic rationale for biological therapies in inflammatory bowel disease. Curr. Opin. Gastroenterol., 21, 431–437 (2005).
Guan, Y. Y., Zhou, J. G., Zhang, Z., Wang, G. L., Cai, B. X., Hong, L., Qiu, Q. Y., and He, H., Ginsenoside-Rd from panax notoginseng blocks Ca2+ influx through receptorand store-operated Ca2+ channels in vascular smooth muscle cells. Eur. J. Pharmacol., 548, 129–136 (2006).
Hagar, H. H., El-Medany, A., El-Eter, E., and Arafa, M., Ameliorative effect of pyrrolidinedithiocarbamate on acetic acid-induced colitis in rats. Eur. J. Pharmacol., 554, 69–77 (2007).
Han, M. and Fang, X. L., Difference in oral absorption of ginsenoside Rg1 between in vitro and in vivo models. Acta. Pharmacol. Sin., 27, 499–505 (2006).
Howard, A., Tahir, I., Javed, S., Waring, S. M., Ford, D., and Hirst, B. H., Glycine transporter GLYT1 is essential for glycine-mediated protection of human intestinal epithelial cells against oxidative damage. J. Physiol., 588, 995–1009 (2010).
Kakkar, P., Das, B., and Viswanathan, P. N., A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys., 21, 130–132 (1984).
Kimura, H., Miura, S., Shigematsu, T., Ohkubo, N., Tsuzuki, Y., Kurose, I., Higuchi, H., Akiba, Y., Hokari, R., Hirokawa, M., Serizawa, H., and Ishii, H., Increased nitric oxide production and inducible nitric oxide synthase activity in colonic mucosa of patients with active ulcerative colitis and Crohn’s disease. Dig. Dis. Sci., 42, 1047–1054 (1997).
Konturek, P. C., Brzozowski, T., Engel, M., Burnat, G., Gaca, P., Kwiecien, S., Pajdo, R., and Konturek, S. J., Ghrelin ameliorates colonic inflammation. Role of nitric oxide and sensory nerves. J. Physiol. Pharmacol., 60, 41–47 (2009).
Krawisz, J. E., Sharon, P., and Stenson, W. F., Quantitative assay for acute intestinal inflammation based on myeloperoxidase activity. Assessment of inflammation in rat and hamster models. Gastroenterology, 87, 1344–1350 (1984).
Kurutas, E. B., Cetinkaya, A., Bulbuloglu, E., and Kantarceken, B., Effects of antioxidant therapy on leukocyte myeloperoxidase and Cu/Zn-superoxide dismutase and plasma malondialdehyde levels in experimental colitis. Mediators Inflamm., 14, 390–394 (2005).
Li, H., Wu, W. K., Li, Z. J., Chan, K. M., Wong, C. C., Ye, C. G., Yu, L., Sung, J. J., Cho, C. H., and Wang, M., 2,3′,4,4′,5′-Pentamethoxy-trans-stilbene, a resveratrol derivative, inhibits colitis-associated colorectal carcinogenesis in mice. Br. J. Pharmacol., 160, 1352–1361 (2010).
Li, X. L., Cai, Y. Q., Qin, H., and Wu, Y. J., Therapeutic effect and mechanism of proanthocyanidins from grape seeds in rats with TNBS-induced ulcerative colitis. Can. J. Physiol. Pharmacol., 86, 841–849 (2008).
Lih-Brody, L., Powell, S. R., Collier, K. P., Reddy, G. M., Cerchia, R., Kahn, E., Weissman, G. S., Katz, S., Floyd, R. A., McKinley, M. J., Fisher, S. E., and Mullin, G. E., Increased oxidative stress and decreased antioxidant defences in mucosa of inflammatory bowel disease. Dig. Dis. Sci., 41, 2078–2086 (1996).
Liu, H., Yang, J., Du, F., Gao, X., Ma, X., Huang, Y., Xu, F., Niu, W., Wang, F., Mao, Y., Sun, Y., Lu, T., Liu, C., Zhang, B., and Li, C., Absorption and disposition of ginsenosides after oral administration of Panax notoginseng extract to rats. Drug Metab. Dispos., 37, 2290–2298 (2009).
Luik, R. M. and Lewis, R. S., New insights into the molecular mechanisms of store-operated Ca2+ signaling in T cells. Trends Mol. Med., 13, 103–107 (2007).
Loguercio, C., D’Argenio, G., Delle Cave, M., Cosenza, V., Della Valle, N., Mazzacca, G., and del Vecchio Blanco, C., Direct evidence of oxidative damage in acute and chronic phases of experimental colitis in rats. Dig. Dis. Sci., 41, 1204–1211 (1996).
Martín, M. C., Martinez, A., Mendoza, J. L., Taxonera, C., Díaz-Rubio, M., Fernández-Arquero M., de la Concha E. G., and Urcelay, E., Influence of the inducible nitric oxide synthase gene (NOS2A) on inflammatory bowel disease susceptibility. Immunogenetics, 59, 833–837 (2007).
McKenzie, S. J., Baker, M. S., Buffinton, G. D., and Doe, W. F., Evidence of oxidant-induced injury to epithelial cells during inflammatory bowel disease. J. Clin. Invest., 98, 136–141 (1996).
Murad-Regadas, S. M., Souza, M. H., Brito, G. A., Rodrigues, L. V., Regadas, F. S., and Vasconcelos, P. R., Effect of soluble fiber or fructooligosaccharide supplementation upon trinitrobenzenesulphonic acid induced colitis in rats. Acta. Cir. Bras., 21, 315–320 (2006).
Neurath, M., Fuss, I., and Strober, W., TNBS-colitis. Int. Rev. Immunol., 19, 51–62 (2000).
Ohkawa, H., Ohishi, N., and Yagi, K., Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 95, 351–358 (1979).
Orhan, H., Marol, S., Hepsen, I. f., and Sahin, G., Effects of some probable antioxidants on selenit-induced cataract formation and oxidative stress-related parameters in rats. Toxicology, 139, 219–232 (1999).
Palatka, K., Serfozo, Z., Veréb, Z., Hargitay, Z., Lontay, B., Erdodi, F., Bánfalvi, G., Nemes, Z., Udvardy, M., and Altorjay, I., Changes in the expression and distribution of the inducible and endothelial nitric oxide synthase in mucosal biopsy specimens of inflammatory bowel disease. Scand. J. Gastroenterol., 40, 670–680 (2005).
Peran, L., Sierra, S., Comalada, M., Lara-Villoslada, F., Bailón, E., Nieto, A., Concha, A., Olivares, M., Zarzuelo, A., Xaus, J., and Gálvez, J., A comparative study of the preventative effects exerted by two probiotics, Lactobacillus reuteri and Lactobacillus fermentum, in the trinitrobenzenesulfonic acid model of rat colitis. Br. J. Nutr., 97, 96–103 (2007).
Sun, F. F., Lai, P. S., Yue, G., Yin, K., Nagele, R. G., Tong, D. M., Krzesicki, R. F., Chin J. E., and Wong P. Y., Pattern of cytokine and adhesion molecule mRNA in hapten-induced relapsing colon inflammation in the rat. Inflammation, 25, 33–45 (2001).
Talero, E., Sánchez-Fidalgo, S., de la Lastra, C. A., Illanes M., Calvo, J. R., and Motilva, V., Acute and chronic responses associated with adrenomedullin administration in experimental colitis. Peptides, 29, 2001–2012 (2008).
Tamura, T., Cui X., Sakaguchi, N., and Akashi, M., Ginsenoside Rd prevents and rescues rat intestinal epithelial cells from irradiation-induced apoptosis. Food Chem. Toxicol., 9, 3080–3089 (2008).
Tüzün, A., Erdil, A., Inal, V., Aydin, A., Bagci, S., Yesilova, Z., Sayal, A., Karaeren, N., and Dagalp, K., Oxidative stress and antioxidant capacity in patients with inflammatory bowel disease. Clin. Biochem., 35, 569–572 (2002).
Wang, W., Wang, G. J., Xie, H. T., Sun, J. G., Zhao, S., Jiang, X. L., Li, H., Lv, H., Xu, M. J., and Wang, R., Determination of ginsenoside Rd in dog plasma by liquid chromatography-mass spectrometry after solid-phase extraction and its application in dog pharmacokinetics studies. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci., 852, 8–14 (2007).
Wang, Y. H., Yang, X. L., Wang, L., Cui, M. X., Cai, Y. Q., Li, X. L., and Wu, Y. J., Effects of proanthocyanidins from grape seed on treatment of recurrent ulcerative colitis in rats. Can. J. Physiol. Pharmacol., 88, 888–898 (2010).
Wu, C. F., Bi, X. L., Yang, J. Y., Zhan, J. Y., Dong, Y. X., Wang, J. H., Wang, J. M., Zhang, R, and Li, X., Differential effects of ginsenosides on NO and TNF-alpha production by LPS-activated N9 microglia. Int. Immunopharmacol., 7, 313–320 (2007).
Wohaieb, S. A. and Godin, D. V., Alterations in free radical tissue-defense mechanisms in streptozocin-induced diabetes in rat. Effects of insulin treatment. Diabetes, 36, 1014–1018 (1987).
Ye, R., Han, J., Kong, X., Zhao, L., Cao, R., Rao, Z., and Zhao, G., Protective effects of ginsenoside Rd on PC12 cells against hydrogen peroxide. Biol. Pharm. Bull., 31, 1923–1927 (2008).
Yokozawa, T., Satoh, A., and Cho, E. J., Ginsenoside-Rd attenuates oxidative damage related to aging in senescenceaccelerated mice. J. Pharm. Pharmacol., 6, 107–113 (2004).
Xie, H. T., Wang, G. J., Sun, J. G., Tucker, I., Zhao, X. C., Xie, Y. Y., Li, H., Jiang, X. L., Wang, R., Xu, M. J., and Wang, W., High performance liquid chromatographicmass spectrometric determination of ginsenoside Rg3 and its metabolites in rat plasma using solid-phase extraction for pharmacokinetic studies. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci., 818, 167–173 (2005).
Zhang, X., Zhang, D., Xu, J., Gu, J., and Zhao, Y., Determination of 25-OH-PPD in rat plasma by highperformance liquid chromatography-mass spectrometry and its application in rat pharmacokinetic studies. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci., 858, 65–70 (2007).
Zhu, F., Qian, J. M., and Pan, G. Z., The establishment of TNBS-induced experimental colitis. Zhongguo Yi Xue Ke Xue Yuan Xue Bao, 20, 271–278 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, XL., Guo, TK., Wang, YH. et al. Therapeutic effect of ginsenoside Rd in rats with TNBS-induced recurrent ulcerative colitis. Arch. Pharm. Res. 35, 1231–1239 (2012). https://doi.org/10.1007/s12272-012-0714-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0714-6