Abstract
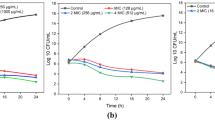
5-O-methylglovanon (5-O-MG) is a bioactive compound that was first isolated and characterized from Glycosmis plants. In this study, we found that chemically synthesized 5-O-MG has antimicrobial ability against eleven clinical ampicillin resistant Staphylococcus aureus and S. epidermidis isolates. The MICs of 5-O-MG against the S. aureus and S. epidermidis isolates were 12.5–50 μg/mL and 25–50 μg/mL, respectively. In combination with ampicillin, a synergistic interaction between 5-O-MG and ampicillin against the eleven resistant Staphylococcus isolates was observed, with fractional inhibitory concentration indices of 0.03–0125. Moreover, the anti-staphylococcal activity of 5-O-MG in combination with ampicillin was comparable with that of clavulanic acid in combination with ampicillin. The drug combination had no antagonistic effects when tested against any of the strains. Time-killing assays confirmed the synergy between 5-O-MG and ampicillin (p < 0.01). The combination of these two agents yielded greater than a 2 log10 cfu/mL decrease in comparison with 5-O-MG or ampicillin alone. These findings suggest that 5-O-MG is a promising compound with the potential for future anti-staphylococcal drug development.
Similar content being viewed by others
References
Arjanova, O. V., Prihoda, N. D., Yurchenko, L. V., Sokolenko, N. I., Vihrova, L. A., Pylypchuk, V. S., Frolov, V. M., and Kutsyna, G. A., Enhancement of efficacy of tuberculosis drugs with immunoxel (Dzherelo) in HIV-infected patients with active pulmonary tuberculosis. Immunotherapy, 1, 549–556 (2009).
Bagamboula, C. F., Uyttendaele, M., and Debevere, J., Antimicrobial effect of spices and herbs on Shigella sonnei and Shigella flexneri. J. Food Prot., 66, 668–673 (2003).
Baker, D. D. and Alvi, K. A., Small-molecule natural products: new structures, new activities. Curr. Opin. Biotechnol., 15, 576–583 (2004).
Baker, D. D., Chu, M., Oza, U., and Rajgarhia, V., The value of natural products to future pharmaceutical discovery. Nat. Prod. Rep., 24, 1225–1244 (2007).
Benton, B. M., Zhang, J. P., Bond, S., Pope, C., Christian, T., Lee, L., Winterberg, K. M., Schmid, M. B., and Buysse, J. M., Large-scale identification of genes required for full virulence of Staphylococcus aureus. J. Bacteriol., 186, 8478–8489 (2004).
CLSI., Performance standards for antimicrobial susceptibility testing. Clinical and Laboratory Standards Institute, 15th informational supplement, pp. M100–S115 (2005).
Credito, K., Lin, G., and Appelbaum, P. C., Activity of daptomycin alone and in combination with rifampin and gentamicin against Staphylococcus aureus assessed by timekill methodology. Antimicrob. Agents Chemother., 51, 1504–1507 (2007).
Darwish, R. M. and Aburjai, T. A., Effect of ethnomedicinal plants used in folklore medicine in Jordan as antibiotic resistant inhibitors on Escherichia coli. BMC Complement. Altern. Med., 10, 9 (2010).
Hendry, E. R., Worthington, T., Conway, B. R., and Lambert, P. A., Antimicrobial efficacy of eucalyptus oil and 1,8-cineole alone and in combination with chlorhexidine digluconate against microorganisms grown in planktonic and biofilm cultures. J. Antimicrob. Chemother., 64, 1219–1225 (2009).
Jang, J., Na, M., Thuong, P. T., Njamen, D., Mbafor, J. T., Fomum, Z. T., Woo, E. R., and Oh, W. K., Prenylated flavonoids with PTP1B inhibitory activity from the root bark of Erythrina mildbraedii. Chem. Pharm. Bull. (Tokyo), 56, 85–88 (2008).
Kim, Y. C., Kim, H. S., Wataya, Y., Sohn, D. H., Kang, T. H., Kim, M. S., Kim, Y. M., Lee, G. M., Chang, J. D., and Park, H., Antimalarial activity of lavandulyl flavanones isolated from the roots of Sophora flavescens. Biol. Pharm. Bull., 27, 748–750 (2004).
Klitgaard, J. K., Skov, M. N., Kallipolitis, B. H., and Kolmos, H. J., Reversal of methicillin resistance in Staphylococcus aureus by thioridazine. J. Antimicrob. Chemother., 62, 1215–1221 (2008).
Kluytmans-Vandenbergh, M. F. and Kluytmans, J. A., Community-acquired methicillin-resistant Staphylococcus aureus: current perspectives. Clin. Microbiol. Infect., 12Suppl 1, 9–15 (2006).
Lechner, D., Stavri, M., Oluwatuyi, M., Pereda-Miranda, R., and Gibbons, S., The anti-staphylococcal activity of Angelica dahurica (Bai Zhi). Phytochemistry, 65, 331–335 (2004).
Lukaseder, B., Vajrodaya, S., Hehenberger, T., Seger, C., Nagl, M., Lutz-Kutschera, G., Robien, W., Greger, H., and Hofer, O., Prenylated flavanones and flavanonols as chemical markers in Glycosmis species (Rutaceae). Phytochemistry, 70, 1030–1037 (2009).
Ma, X. M., Bao, G. Sh., Wan, J. M., Liao, D. J., Yin, Sh. F., Meng, X. Q., Zhou, G. K., Lu, X. M., and Li, H. Y., Therapeutic effects of Sophora moorcroftiana alkaloids in combination with albendazole in mice experimentally infected with protoscolices of Echinococcus granulosus. Braz. J. Med. Biol. Res., 40, 1403–1408 (2007).
Mulders, M. N., Haenen, A. P., Geenen, P. L., Vesseur, P. C., Poldervaart, E. S., Bosch, T., Huijsdens, X. W., Hengeveld, P. D., Dam-Deisz, W. D., Graat, E. A., Mevius, D., Voss, A., and Van De Giessen, A. W., Prevalence of livestockassociated MRSA in broiler flocks and risk factors for slaughterhouse personnel in The Netherlands. Epidemiol. Infect., 138, 743–755 (2010).
Na, M., Jang, J., Njamen, D., Mbafor, J. T., Fomum, Z. T., Kim, B. Y., Oh, W. K., and Ahn, J. S., Protein tyrosine phosphatase-1B inhibitory activity of isoprenylated flavonoids isolated from Erythrina mildbraedii. J. Nat. Prod., 69, 1572–1576 (2006).
Oo, T. Z., Cole, N., Garthwaite, L., Willcox, M. D., and Zhu, H., Evaluation of synergistic activity of bovine lactoferricin with antibiotics in corneal infection. J. Antimicrob. Chemother., 65, 1243–1251 (2010).
Petersen, P. J., Labthavikul, P., Jones, C. H., and Bradford, P. A., In vitro antibacterial activities of tigecycline in combination with other antimicrobial agents determined by chequerboard and time-kill kinetic analysis. J. Antimicrob. Chemother., 57, 573–576 (2006).
Sun, L., Sun, S., Cheng, A., Wu, X., Zhang, Y., and Lou, H., In vitro activities of retigeric acid B alone and in combination with azole antifungal agents against Candida albicans. Antimicrob. Agents Chemother., 53, 1586–1591 (2009).
van Cleef, B. A., Verkade, E. J., Wulf, M. W., Buiting, A. G., Voss, A., Huijsdens, X. W., van Pelt, W., Mulders, M. N., and Kluytmans, J. A., Prevalence of livestock-associated MRSA in communities with high pig-densities in The Netherlands. PLoS One, 5, e9385 (2010).
Voss, A., Loeffen, F., Bakker, J., Klaassen, C., and Wulf, M., Methicillin-resistant Staphylococcus aureus in pig farming. Emerg. Infect. Dis., 11, 1965–1966 (2005).
Vuong, C. and Otto, M., Staphylococcus epidermidis infections. Microbes Infect., 4, 481–489 (2002).
Yao, Y., Sturdevant, D. E., Villaruz, A., Xu, L., Gao, Q., and Otto, M., Factors characterizing Staphylococcus epidermidis invasiveness determined by comparative genomics. Infect. Immun., 73, 1856–1860 (2005).
Yu, Y. Y., Wang, H., Zhang, S. W., and Wang, B. E., Inhibition of methicillin-resistant Staphylococcus aureus by the compound Qingre granules. Chin. Med. J. (Engl), 123, 1017–1020 (2010).
Zuo, G. Y., Wang, G. C., Zhao, Y. B., Xu, G. L., Hao, X. Y., Han, J., and Zhao, Q., Screening of Chinese medicinal plants for inhibition against clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA). J. Ethnopharmacol., 120, 287–290 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, X., Jia, F., Liu, X. et al. In Vitro synergistic interaction of 5-O-methylglovanon and ampicillin against ampicillin resistant Staphylococcus aureus and Staphylococcus epidermidis isolates. Arch. Pharm. Res. 34, 1751–1757 (2011). https://doi.org/10.1007/s12272-011-1019-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-1019-x