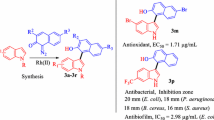
Abstract
Aromatic ester derivatives of ferulic acid where the phenolic hydroxyl is free (6a–d) or acetylated (5a–d) were evaluated for their antioxidant and antimicrobial properties. The superoxide radical scavenging capacity of compounds 5d and 6d–e (IC50 of 0.19, 0.27 and 0.20 mM, respectively) was found to be twice as active as α-tocopherol (IC50 = 0.51 mM). DPPH radical scavenging capacity was moderate and only found in compounds bearing free phenolic hydroxyl groups (6a–e). With regard to antimicrobial properties, compounds 6b and 6c displayed significant activity against Enterococcus faecalis (MICs = 16 μg/mL) and vancomycin-resistant E. faecalis (MIC for 6b, 32 and for 6c, 16 μg/mL). Compound 6c also demonstrated prominent activity against planktonic Staphylococcus aureus with a MIC value of <8 μg/mL and it inhibited bacterial biofilm formation by S. aureus with a MBEC value of <8 μg/mL, which was 64 and 128 times more potent than ofloxacin and vancomycin, respectively.
Similar content being viewed by others
References
Abdi-Ali, A., Mohammadi-Mehr, M., and Agha Alaei, Y., Bactericidal activity of various antibiotics against biofilmproducing Pseudomonas aeruginosa. Int. J. Antimicrob. Agents, 27, 196–200 (2006).
Ali, L., Khambaty, F., and Diachenko, G., Investigating the suitability of the Calgary Biofilm Device for assessing the antimicrobial efficacy of new agents. Bioresour. Technol., 97, 1887–1893 (2006).
Amorena, B., Gracia, E., Monzón, M., Leiva, J., Oteiza, C., Pérez, M., Alabart, J. L., and Hernández-Yago, J., Antibiotic susceptibility assay for Staphylococcus aureus in biofilms developed in vitro. J. Antimicrob. Chemother., 44, 43–55 (1999).
Anselmi, C., Centini, M., Granata, P., Sega, A., Buonocore, A., Bernini, A., and Facino, R. M., Antioxidant activity of ferulic acid alkyl esters in a heterophasic system: a mechanistic insight. J. Agric. Food Chem., 52, 6425–6432 (2004).
Bakalbassis, E. G., Chatzopoulou, A., Melissas, V. S., Tsimidou, M., Tsolaki, M., and Vafiadis, A., Ab initio and density functional theory studies for the explanation of the antioxidant activity of certain phenolic acids. Lipids, 36, 181–190 (2001).
Blois, M. S., Antioxidant determination by the use of stable free radical. Nature, 181, 1199–1200 (1958).
Bodet, C., Epifano, F., Genovese, S., Curini, M., and Grenier, D., Effects of 3-(4′-geranyloxy-3′-methoxyphenyl)-2-trans propenoic acid and its ester derivatives on biofilm formation by two oral pathogens, Porphyromonas gingivalis and Streptococcus mutans. Eur. J. Med. Chem., 43, 1612–1620 (2008).
Caliskan-Ergün, B., Süküroğlu, M., Coban, T., Banoğlu, E., and Suzen, S., Screening and evaluation of antioxidant activity of some pyridazine derivatives. J. Enzyme Inhib. Med. Chem., 23, 225–229 (2008).
Ceri, H., Olson, M. E., Stremick, C., Read, R. R., Morck, D., and Buret, A., The Calgary Biofilm Device: new technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol., 37, 1771–1776 (1999).
Costerton, J. W., Lewandowski, Z., Caldwell, D. E., Korber, D. R., and Lappin-Scott, H. M., Microbial biofilms. Annu. Rev. Microbiol., 49, 711–745 (1995).
Costerton, J. W., Stewart, P. S., and Greenberg, E. P., Bacterial biofilms: a common cause of persistent infections. Science, 284, 1318–1322 (1999).
Davies, D. G., Parsek, M. R., Pearson, J. P., Iglewski, B. H., Costerton, J. W., and Greenberg, E. P., The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science, 280, 295–298 (1998).
Del Soldato, P., Benedini, F., and Antognazza, P., Nitro derivatives as drugs for diseases having antiinflammatory basis. WO200230866 (2002).
Dunne, W. M. Jr., Bacterial adhesion: seen any good biofilms lately? Clin. Microbiol. Rev., 15, 155–166 (2002).
Galey, J. B. and Terranova, E., Monoesters and diesters of cinnamic acid or one of the derivatives tehreof and of vitamin C, process for the preparation thereof, and use as antioxidants in cosmetic, pharmaceutical or nutritional compositions. L’Oreal, Paris, France, US 5,536,500 (1996).
Graf, E., Antioxidant potential of ferulic acid. Free Radic. Biol. Med., 13, 435–448 (1992).
Halliwell, B., Free radicals and antioxidants: a personal view. Nutr. Rev., 52, 253–265 (1994).
Jayaprakasam, B., Vanisree, M., Zhang, Y., Dewitt, D. L., and Nair, M. G., Impact of alkyl esters of caffeic and ferulic acids on tumor cell proliferation, cyclooxygenase enzyme, and lipid peroxidation. J. Agric. Food Chem., 54, 5375–5381 (2006).
Jeong, Y. G., Jae, H. M., and Keun, H. P., Isolation and identification of 3-methoxy-4-hydroxybenzoic acid and 3-methoxy-4-hydroxycinnamic acid from hot water extracts of Hovenia dulcis Thumb and confirmation of their antioxidant and antimicrobial activity. Korean J. Food Sci. Technol., 32, 1403–1408 (2000).
Lo, H. H. and Chung, J. G., The effects of plant phenolics, caffeic acid, chlorogenic acid and ferulic acid on arylamine N-acetyltransferase activities in human gastrointestinal microflora. Anticancer Res., 19, 133–139 (1999).
Masuda, T., Yamada, K., Maekawa, T., Takeda, Y., and Yamaguchi, H., Antioxidant mechanism studies on ferulic acid: identification of oxidative coupling products from methyl ferulate and linoleate. J. Agric. Food Chem., 54, 6069–6074 (2006).
McCord, J. M. and Fridovich, I., Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J. Biol. Chem., 244, 6049–6055 (1969).
Melchior, M. B., Fink-Gremmels, J., and Gaastra, W., Comparative assessment of the antimicrobial susceptibility of Staphylococcus aureus isolates from bovine mastitis in biofilm versus planktonic culture. J. Vet. Med. B Infect. Dis. Vet. Public Health, 53, 326–332 (2006).
Michiyo, N., Shigeaki, I., Hisako, Y., Yoshihiko, O., Takuo, T., Eisaku, N., Asao, H., and Hisaji, T., Antimicrobial activities of synthetic ferulic acid derivatives. Food Preservation Science, 28, 183–188 (2002).
Mihara, M., Uchiyama, M., and Fukuzawa, K., Thiobarbituric acid value on fresh homogenate of rat as a parameter of lipid peroxidation in aging, CCl4 intoxication, and vitamin E deficiency. Biochem. Med., 23, 302–311 (1980).
Mori, H., Kawabata, K., Yoshimi, N., Tanaka, T., Murakami, T., Okada, T., and Murai, H., Chemopreventive effects of ferulic acid on oral and rice germ on large bowel carcinogenesis. Anticancer Res., 19, 3775–3778 (1999).
Murakami, A., Kadota, M., Takahashi, D., Taniguchi, H., Nomura, E., Hosoda, A., Tsuno, T., Maruta, Y., Ohigashi, H., and Koshimizu, K., Suppressive effects of novel ferulic acid derivatives on cellular responses induced by phorbol ester, and by combined lipopolysaccharide and interferon-γ. Cancer Lett., 157, 77–85 (2000).
Murakami, A., Nakamura, Y., Koshimizu, K., Takahashi, D., Matsumoto, K., Hagihara, K., Taniguchi, H., Nomura, E., Hosoda, A., Tsuno, T., Maruta, Y., Kim, H. W., Kawabata, K., and Ohigashi, H., FA15, a hydrophobic derivative of ferulic acid, suppresses inflammatory responses and skin tumor promotion: comparison with ferulic acid. Cancer Lett., 180, 121–129 (2002).
Nenadis, N., Zhang, H. Y., and Tsimidou, M. Z., Structureantioxidant activity relationship of ferulic acid derivatives: effect of carbon side chain characteristic groups. J. Agric. Food Chem., 51, 1874–1879 (2003).
Nomura, E., Hosoda, A., Morishita, H., Murakami, A., Koshimizu, K., Ohigashi, H., and Taniguchi, H., Synthesis of novel polyphenols consisted of ferulic and gallic acids, and their inhibitory effects on phorbol ester-induced Epstein-Barr virus activation and superoxide generation. Bioorg. Med. Chem., 10, 1069–1075 (2002).
Olson, M. E., Ceri, H., Morck, D. W., Buret, A. G., and Read, R. R., Biofilm bacteria: formation and comparative susceptibility to antibiotics. Can. J. Vet. Res., 66, 86–92 (2002).
Ou, S. and Kwok, K. C., Ferulic acid: pharmaceutical functions, preparation and applications in foods. J. Science Food Agricul., 84, 1261–1269 (2004).
Panizzi, L., Caponi, C., Catalano, S., Cioni, P. L., and Morelli, I., In vitro antimicrobial activity of extracts and isolated constituents of Rubus ulmifolius. J. Ethnopharmacol., 79, 165–168 (2002).
Proestos, C., Chorianopoulos, N., Nychas, G. J., and Komaitis, M., RP-HPLC analysis of the phenolic compounds of plant extracts. investigation of their antioxidant capacity and antimicrobial activity. J. Agric. Food Chem., 53, 1190–1195 (2005).
Rex, J. H., Alexander, B. D., Andes, D., Arthington-Skaggs, B., and Brown, S. D., Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeast Approved Standard, M27-A3. Clinical and Laboratory Standards Institute (CLSI), Clinical and Laboratory Standards Institute, 940 West Valley Road, Wayne, Pennsylvania, USA (2008).
Stewart, P. S., Theoretical aspects of antibiotic diffusion into microbial biofilms. Antimicrob. Agents Chemother., 40, 2517–2522 (1996).
Taniguchi, H., Hosoda, A., Tsuno, T., Maruta, Y., and Nomura, E., Preparation of ferulic acid and its application for the synthesis of cancer chemopreventive agents. Anticancer Res., 19, 3757–3761 (1999).
Vijay Kumar, H. and Naik, N., Synthesis and antioxidant properties of some novel 5H-dibenz[b,f]azepine derivatives in different in vitro model systems. Eur. J. Med. Chem., 45, 2–10 (2010).
Vuong, C. and Otto, M., Staphylococcus epidermidis infections. Microbes Infect., 4, 481–489 (2002).
Wikler, M. A., Cockerill, F. R., Bush, K., Dudley, M. N., and Eliopoulos, G. M., Performance Standards for Antimicrobial Susceptibility Testing, 19th Informational Supplement. CLSI document M100-S19. Clinical and Laboratory Standards Institute (CLSI), 940 West Valley Road, Wayne, Pennsylvania, pp. 19087–19898, (2009).
Wilson, M., Susceptibility of oral bacterial biofilms to antimicrobial agents. J. Med. Microbiol., 44, 79–87 (1996).
Xia, C. N., Li, H. B., Liu, F., and Hu, W. X., Synthesis of transcaffeate analogues and their bioactivities against HIV-1 integrase and cancer cell lines. Bioorg. Med. Chem. Lett., 18, 6553–6557 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ergün, B.Ç., Çoban, T., Onurdag, F.K. et al. Synthesis, antioxidant and antimicrobial evaluation of simple aromatic esters of ferulic acid. Arch. Pharm. Res. 34, 1251–1261 (2011). https://doi.org/10.1007/s12272-011-0803-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0803-y