Abstract
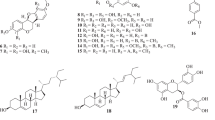
A new diterpene glycoside, tomentoside I (1), along with eleven known compounds, including the four coumarins, 4,5-dimethoxyl-7-methylcoumarin (2), 4,7-dimethoxyl-5-methylcoumarin (3), isofraxidin (4) and fraxidin (5) as well as the seven triterpenoids, oleanolic acid (6), oleanolic acid 3-O-α-L-arabinopyranoside (7), oleanolic acid 3-O-β-D-galactopyranosyl-(1→3)-β-D-glucopyranoside (8), hederagenin 3-O-α-L-arabinopyranoside (9), betulinic acid (10), 18-hydroxyursolic acid (11) and 2α,3β,23-trihydroxyurs-12-en-28-oic acid (12) were isolated from the ethanolic extract of the root of Anemone tomentosa and their chemical structures were elucidated by spectroscopic methods. The antimicrobial activities of compounds 1–12 were measured using the agar disc-diffusion method. Also, their antioxidant activities against 1,1-diphenyl-2-picrylhydrazyl (DPPH) were evaluated.
Similar content being viewed by others
References
Cao, P., Wu, F. E., and Ding, L. S., Advances in the studies on the chemical constituents and biologic activities for Anemone species. Nat. Prod. Res. Dev., 16 581–584 (2004).
Chemli, R., Babadjamian, A., Faure, R., Boukef, K., Balansard, G., and Vidal, E., Arvensoside A and B, triterpenoid saponins from Calendula arvensis. Phytochemistry, 26, 1785–1788 (1987).
Delectis Florae Reipublicae Popularis Sinicae, Agendae Academiae Sinicae Edits. Flora Reipublicae Popularis Sinicae. Science Press, Beijing, Vol. 28, pp. 29–31, (1980).
Denton, R. W., Harding, W. W., Anderson, C. I., Jacobs, H., McLean, S., and Reynolds, W. F., New diterpenes from Jatropha divaricata. J. Nat. Prod., 64, 829–831 (2001).
Fan, L., Lu, J., Wang, J., Cheng, W., Yao, Y., Liu, R., and Zhang, H., Two new triterpenoid saponins from rhizome of Anemone raddeana Regel. J. Nat. Med., 64, 50–54 (2010).
Gopalsamy, N., Gueho, J., Julien, H. R., Owadally, A. W., and Hostettmann, K., Molluscicidal saponins of Polyscias dichroostachya. Phytochemistry, 29, 793–795 (1990).
Karikas, G. A., Euerby, M. R., and Waigh, R. D., Constituents of the stems of Arbutus unedo. Planta Med., 53, 223–224 (1987).
Kawai, H., Kuroyanagi, M., Umehara, K., Ueno, A., and Satake, M., Studies on the saponins of Lonicera japonica Thunb. Chem. Pharm. Bull., 36, 4769–4775 (1988).
Kuo, Y. H., Yang, I. C., Chen, C. S., and Lin, Y. T., Five new sesquiterpenes from the heartwood of Juniperus squamata Lamb. J. Chin. Chem. Soc., 34, 125–134 (1987).
Li, Z. X., Li, X., Li, N., Li, W., and Sha, Y., Studies on the chemical constituents of the husks of Xanthoceras sorbifolia Bunge. J. Shenyang Pharm. Univ., 22, 271–273 (2005).
Liao, X., Peng, S. L., Li, B. G., Chen, Y. Z., and Ding, L. S., A new triterpenoid saponin from Anemone tomentosa. Chin. Chem. Lett., 10, 1035–1037 (1999).
Liu, X., Wang, C. H., Lin, M. X., Qin, S. Y., and Liu, Z. Y., Study on resources of medicinal plant Anemone L. in Chongqing. Resour. Dev. Mark., 24, 450–467 (2008).
Liu, Y. H., Liu, D. Y., and Liang, L., New medical advances of Anemone. Lishizhen Med. Mat. Med. Res., 10, 176–179 (1999).
Mahato, S. B. and Nandy, A. K., Triterpenoid saponins discovered between 1987 and 1989. Phytochemistry, 30, 1357–1390 (1991).
Mahato, S. B. and Kundu, A. P., 13C NMR spectra of pentacyclic triterpenoids—a compilation and some salient features. Phytochemistry, 37, 1517–1575 (1994).
NCCLS (The National Committee for Clinical Laboratory Standards), Performance standards for antimicrobial disk susceptibility test. Approved Standard: NCCLS document M2-A6 (6th ed.). Pennsylvania: Wayne, pp. 1–83, (1997).
Northwest Institute of Botany, Chinese Academy of Science. Flora of Qinling. Science Press, Beijing, pp. 278–279, (1974).
Numata, A., Takahashi, C., Miyamoto, T., Yoneda, M., and Yang, P. M., New triterpenes from a Chinese medicine, goreishi. Chem. Pharm. Bull., 38, 942–944 (1990).
Osborne, A. G., 13C NMR spectral studies of some methoxycoumarin derivatives. A re-assignment for citropten (limettin) and an examination of Peri-proximity effects for the methyl-methoxy and methoxy-methyl couples. Magn. Reson. Chem., 27, 348–354 (1989).
Pettit, G. R., Tan, R., Northen, J. S., Herald, D. L., Chapuis, J. C., and Pettit, R. K., Antineoplastic agents. 529. Isolation and structure of nootkastatins 1 and 2 from the Alaskan yellow cedar Chamaecyparis nootkatensis. J. Nat. Prod., 67, 1476–1482 (2004).
Tsukamoto, H., Hisada, S., and Nishibe, S., Coumarins from bark of Fraxinus japonica and F. mandshurica var. japonica. Chem. Pharm. Bull., 33, 4069–4073 (1985).
Wandji, J., Awanchiri, S. S., Fomum, Z. T., Tillequin, F., and Michel-Daniwicz, S., Prenylated isoflavonoids from Erythrina sensegalensis. Phytochemistry, 38, 1309–1313 (1995).
Wang, G., Chen, Y., Xu, J., Yuan, X., and Zhou, Y., Chemical constituents in the leaves of Acanthopanax sessiliflorus. Chin. Pharm. J., 32, 11–13 (1997).
Wang, J., Ma, J., Peng, S., Feng, J., and Ding, L., Chemical constituents and anfifeeding activity of Anemone tomentosa. Xibei Zhiwu Xuebao, 18, 643–644 (1998).
Wang, J. R., Peng, S. L., Wang, M. K., Feng, J. T., and Ding, L. S., Chemical constituents of the Anemone tomentosa root. Acta Bot. Sin., 41, 107–110 (1999).
Wang, W. S., Li, E. W., Jia, Z. J., Wang, C. M., and Zheng, R. L., A new anticancer diterpene from Juniperus squamata Lamb. J. Lanzhou Univ., 38, 121–122 (2002).
Wu, F. E. and Chu, T. T., Studies on the chemical constituents of the Chinese medicinal herb Anemone raddeana Regel. Chem. J. Chin. Univ., 4, 595–599 (1983).
Yoshida, T., Mori, K., Hatano, T., Okumura, T., Uehara, I., Komagoe, K., Fujita, Y., and Okuda, T., Studies on inhibition mechanism of autoxidation by tannins and flavonoids. V. Radical-scavenging effects of tannins and related polyphenols on 1,1-diphenyl-2-picrylhydrazyl radical. Chem. Pharm. Bull., 37, 1919–1921 (1989).
Zhao, J., Nakamura, N., Hattori, M., Yang, X. W., Komatsu, K., and Qiu, M. H., New triterpenoid saponins from the roots of Sinocrassula asclepiadea. Chem. Pharm. Bull., 52, 230–237 (2004).
Zheng, Z. B., Zhao, S. X., Deng, J. Z., Zhao, H. R., Ye, W. C., and Wang, M. S., Triterpenes from root bark of Shorea wangtianshuea. J. Chin. Pharm. Univ., 25, 262–264 (1994).
Zou, Z. J., Liu, H. X., and Yang, J. S., Chemical constituents and pharmacological activities of genus Anemone. Chin. Pharm. J., 39, 493–495 (2004).
Zuo, G., He, H., Hong, X., Shen, Y. M., and Hao, X. J., Chemical constituents of Spiraea japonica var. ovalifolia. Yunnan Zhi Wu Yan Jiu, 27, 101–106 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, HB., Zheng, XD., Jian, YF. et al. Constituents of the root of Anemone tomentosa . Arch. Pharm. Res. 34, 1097–1105 (2011). https://doi.org/10.1007/s12272-011-0707-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0707-x