Abstract
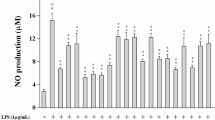
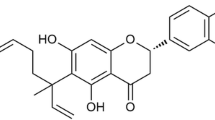
Flavonoids have biological activities including anti-allergic, anti-inflammatory, antimicrobial and anticancer activities shown from in vitro studies. Of these biological activities, the anti-inflammatory capacity of flavonoids has long been emphasized in Chinese medicine. In this study, I investigated that what flavonoid can be applied to the suppression of lipopolysaccharide (LPS)-induced inflammatory responses in macrophages among the four similar structure flavonoids. Eriodictyol was found to reduce nitric oxide (NO) production from LPS-stimulated Raw 264.7 cells in non-cytotoxic concentrations. Moreover, eriodictyol strongly suppressed the phagocytic activity of activated macrophages. Pre-treatment of Raw 264.7 cells with eriodictyol reduced the expression of mRNA and the secretion of pro-inflammatory cytokines. These inhibitory effects were found to be caused by blockage of nuclear factor kappa-light-chainenhancer of activated B cells (NF-κB) activation and phosphorylation of p38 mitogen-activated protein kinase (MAPK), extracellular signal-regulated kinases 1 and 2 (ERK1/2) and c-Jun Nterminal kinase (JNK).
Similar content being viewed by others
References
Amirzargar, A., Khosravi, F., Dianat, S., Hushmand, F., Maryousef, P., Foroushani, A. R., Lotfi, J., and Nikbin, B., Profile of cytokine gene polymorphisms in Iranian multiple sclerosis patients. Mult. Scler., 13, 253–255 (2007).
Bai, X. C., Lu, D., Liu, A. L., Zhang, Z. M., Li, X. M., Zou, Z.P., Zeng, W. S., Cheng, B. L., and Luo, S. Q., Reactive oxygen species stimulates receptor activator of NFkappaB ligand expression in osteoblast. J. Biol. Chem., 280, 17497–17506 (2005).
Beckman, J. S. and Koppenol, W. H., Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am. J. Physiol., 271, C1424–C1437 (1996).
Bian, Z. M., Elner, S. G., Yoshida, A., and Elner, V. M., Human RPE-monocyte co-culture induces chemokine gene expression through activation of MAPK and NIK cascade. Exp. Eye Res., 76, 573–583 (2003).
Brennan, F. M., Maini, R. N., and Feldmann, M., Role of proinflammatory cytokines in rheumatoid arthritis. Springer Semin. Immunopathol., 20, 133–147 (1998).
Butler, D. M., Malfait, A. M., Mason, L. J., Warden, P. J., Kollias, G., Maini, R. N., Feldmann, M., and Brennan, F. M., DBA/1 mice expressing the human TNF-alpha transgene develop a severe, erosive arthritis: characterization of the cytokine cascade and cellular composition. J. Immunol., 159, 2867–2876 (1997).
Cheon, B. S., Kim, Y. H., Son, K. S., Chang, H. W., Kang, S.S., and Kim, H. P., Effects of prenylated flavonoids and biflavonoids on lipopolysaccharide-induced nitric oxide production from the mouse macrophage cell line Raw 264.7. Planta Med., 66, 596–600 (2000).
Choo, Q. Y., Ho, P. C., and Lin, H. S., Histone deacetylase inhibitors: new hope for rheumatoid arthritis? Curr. Pharm. Des., 14, 803–820 (2008).
Comalada, M., Ballester, I., Bailón, E., Sierra, S., Xaus, J., Gálvez, J., de Medina, F. S., and Zarzuelo, A., Inhibition of pro-inflammatory markers in primary bone marrowderived mouse macrophages by naturally occurring flavonoids: analysis of the structure-activity relationship. Biochem. Pharmacol., 72, 1010–1021 (2006).
Dinarello, C. A., Biologic basis for interleukin-1 in disease. Blood, 87, 2095–2147 (1996).
Duval, D. L., Miller, D. R., Collier, J., and Billings, R. E., Characterization of hepatic nitric oxide synthase: identification as the cytokine-inducible form primarily regulated by oxidants. Mol. Pharmacol., 50, 277–284 (1996).
Havsteen B., Flavonoids, a class of natural products of high pharmacological potency. Biochem. Pharmacol., 32, 1141–1148 (1983).
Huang, D. W., Chung, C. P., Kuo, Y. H., Lin, Y. L., and Chiang, W., Identification of compounds in adlay (Coix lachryma-jobi L. var. ma-yuen Stapf) seed hull extracts that inhibit lipopolysaccharide-induced inflammation in Raw 264.7 macrophages. J. Agric. Food Chem., 57, 10651–10657 (2009).
Huk, I., Brovkovych, V., Nanobash Vili, J., Weigel, G., Neumayer, C., Partyka, L., Patton, S., and Malinski, T., Bioflavonoid quercetin scavenges superoxide and increases nitric oxide concentration in ischaemia-reperfusion injury: an experimental study. Br. J. Surg., 85, 1080–1085 (1998).
Ialenti, A., Ianaro, A., Moncada, S., and Di Rosa, M., Modulation of acute inflammation by endogenous nitric oxide. Eur. J. Pharmacol., 211, 177–182 (1992).
Ignarro, L. J., Regulation of cytosolic guanylyl cyclase by porphyrins and metalloporphyrins. Adv. Pharmacol., 26, 35–65 (1994).
Ito, H., Onoue, S., and Yoshida, T., Isoflavonoids from Belamcanda chinensis. Chem. Pharm. Bull. (Tokyo), 49, 1229–1231 (2001).
Johnson, G. L. and Lapadat, R., Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science, 298, 1911–1912 (2002).
Kean, R. B., Spitsin, S. V., Mikheeva, T., Scott, G. S., and Hooper, D. C., The peroxynitrite scavenger uric acid prevents inflammatory cell invasion into the central nervous system in experimental allergic encephalomyelitis through maintenance of blood-central nervous system barrier integrity. J. Immunol., 165, 6511–6518 (2000).
Kim, H. K., Cheon, B. S., Kim, Y. H., Kim, S. Y., and Kim, H. P., Effects of naturally occurring flavonoids on nitric oxide production in the macrophage cell line Raw 264.7 and their structure-activity relationships. Biochem. Pharmacol., 58, 759–765 (1999).
Kim, S. H., Kim, J., and Sharma, R. P., Inhibition of p38 and ERK MAP kinases blocks endotoxin-induced nitric oxide production and differentially modulates cytokine expression. Pharmacol. Res., 49, 433–439 (2004).
Kinne, R. W., Bräuer, R., Stuhlmüller, B., Palombo-Kinne, E., and Burmester, G. R., Macrophages in rheumatoid arthritis. Arthritis Res., 2, 189–202 (2000).
Kishimoto, T., Akira, S., Narazaki, M., and Taga, T., Interleukin-6 family of cytokines and gp130. Blood, 86, 1243–1254 (1995).
LaDuca, J. R. and Gaspari, A. A., Targeting tumor necrosis factor alpha. New drugs used to modulate inflammatory diseases. Dermatol. Clin., 19, 617–635 (2001).
Lawrence, T., Gilroy, D. W., Colville-Nash, P. R., and Willoughby, D. A., Possible new role for NF-kappaB in the resolution of inflammation. Nat. Med., 7, 1291–1297 (2001).
Lee, J. K., Kim, S. Y., Kim, Y. S., Lee, W. H., Hwang, D. H., and Lee, J. Y., Suppression of the TRIF-dependent signaling pathway of Toll-like receptors by luteolin. Biochem. Pharmacol., 77, 1391–1400 (2009).
Lee, S. Y. and Cho, J. Y., Inhibitory effects of honokiol on LPS and PMA-induced cellular responses of macrophages and monocytes. BMB Rep., 42, 574–579 (2009).
Liang, Y. C., Huang, Y. T., Tsai, S. H., Lin-Shiau, S. Y., Chen, C. F., and Lin, J. K., Suppression of inducible cyclooxygenase and inducible nitric oxide synthase by apigenin and related flavonoids in mouse macrophages. Carcinogenesis, 20, 1945–1952 (1999).
Lowenstein, C. J., Hill, S. L., Lafond-Walker, A., Wu, J., Allen, G., Landavere, M., Rose, N. R., and Herskowitz, A., Nitric oxide inhibits viral replication in murine myocarditis. J. Clin. Invest., 97, 1837–1843 (1996).
Middleton, E. Jr., Effect of plant flavonoids on immune and inflammatory cell function. Adv. Exp. Med. Biol., 439, 175–182 (1998).
Morgan, M. M., Clayton, C. C., and Heinricher, M. M., Dissociation of hyperalgesia from fever following intracerebroventricular administration of interleukin-1beta in the rat. Brain Res., 1022, 96–100 (2004).
Narumi, S., Finke, J. H., and Hamilton, T. A., Interferon gamma and interleukin 2 synergize to induce selective monokine expression in murine peritoneal macrophages. J. Biol. Chem., 265, 7036–7041 (1990).
Stichtenoth, D. O. and Frölich, J. C., Nitric oxide and inflammatory joint diseases. Br. J. Rheumatol., 37, 246–257 (1998).
Stuehr, D. J. and Marletta, M. A., Induction of nitrite/nitrate synthesis in murine macrophages by BCG infection, lymphokines, or interferon-gamma. J. Immunol., 139, 518–525 (1987).
Xagorari, A., Papapetropoulos, A., Mauromatis, A., Economou, M., Fotsis, T., and Roussos, C., Luteolin inhibits an endotoxin-stimulated phosphorylation cascade and proinflammatory cytokine production in macrophages. J. Pharmacol. Exp. Ther., 296, 181–187 (2001).
Xing, Z., Gauldie, J., Cox, G., Baumann, H., Jordana, M., Lei, X. F., and Achong, M. K., IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J. Clin. Invest., 101, 311–320 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.K. Anti-inflammatory effects of eriodictyol in lipopolysaccharidestimulated raw 264.7 murine macrophages. Arch. Pharm. Res. 34, 671–679 (2011). https://doi.org/10.1007/s12272-011-0418-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0418-3