Abstract
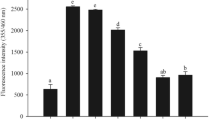
Advanced glycation end products (AGEs) play an important role in the development of chronic diabetic complications. Chlorogenic acid (CGA) is a phenolic compound formed by the esterification of caffeic and quinic acids. In this study, we evaluated the inhibitory effects of CGA against the formation of AGEs and AGEs protein cross-linking in vitro. An in vitro assay for glycation of bovine serum albumin by high glucose showed that CGA inhibited AGEs formation with an IC50 value of 148.32 μM and was found to be more effective than aminoguanidine, a well-known AGEs inhibitor (IC50; 807.67 μM). In an indirect AGE-ELISA assay, the CGA exhibited more potent inhibitory activity on the cross-linking of AGEs to collagen than aminoguanidine. In addition, the inhibitory effects of CGA on AGEs formation and on its cross-linking with collagen might be caused by its interactions with reactive decarbonyl compounds, such as methylglyoxal. These results suggest that CGA could be beneficial in the prevention of AGEs progression in patients with diabetes because CGA can attenuate AGEs deposition in glucose.
Similar content being viewed by others
References
Andrade-Cetto, A. and Wiedenfeld, H., Hypoglycemic effect of Cecropia obtusifolia on streptozotocin diabetic rats. J. Ethnopharmacol., 78, 145–149 (2001).
Baynes, J. W. and Thorpe, S. R., Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes, 48, 1–9 (1999).
Bourajjaj, M., Stehouwer, C. D., Van Hinsbergh, V. W., and Schalkwijk, C. G., Role of methylglyoxal adducts in the development of vascular complications in diabetes mellitus. Biochem. Soc. Trans., 31, 1400–1402 (2003).
Brownlee, M., Advanced protein glycosylation in diabetes and aging. Annu. Rev. Med., 46, 223–234 (1995).
Brownlee, M., Biochemistry and molecular cell biology of diabetic complications. Nature, 414, 813–820 (2001).
Degenhardt, T. P., Alderson, N. L., Arrington, D. D., Beattie, R. J., Basgen, J. M., Steffes, M. W., Thorpe, S. R., and Baynes, J. W., Pyridoxamine inhibits early renal disease and dyslipidemia in the streptozotocin-diabetic rat. Kidney Int., 61, 939–950 (2002).
Edelstein, D. and Brownlee, M., Mechanistic studies of advanced glycosylation end product inhibition by aminoguanidine. Diabetes, 41, 26–29 (1992).
Forbes, J. M., Soulis, T., Thallas, V., Panagiotopoulos, S., Long, D. M., Vasan, S., Wagle, D., Jerums, G., and Cooper, M. E., Renoprotective effects of a novel inhibitor of advanced glycation. Diabetologia, 44, 108–114 (2001).
Gugliucci, A., Bastos, D. H., Schulze, J., and Souza, M. F., Caffeic and chlorogenic acids in Ilex paraguariensis extracts are the main inhibitors of AGE generation by methylglyoxal in model proteins. Fitoterapia, 80, 339–344 (2009).
Haik, G. M., Jr., Lo, T. W., and Thornalley, P. J., Methylglyoxal concentration and glyoxalase activities in the human lens. Exp. Eye Res., 59, 497–500 (1994).
Herrera-Arellano, A., Aguilar-Santamaria, L., Garcia-Hernandez, B., Nicasio-Torres, P., and Tortoriello, J., Clinical trial of Cecropia obtusifolia and Marrubium vulgare leaf extracts on blood glucose and serum lipids in type 2 diabetics. Phytomedicine, 11, 561–566 (2004).
Jang, D. S., Yoo, N. H., Lee, Y. M., Yoo, J. L., Kim, Y. S., and Kim, J. S., Constituents of the flowers of Erigeron annuus with inhibitory activity on the formation of advanced glycation end products (AGEs) and aldose reductase. Arch. Pharm. Res., 31, 900–904 (2008).
Kiho, T., Usui, S., Hirano, K., Aizawa, K., and Inakuma, T., Tomato paste fraction inhibiting the formation of advanced glycation end-products. Biosci. Biotechnol. Biochem., 68, 200–205 (2004).
Kono, Y., Kashine, S., Yoneyama, T., Sakamoto, Y., Matsui, Y., and Shibata, H., Iron chelation by chlorogenic acid as a natural antioxidant. Biosci. Biotechnol. Biochem., 62, 22–27 (1998).
Lo, T. W., Selwood, T., and Thornalley, P. J., The reaction of methylglyoxal with aminoguanidine under physiological conditions and prevention of methylglyoxal binding to plasma proteins. Biochem. Pharmacol., 48, 1865–1870 (1994).
Loske, C., Gerdemann, A., Schepl, W., Wycislo, M., Schinzel, R., Palm, D., Riederer, P., and Munch, G., Transition metal-mediated glycoxidation accelerates cross-linking of beta-amyloid peptide. Eur. J. Biochem., 267, 4171–4178 (2000).
Miyata, T., Wada, Y., Cai, Z., Iida, Y., Horie, K., Yasuda, Y., Maeda, K., Kurokawa, K., and Van Ypersele De Strihou, C., Implication of an increased oxidative stress in the formation of advanced glycation end products in patients with end-stage renal failure. Kidney Int., 51, 1170–1181 (1997).
Nagaraj, R. H., Sarkar, P., Mally, A., Biemel, K. M., Lederer, M. O., and Padayatti, P. S., Effect of pyridoxamine on chemical modification of proteins by carbonyls in diabetic rats: characterization of a major product from the reaction of pyridoxamine and methylglyoxal. Arch. Biochem. Biophys., 402, 110–119 (2002).
Osawa, T. and Kato, Y., Protective role of antioxidative food factors in oxidative stress caused by hyperglycemia. Ann. N. Y. Acad. Sci., 1043, 440–451 (2005).
Pokupec, R., Kalauz, M., Turk, N., and Turk, Z., Advanced glycation endproducts in human diabetic and non-diabetic cataractous lenses. Graefes Arch. Clin. Exp. Ophthalmol., 241, 378–384 (2003).
Price, D. L., Rhett, P. M., Thorpe, S. R., and Baynes, J. W., Chelating activity of advanced glycation end-product inhibitors. J. Biol. Chem., 276, 48967–48972 (2001).
Rodriguez De Sotillo, D. V. and Hadley, M., Chlorogenic acid modifies plasma and liver concentrations of: cholesterol, triacylglycerol, and minerals in (fa/fa) Zucker rats. J. Nutr. Biochem., 13, 717–726 (2002).
Sato, T., Iwaki, M., Shimogaito, N., Wu, X., Yamagishi, S., and Takeuchi, M., TAGE (toxic AGEs) theory in diabetic complications. Curr. Mol. Med., 6, 351–358 (2006).
Sensi, M., Pricci, F., Andreani, D., and Di Mario, U., Advanced nonenzymatic glycation endproducts (AGE): their relevance to aging and the pathogenesis of late diabetic complications. Diabetes Res., 16, 1–9 (1991).
Shamsi, F. A., Partal, A., Sady, C., Glomb, M. A., and Nagaraj, R. H., Immunological evidence for methylglyoxal-derived modifications in vivo. Determination of antigenic epitopes. J. Biol. Chem., 273, 6928–6936 (1998).
Singh, R., Barden, A., Mori, T., and Beilin, L., Advanced glycation end-products: a review. Diabetologia, 44, 129–146 (2001).
Sohn, E., Kim, J., Kim, C. S., Kim, Y. S., Jang, D. S., and Kim, J. S., Extract of the aerial parts of Aster koraiensis reduced development of diabetic nephropathy via antiapoptosis of podocytes in streptozotocin-induced diabetic rats. Biochem. Biophys. Res. Commun., 391, 733–738 (2010).
Stitt, A., Gardiner, T. A., Alderson, N. L., Canning, P., Frizzell, N., Duffy, N., Boyle, C., Januszewski, A. S., Chachich, M., Baynes, J. W., and Thorpe, S. R., The AGE inhibitor pyridoxamine inhibits development of retinopathy in experimental diabetes. Diabetes, 51, 2826–2832 (2002).
Stitt, A. W., The role of advanced glycation in the pathogenesis of diabetic retinopathy. Exp. Mol. Pathol., 75, 95–108 (2003).
Svilaas, A., Sakhi, A. K., Andersen, L. F., Svilaas, T., Strom, E. C., Jacobs, D. R., Jr., Ose, L., and Blomhoff, R., Intakes of antioxidants in coffee, wine, and vegetables are correlated with plasma carotenoids in humans. J. Nutr., 134, 562–567 (2004).
Thornalley, P. J., Langborg, A., and Minhas, H. S., Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J., 344Pt 1, 109–116 (1999).
Thornalley, P. J., Yurek-George, A., and Argirov, O. K., Kinetics and mechanism of the reaction of aminoguanidine with the alpha-oxoaldehydes glyoxal, methylglyoxal, and 3-deoxyglucosone under physiological conditions. Biochem. Pharmacol., 60, 55–65 (2000).
Van Dam, R. M. and Feskens, E. J., Coffee consumption and risk of type 2 diabetes mellitus. Lancet, 360, 1477–1478 (2002).
Vasan, S., Zhang, X., Kapurniotu, A., Bernhagen, J., Teichberg, S., Basgen, J., Wagle, D., Shih, D., Terlecky, I., Bucala, R., Cerami, A., Egan, J., and Ulrich, P., An agent cleaving glucose-derived protein crosslinks in vitro and in vivo. Nature, 382, 275–278 (1996).
Vinson, J. A. and Howard III, T. B., Inhibition of protein glycation and advanced glycation end products by ascorbic acid and other vitamins and nutrients. J. Nutr. Biochem., 7, 659–663 (1996).
Wolff, S. P., Bascal, Z. A., and Hunt, J. V., Autoxidative glycosylation: free radicals and glycation theory. Prog. Clin. Biol. Res., 304, 259–275 (1989).
Wolff, S. P., Jiang, Z. Y., and Hunt, J. V., Protein glycation and oxidative stress in diabetes mellitus and ageing. Free Radic. Biol. Med., 10, 339–352 (1991).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kim, J., Jeong, IH., Kim, CS. et al. Chlorogenic acid inhibits the formation of advanced glycation end products and associated protein cross-linking. Arch. Pharm. Res. 34, 495–500 (2011). https://doi.org/10.1007/s12272-011-0319-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0319-5