Abstract
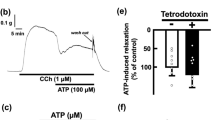
Lysophosphatidic acid (LPA) is a lipid mediator that is involved in many biological responses, such as, in the stimulation/inhibition of proliferation, in cell migration, antiapoptosis, tumor cell invasion, platelet aggregation, vascular remodeling, and neurotransmitter release. In addition, LPA indirectly enhances the contractility of smooth muscle. Furthermore, electric field stimulation (EFS) causes contractions of isolated cat esophageal smooth muscle and relaxations of isolated cat lower esophageal sphincter (LES). To test whether or not LPA enhances postsynaptically-mediated contraction in cat esophageal smooth muscle and LES, both types of muscle strips were stimulated with muscarinic agonists. However, no significant effects were observed, and therefore, to investigate whether LPA is involved in presynaptic signal transduction, cat esophageal smooth muscle and LES were pretreated with LPA and stimulated using EFS. LPA had no effect on EFS-induced contraction in esophageal smooth muscle but the EFSinduced LES relaxation was dose-dependently inhibited by LPA. To identify the LPA receptor subtype that inhibits EFS-induced LES relaxation, we used the specific LPA1/LPA3 antagonist Ki16425 and the LPA3 agonist OMPT. Ki16425 significantly blocked the inhibitory effect of LPA on EFS-induced relaxation, but OMPT did not enhance the effect of LPA. These results suggest that LPA inhibits EFS-induced relaxation in LES via LPA1 receptor-mediated signaling. It is well known that EFS-induced LES relaxation is related to the release of neurotransmitters, such as, nitric oxide, vasoactive intestinal polypeptide, and calcitonin gene-related peptide. We then investigated whether LPA selectively blocks NO-mediated signaling. Sodium nitroprusside-induced LES relaxation was found to be inhibited in the same manner as EFS-induced LES relaxation by LPA. This result suggests that LPA partially blocks NO uptake by presynaptic pathways, and thus, inhibits LES relaxation.
Similar content being viewed by others
References
Bell, N. J., Burget, D., Howden, C. W., Wilkinson, J., and Hunt, R. H., Appropriate acid suppression for the management of gastro-esophageal reflux disease. Digestion, 51, 59–67 (1992).
Biancani, P., Sohn, U. D., Rich, H. G., Harnett, K. M., and Behar, J., Signal transduction pathways in esophageal and lower esophageal sphincter circular muscle. Am. J. Med., 103, 23S–28S (1997).
Burnstock, G., Campbell, G., Satchell, D., and Smythe, A., Evidence that adenosine triphosphate or a related nucleotide is the transmitter substance released by non-adrenergic inhibitory nerves in the gut. Br. J. Pharmacol., 40, 668–688 (1970).
Contos, J. J., Ishii, I., and Chun, J., Lysophosphatidic acid receptors. Mol. Pharmacol., 58, 1188–1196 (2000).
Hama, K., Bandoh, K., Kakehi, Y., Aoki, J., and Arai, H., Lysophosphatidic acid (LPA) receptors are activated differentially by biological fluids: possible role of LPA-binding proteins in activation of LPA receptors. FEBS Lett., 523, 187–192 (2002).
Holian, O., Astumian, R. D., Lee, R. C., Reyes, H. M., Attar, B. M., and Walter, R. J., Protein Kinase C activity is altered in Hl60 cells exposed to 60 Hz AC dielectric fields. Bioelectromagnetics, 17, 504–509 (1996).
Ishii, I., Contos, J. J., Fukushima, N., and Chun, J., Functional comparisons of the lysophosphatidic acid receptors, LP(A1)/VZG-1/EDG-2, LP(A2)/EDG-4, and LP(A3)/EDG-7 in neuronal cell lines using a retrovirus expression system. Mol. Pharmacol., 58, 895–902 (2000).
Kadowaki, S., Chikumi, H., Yamamoto, H., Yoneda, K., Yamasaki, A., Sato, K., and Shimizu, E., Down-regulation of inducible nitric oxide synthase by lysophosphatidic acid in human respiratory epithelial cells. Mol. Cell. Biochem., 262, 51–59 (2004).
Lundberg, J. M., Pharmacology of cotransmission in the autonomic nervous system: integrative aspects on amines, neuropeptides, adenosine triphosphate, amino acids and nitric oxide. Pharmacol. Rev., 48, 113–178 (1996).
Mills, G. B. and Moolenaar, W. H., The emerging role of lysophosphatidic acid in cancer. Nat. Rev. Cancer, 3, 582–591 (2003).
Moolenaar, W. H., Lysophosphatidic acid, a multifunctional phospholipid messenger. J. Biol. Chem., 270, 12949–12952 (1995).
Moolenaar, W. H., Van Meeteren, L. A., and Giepmans, B. N., The ins and outs of lysophosphatidic acid signaling. Bioessays, 26, 870–881 (2004).
Noguchi, K., Ishii, S., and Shimizu, T., Identification of p2y9/GPR23 as a novel G-protein-coupled receptor for lysophosphatidic acid, structurally distant from the family. J. Biol. Chem., 278, 25600–25606 (2003).
Park, S. Y., Park, S. U., and Sohn, U. D., Regulators involved in the electrically stimulated response of feline esophageal smooth muscle. Pharmacology, 84, 346–355 (2009).
Park, S. Y., Shim, J. H., Kim, M., Sun, Y. H., Kwak, H. S., Yan, X., Choi, B. C., Im, C., Sim, S. S., Jeong, J. H., Kim, I. K., Min, Y. S., and Sohn, U. D., MLCK and PKC involvements via Gi and RhoA protein in contraction by the electrical field stimulation in feline esophageal smooth muscle. Korean J. Physiol. Pharmacol., 14, 29–35 (2010).
Sriwai, W., Zhou, H., and Murthy, K. S., Gq-dependent signalling by the lysophosphatidic acid receptor LPA3 in gastric smooth muscle: reciprocal regulation of MYPT1 phosphorylation by Rho kinase and cAMP-dependent PKA. Biochem. J., 411, 543–551 (2008).
Tigyi, G., Hong, L., Yakubu, M., Parfenova, H., Shibata, M., and Leffler, C. W., Lysophosphatidic acid alters cerebrovascular reactivity in piglets. Am. J. Physiol., 268, H2048–H2055 (1995).
Toews, M. L., Ustinova, E. E., and Schultz, H. D., Lysophosphatidic acid enhances contractility of isolated airway smooth muscle. J. Appl. Physiol., 83, 1216–1222 (1997).
Tokumura, A., Fukuzawa, K., and Tsukatani, H., Effects of synthetic and natural lysophosphatidic acids in the arterial blood pressure of different animal species. Lipids, 13, 572–574 (1978).
Tokumura, A., Fukuzawa, K., Yamada, S., and Tsukatani, H., Stimulatory effect of lysophosphatidic acids on uterine smooth muscles of non-pregnant rats. Arch. Int. Pharmacodyn. Ther., 245, 74–83 (1980).
Tokumura, A., Fukuzawa, K., and Tsukatani, H., Contractile actions of lysophosphatidic acids with a chemically-defined fatty acyl group on longitudinal muscle from guineapig ileum. J. Pharm. Pharmacol., 34, 514–516 (1982).
Tokumura, A., Yube, N., Fujimoto, H., and Tsukatani, H., Lysophosphatidic acids induce contraction of rat isolated colon by two different mechanism. J. Pharm. Pharmacol., 43, 774–778 (1991).
Uc, A., Oh, S. T., Murray, J. A., Clark, E., and Conklin, J. L., Biphasic relaxation of the opossum lower esophageal sphincter: roles of NO, VIP, and CGRP. Am. J. Physiol., 277, G548–G554 (1999).
Van Der Bend, R. L., Brunner, J., Jalink, K., Van Corven, E. J., Moolenaar, W. H., and Van Blitterswijk, W. J., Identification of a putative membrane receptor for the bioactive phospholipids, lysophosphatidic acid. EMBO J., 11, 2495–2501 (1992).
Yamboliev, I. A. and Mutafova-Yambolieva, V. N., PI3K and PKC contribute to membrane depolarization mediated by alpha2-adrenoceptors in the canine isolated mesenteric vein. BMC Physiol., 5, 9 (2005).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Lee, J.W., Kim, CH., Wang, YY. et al. Lysophosphatidic acid presynaptically blocks NO uptake during electric field stimulation-induced relaxation via LPA1 receptor in cat lower esophageal sphincter. Arch. Pharm. Res. 34, 169–176 (2011). https://doi.org/10.1007/s12272-011-0120-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-011-0120-5