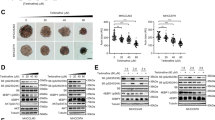
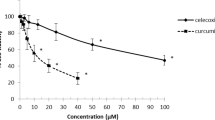
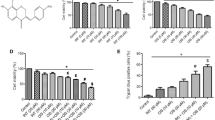
Abstract
The fungal cyclic peptide cyclosporin A (CsA) and fungal macrolide compound sanglifehrin A (SFA) have been developed as immunosuppressive drugs and both bind to cyclophilin A (CypA). CypA has been reported to be upregulated in diverse human cancers including hepatocellular carcinomas (HCC). CypA overexpression induces resistance to hypoxia and chemotherapeutic agents such as cisplatin in cancer cells. In this report, it was shown that CsA or SFA can synergistically increase apoptotic cell death in HCC cells, when combined with cisplatin. The increased cytotoxicity was accompanied by enhanced oxidative stress. The effects of CsA and SFA were due to inhibition of CypA activity. It was also shown that CsA and SFA abrogate cisplatin resistance as well as protection against hypoxia that is provided by CypA overexpression. Importantly, the synergistic effect of CsA or SFA with cisplatin was shown even in p53 defective Hep3B cells. Finally, overexpression of CypA was observed in human HCC tissues. In conclusion, CsA or SFA synergistically enhances cisplatin-induced apoptosis in HCC cells including the p53-defective Hep3B.
Similar content being viewed by others
References
Bassan, R., Lerede, T., Borleri, G., Chiodini, B., Rossi, A., Buelli, M., Rambaldi, A., Viero, P., and Barbui, T., Phase I trial with escalating doses of idarubicin and multidrug resistance reversal by short-course cyclosporin A, sequential high-dose cytosine arabinoside, and granulocyte colony-stimulating factor for adult patients with refractory acute leukemia. Haematologica, 87, 257–263 (2002).
Choi, K. J., Piao, Y. J., Lim, M. J., Kim, J. H., Ha, J., Choe, W., and Kim, S. S., Overexpressed cyclophilin A in cancer cells renders resistance to hypoxia-and cisplatin-induced cell death. Cancer Res., 67, 3654–3662 (2007).
Clarke, S. J., McStay, G. P., and Halestrap, A. P., Sanglifehrin A acts as a potent inhibitor of the mitochondrial permeability transition and reperfusion injury of the heart by binding to cyclophilin-D at a different site from cyclosporine A. J. Biol. Chem., 277, 34793–34799 (2002).
Cohen, G. M., Caspases: The executioners of apoptosis. Biochem. J., 326, 1–16 (1997).
Doyle, V., Virji, S., and Crompton, M., Evidence that cyclophilin-A protects cells against oxidative stress. Biochem. J., 341, 127–132 (1999).
Fehr, T., Kallen, J., Oberer, L., Sanglier, J. J., and Schilling, W., Sanglifehrins A, B, C and D, novel cyclophilin-binding compounds isolated from Streptomyces sp. A92-308110. II. Structure elucidation, stereochemistry and physicochemical properties. J. Antibiot (Tokyo)., 52, 474–479 (1999).
Fillies, T., Werkmeister, R., van Diest, P. J., Brandt, B., Joos, U., and Buerger, H., HIF1-alpha overexpression indicates a good prognosis in early stage squamous cell carcinomas of the oral floor. BMC Cancer, 5, 84 (2004).
Frei, U. A., Neumayer, H. H., Buchholz, B., Niese, D., and Mueller, E. A., Randomized, double-blind, one-year study of the safety and tolerability of cyclosporine microemulsion compared with conventional cyclosporine in renal transplant patients. International Sandimmun Neoral Study Group. Transplantation, 65, 1455–1460 (1998).
Handschumacher, R. E., Harding, M. W., Rice, J., Drugge, R. J., and Speicher, D. W., Cyclophilin: a specific cytosolic binding protein for cyclosporin A. Science, 226, 544–547 (1984).
Hinkle, R. T., Hodge, K. M., Cody, D. B., Sheldon, R. J., Kobilka, B. K., and Isfort, R. J., Skeletal muscle hypertrophy is mediated by a Ca2+-dependent calcineurin signalling pathway. Nature, 400, 576–581 (1999).
Hirsch-Ernst, K. I., Ziemann, C., Rustenbeck, I., and Kahl, G. F., Inhibitors of mdr1-dependent transport activity delay accumulation of the mdr1 substrate rhodamine 123 in primary rat hepatocyte cultures. Toxicology, 167, 47–57 (2001).
Ho, I. C., Kim, J. H., Rooney, J. W., Spiegelman, B. M., and Glimcher, L. H., A potential role for the nuclear factor of activated T cells family of transcriptional regulatory proteins in adipogenesis. Proc. Natl. Acad. Sci. U. S. A., 95, 15537–15541 (1998).
Hong, F., Lee, J., Song, J. W., Lee, S. J., Ahn, H., Cho, J. J., Ha, J., and Kim, S. S., Cyclosporin A blocks muscle differentiation by inducing oxidative stress and inhibiting the peptidyl-prolyl-cis-trans isomerase activity of cyclophilin A: cyclophilin A protects myoblasts from cyclosporin Ainduced cytotoxicity. FASEB J., 16, 1633–1635 (2002).
Hong, F., Lee, J., Piao, Y. J., Jae, Y. K., Kim, Y. J., Oh, C., Seo, J. S., Yun, Y. S., Yang, C. W., Ha, J., and Kim, S. S., Transgenic mice overexpressing cyclophilin A are resistant to cyclosporin A-induced nephrotoxicity via peptidylprolyl cis-trans isomerase activity. Biochem. Biophys. Res. Commun., 316, 1073–1080 (2004).
Howard, B. A., Zheng, Z., Campa, M. J., Wang, M. Z., Sharma, A., Haura, E., Herndon, J. E., 2nd, Fitzgerald, M. C., Bepler, G., and Patz, E. F. Jr., Translating biomarkers into clinical practice: prognostic implications of cyclophilin A and macrophage migratory inhibitory factor identified from protein expression profiles in non-small cell lung cancer. Lung Cancer, 46, 313–323 (2004).
Howard, B. A., Furumai, R., Campa, M. J., Rabbani, Z. N., Vujaskovic, Z., Wang, X. F., and Patz, E. F. Jr., Stable RNA interference-mediated suppression of cyclophilin A diminishes non-small-cell lung tumor growth in vivo. Cancer Res., 65, 8853–8860 (2005).
Ivery, M. T., Immunophilins: switched on protein binding domains? Med. Res. Rev., 20, 452–484 (2000).
Jeon, S. H., Piao, Y. J., Choi, K. J., Hong, F., Baek, H. W., Kang, I., Ha, J., and Kim, S. S., Chang, S. G., Prednisolone suppresses cyclosporin A-induced apoptosis but not cell cycle arrest in MDCK cells. Arch. Biochem. Biophys., 435, 382–392 (2005).
Jordan, P. and Carmo-Fonseca, M., Molecular mechanisms involved in cisplatin cytotoxicity. Cell. Mol. Life Sci., 57, 1229–1235 (2000).
Kartalou, M. and Essigmann, J. M., Mechanisms of resistance to cisplatin. Mutat. Res., 478, 23–43 (2001).
Li, M., Wang, H., Li, F., Fisher, W. E., Chen, C., and Yao, Q., Effect of cyclophilin A on gene expression in human pancreatic cancer cells. Am. J. Surg., 190, 739–745 (2005).
Llovet, J. M., Bustamante, J., Castells, A., Vilana, R., Ayuso Mdel, C., Sala, M., Bru, C., Rodes, J., and Bruix, J., Natural history of untreated nonsurgical hepatocellular carcinoma: rationale for the design and evaluation of therapeutic trials. Hepatology, 29, 62–67 (1999).
Llovet, J. M., Burroughs, A., and Bruix, J., Hepatocellular carcinoma. Lancet, 362, 1907–1917 (2003).
Lum, B. L., Kaubisch, S., Fisher, G. A., Brown, B. W., and Sikic, B. I., Effect of high-dose cyclosporine on etoposide pharmacodynamics in a trial to reverse P-glycoprotein (MDR1 gene) mediated drug resistance. Cancer Chemother. Pharmacol., 45, 305–311 (2000).
Musaro, A., McCullagh, K. J., Naya, F. J., Olson, E. N., and Rosenthal, N., IGF-1 induces skeletal myocyte hypertrophy through calcineurin in association with GATA-2 and NF-ATc1. Nature, 400, 581–585 (1999).
Nakagawa, M., Sakamoto, N., Tanabe, Y., Koyama, T., Itsui, Y., Takeda, Y., Chen, C. H., Kakinuma, S., Oooka, S., Maekawa, S., Enomoto, N., and Watanabe, M., Suppression of hepatitis C virus replication by cyclosporin A is mediated by blockade of cyclophilins. Gastroenterology, 129, 1031–1041 (2005).
Nakahara, C., Nakamura, K., Yamanaka, N., Baba, E., Wada, M., Matsunaga, H., Noshiro, H., Tanaka, M., Morisaki, T., and Katano, M., Cyclosporin-A enhances docetaxel-induced apoptosis through inhibition of nuclear factorkappaB activation in human gastric carcinoma cells. Clin. Cancer Res., 9, 5409–5416 (2003).
Orth, K., Chinnaiyan, A. M., Garg, M., Froelich, C. J., and Dixit, V. M., The CED-3/ICE-like protease Mch2 is activated during apoptosis and cleaves the death substrate lamin A. J. Biol. Chem., 271, 16443–16446 (1996).
Parkin, D. M., Pisani, P., and Ferlay, J., Estimates of the worldwide incidence of 25 major cancers in 1990. Int. J. Cancer, 80, 827–884 (1999).
Pyrzynska, B., Serrano, M., Martinez, A., and Kaminska, B., Tumor suppressor p53 mediates apoptotic cell death triggered by cyclosporin A. J. Biol. Chem., 277, 14102–14108 (2002).
Rey, O., Baluda, M. A., and Park, N. H., Differential gene expression in neoplastic and human papillomavirus-immortalized oral keratinocytes. Oncogene, 18, 827–831 (1999).
Seko, Y., Fujimura, T., Taka, H., Mineki, R., Murayama, K., and Nagai, R., Hypoxia followed by reoxygenation induces secretion of cyclophilin A from cultured rat cardiac myocytes. Biochem. Biophys. Res. Commun., 317, 162–168 (2004).
Shen, J., Person, M. D., Zhu, J., Abbruzzese, J. L., and Li, D., Protein expression profiles in pancreatic adenocarcinoma compared with normal pancreatic tissue and tissue affected by pancreatitis as detected by two-dimensional gel electrophoresis and mass spectrometry. Cancer Res., 64, 9018–9026 (2004).
Siddik, Z. H., Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene, 22, 7265–7279 (2003).
Sigal, N. H., Dumont, F., Durette, P., Siekierka, J. J., Peterson, L., Rich, D. H., Dunlap, B. E., Staruch, M. J., Melino, M. R., Koprak, S. L., Williams, D., Witzel, B., and Pisano, J. M., Is cyclophilin involved in the immunosuppressive and nephrotoxic mechanism of action of cyclosporin A? J. Exp. Med., 173, 619–628 (1991).
Wenger, R. M., Cyclosporine: conformation and analogues as tools for studying its mechanism of action. Transplant. Proc., 2, 313–318 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J. Novel combinational treatment of cisplatin with cyclophilin a inhibitors in human heptocellular carcinomas. Arch. Pharm. Res. 33, 1401–1409 (2010). https://doi.org/10.1007/s12272-010-0914-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0914-x