Abstract
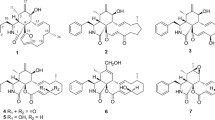
Nine 10-(indol-3-yl)-[13]cytochalasans such as a new chaetoglobosin V (1); two new natural products, prochaetoglobosin III (2) and prochaetoglobosin IIIed (3); six known chaetoglobosins B-D (4–6), F (7), and G (8) and isochaetoglobosin D (9) in addition to two known sterols, 24(R)-5α,8α-epidioxyergosta-6–22-diene-3β-ol (10) and ergosterol (11), were isolated from the fungus Chaetomium elatum ChE01. The structures of these compounds were elucidated by spectroscopic methods. Compounds 1–9 showed cytotoxicity against the human breast cancer (IC50 2.54–21.29 μM) and cholangiocarcinoma cell lines (IC50 3.41–86.95 μM).
Similar content being viewed by others
References
Bashyal, B. P., Wijeratne, E. M. K., Faeth, S. H., and Gunatilaka, A. A. L., Globosumones A-C, cytotoxic orsellinic acid esters from the Sonoran Desert endophytic fungus Chaetomium globosum. J. Nat. Prod., 68, 724–728 (2005).
Bok, J. W., Lermer, L., Chilton, J., Klingeman, H. G., and Towers, G. H., Antitumor sterols from the mycelia of Cordyceps sinensis. Phytochemistry, 51, 891–898 (1999).
Brewer, D., Jerram, W. A., and Taylor, A., Production of cochliodinol and a related metabolite by Chaetomium species. Can. J. Microbiol., 14, 861–866 (1968).
Buranrat, B., Prawan, A., Sripa, B., and Kukongviriyapan, V., Inflammatory cytokines suppress arylamine N-acetyltransferase 1 in cholangiocarcinoma cells. World J. Gastroenterol., 13, 6219–6225 (2007).
Ding, G., Song, Y. C., Chen, J. R., Xu, C., Ge, H. M., Wang, X. T., and Tan, R. X., Chaetoglobosin U, a cytochalasan alkaloid from endophytic Chaetomium globosum IFBE019. J. Nat. Prod., 69, 302–304 (2006).
Kanokmedhakul, S., Kanokmedhakul, K., Phonkerd, N., Soytong, K., Kongsaeree, P., and Suksamrarn, A., Antimycobacterial anthraquinone-chromanone compound and diketopiperazine alkaloid from the fungus Chaetomium globosum KMITL-N0802. Planta Med., 68, 834–836 (2002).
Kanokmedhakul, S., Kanokmedhakul, K., Nasomjai, P., Soytong, K., Isobe, M., Kongsaeree, P., Prabpai, S., and Suksamrarn, A., Antifungal azaphilones from the fungus Chaetomium cupreum CC3003. J. Nat. Prod., 69, 891–895 (2006).
Khumkomkhet, P., Kanokmedhakul, S., Kanokmedhakul, K., Hahnvajanawong, C., and Soytong, K., Antimalarial and cytotoxic depsidones from the fungus Chaetomium brasiliense. J. Nat. Prod., 72, 1487–1491 (2009).
Li, G. Y., Li, B. G., Yang, T., Liu, G. Y., and Zhang, G. L., Secondary metabolites from the fungus Chaetomium brasiliense. Helv. Chim. Acta, 91, 124–129 (2008).
Oh, H., Swenson, D. C., Gloer, J. B., Wicklow, D. T., and Dowd, P. F., Chaetochalasin A: A new bioactive metabolite from Chaetomium brasiliense. Tetrahedron Lett., 39, 7633–7636 (1998).
Oikawa, H., Murakami, Y., and Ichihara, A., New plausible precursors of chaetoglobosin A accumulated by treatment of Chaetomium subaffine with cytochrome P-450 inhibitors. Tetrahedron Lett., 35, 4533–4536 (1991).
Oikawa, H., Murakami, Y., and Ichihara, A., Useful approach to find the plausible biosynthetic precursors of secondary metabolites using P-450 inhibitors: postulated intermediates of chaetoglobosin A. J. Chem. Soc. Perkin Trans. I, 2949–2953 (1992).
Petcharat, V. and Soytong, K., Cheatomium in soil under para rubber. Songklanakarin J. Sci. Technol., 13, 129–132 (1991).
Phonkerd, N., Kanokmedhakul, S., Kanokmedhakul, K., Soytong, K., Prabpai, S., and Kongsaeree, P., Bis-spiro-Azaphilones and azaphilones from the fungi Chaetomium cochliodes VTh01 and C. cochliodes CTh05. Tetrahedron, 64, 9636–9645 (2008).
Pornsuriya, C., Lin, F. C., Kanokmedhakul, S., and Soytong, K., New record of Chaetomium species isolated from soil under pineapple plantation in Thailand. J. Agric. Tech., 4, 91–103 (2008).
Prawan, A., Buranrat, B., Kukongviriyapan, U., Sripa, B., and Kukongviriyapan, V., Inflammatory cytokines suppress NAD(P)H:quinone oxidoreductase-1 and induce oxidative stress in cholangiocarcinoma cells. J. Cancer Res. Clin. Oncol., 135, 515–522 (2009).
Probst, A. and Tamm, C., 19-O-Acetylchaetoglobosin B and 19-O-acetylchaetoglobosin D, two new metabolites of Chaetomium globosum. Helv. Chim. Acta, 64, 2056–2064 (1981).
Safe, S. and Taylor, A., Sporidesmins. XIII. Ovine Ill-thrift in Nova Scotia. III. Characterization of chetomin, a toxic metabolite of Chaetomium cochliodes and Chaetomium globosum. J. Chem. Soc. Perkin Trans. I, 472–479 (1972).
Sekita, S., Yoshihira, K., Natori, S., and Kuwano, H., Structures of chaetoglobosins C, D, E, and F, cytotoxic indol-3-yl-[13]cytochalasans from Chaetomium globosum. Tetrahedron Lett., 17, 1351–1354 (1976).
Sekita, S., Yoshihira, K., Natori, S., and Kuwano, H., Chaetoglobosins, cytotoxic 10-(indol-3-yl)-[13]cytochalasans from Chaetomium spp. III. Structures of chaetoglobosins C, E, F, G, and J. Chem. Pharm. Bull., 30, 1629–1638 (1982).
Sekita, S., Yoshihira, K., and Natori, S., Chaetoglobosins, cytotoxic 10-(indol-3-yl)-[13]cytochalasans from Chaetomium spp. IV. Carbon-13 nuclear magnetic resonance spectra and their application to a biosynthetic study. Chem. Pharm. Bull., 31, 490–498 (1983).
Silverton, J. V., Akiyama, T., Kabuto, C., Sekita, S., Yoshihira, K., and Natori, S., X-ray analysis of chaetoglobosin A, an indol-3-yl-[13]-cytochalasan from Chaetomium globosum. Tetrahedron Lett., 17, 1349–1350 (1976).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, J. T., Bokesch, H., Kenney, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst., 82, 1107–1112 (1990).
Soytong, K., Chaetomium in Thailand soils. Thai Phytopathog., 11, 86–111 (1991).
Takahashi, M., Koyama, K., and Natori, S., Four new azaphilones from Chaetomium globosum var. flavo-viridae. Chem. Pharm. Bull., 38, 625–628 (1990).
Udagawa, S., Muroi, T., Kurata, H., Sekita, S., Yoshihira, K., Natori, S., and Umeda, M., The production of chaetoglobosins, sterigmatocystin, O-methylsterigmatocystin, and chaetocin by Chaetomium subspecies and related fungi. Can. J. Microbiol., 25, 170–177 (1979).
Voigt, W., Sulforhodamine B assay and chemosensitivity. Methods Mol. Med., 110, 39–48 (2005).
Von Arx, J. A., The Ascomycete Genus Chaetomium (Nova Hedwigia Beihefte)., Lubrecht & Cramer Ltd, UK, pp. 1–162, (1986).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thohinung, S., Kanokmedhakul, S., Kanokmedhakul, K. et al. Cytotoxic 10-(indol-3-yl)-[13]cytochalasans from the fungus Chaetomium elatum ChE01. Arch. Pharm. Res. 33, 1135–1141 (2010). https://doi.org/10.1007/s12272-010-0801-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0801-5