Abstract
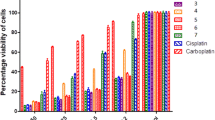
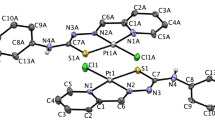
A novel mixed NH3/NH2OH platinum(II) complex cis-[Pt(NH3)(NH2OH)Cl2] was synthesized and characterized by elemental analysis, FAB-MS, FT-IR and 1H NMR spectroscopy. This complex was determined to have a good water-solubility and satisfactory stability. The pertinent complex was evaluated for its in vitro cytotoxicity against 3AO, HCT-116, LNcap, A549/ATCC and SGC-7901 human carcinoma cell lines. It shows appreciable cytotoxic activity that is comparable with cisplatin and is much more active than carboplatin.
Similar content being viewed by others
References
Abu-Surrah, A. S. and Kettunen, M., Platinum group antitumor chemistry: Design and development of new anticancer drugs complementary to cisplatin. Curr. Med. Chem., 13, 1337–1357 (2006).
Chen, X.-Z., Xie, M.-J., Liu, W.-P., Ye, Q.-S., Yu, Y., Hou, S.-Q., Gao, W.-G., and Liu, Y., Synthesis and structure of platinum (II) complexes containing an asymmetric chelating diamine 2-morpholinoethylamine as the carrier. Inorganica Chim. Acta, 360, 2851–2856 (2007).
Cossa, G., Gatti, L., Zunino F., and Perego, P., Strategies to Improve the Efficacy of Platinum Compounds. Curr. Med. Chem., 16, 2355–2365 (2009).
Galanski, M., Recent developments in the field of anticancer platinum complexes. Recent Pat. Anticancer Drug Discov., 1, 285–295 (2006).
Ho, Y.-P., Au-Yeung, S. C. F., and To, K. K. W., Platinum-based anticancer agents: innovative design strategies and biological perspectives. Med. Res. Rev., 23, 633–655 (2003).
Jakupec, M. A., Galanski, M., and Keppler, B. K., Tumour-inhibiting platinum complexes-state of the art and future perspectives. Rev. Physiol. Biochem. Pharmacol., 146, 1–54 (2003).
Kelland, L., The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer, 7, 573–584 (2007).
Kim, J. C., Lee, M.-H., and Choi, S. K., Synthesis and antitumor evaluation of cis-(1,2-diaminoethane) dichloroplatinum (II) complexes linked to 5- and 6-methy-leneuracil and -uridine analogues. Arch. Pharm. Res., 21, 465–469 (1998).
Kostova, I., Platinum complexes as anticancer agents. Recent Pat. Anticancer Drug Discov., 1, 1–22 (2006).
Liu W.-P., Chen X.-Z., Xie M.-J., Lou L.-G., Ye Q.-S., Yu Y., and Hou S.-Q., Synthesis and anticancer activity of [2-hydroxy-1,3-diaminopropane-κ2N,N′] platinum (II) complexes. J. Inorg. Biochem., 102, 1942–1946 (2008).
Momekov, G., Bakalova, A., and Karaivanova, M., Novel approaches towards development of non-classical platinum-based antineoplastic agents: design of platinum complexes characterized by an alternative DNA-binding pattern and/or tumor-targeted cytotoxicity. Curr. Med. Chem., 12, 2177–2191 (2005).
Montana, A. M. and Batalla, C., The rational design of anticancer platinum complexes: The importance of the structure-activity relationship. Curr. Med. Chem., 16, 2235–2260 (2009).
Nakamoto, K., McCarthy, P. J., Fujita, J., Condrate, R. A., and Behnke, G. T., Infrared studies of ligand-ligand interaction in dihalogenodiammineplatinum (II) complexes. Inorg. Chem., 4, 36–43 (1965).
Rosenberg, B., VanCamp, L., and Krigas, T., Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature, 205, 698–699 (1965).
Rosenberg, B., VanCamp, L., Trosko, J. E., and Mansour, H. V., Platinum compounds: A new class of potent antitumour agents. Nature, 222, 385–386 (1969).
Starkov, A. K. and Kozhukhovskaya, G. A., Conversion of magnus salt into diamminedichloroplatinum (II) isomers in aqueous solution. Russ. J. Inorg. Chem., 53, 1426–1429 (2008).
Wong, E. and Giandomenico, C. M., Current status of platinum-based antitumor drugs. Chem. Rev., 99, 2451–2466 (1999).
Ye, Q.-S., Lou, L.-G., Liu, Z.-Y., Liu, W.-P., Hou, S.-Q., Chen, X.-Z., and Yu, Y., Synthesis, characterization and cytotoxicity of diam(m)ineplatinum (II) complexes containing β-phenylisosuccinate ligand. Arch. Pharm. (Weinheim), 340, 599–602 (2007).
Ye, Q.-S., Xie, M.-J., Liu, W.-P., Chen, X.-Z., Yu, Y., Chang, Q.-W., and Hou, S.-Q., Synthesis, Characterization and Cytotoxicity of Dihalogeno-platinum (II) Complexes with L-Histidine Ligand. Chem. Pharm. Bull., 57, 424–427 (2009).
Zorbas-Seifried, S., Jakupec, M. A., Kukushkin, N. V., Groessl, M., Hartinger, C. G., Semenova, O., Zorbas, H., Kukushkin, V. Y., and Keppler, B. K., Reversion of structure-activity relationships of antitumor platinum complexes by acetoxime but not hydroxylamine ligands. Mol. Pharmacol., 71, 357–365 (2007).
Zutphen, S. V. and Reedijk, J., Targeting platinum antitumour drugs: overview of strategies employed to reduce systemic toxicity. Coord. Chem. Rev., 249, 2845–2853 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ye, QS., Liu, WP., Chen, XZ. et al. Synthesis and In Vitro cytotoxicity of Cis-[Pt(NH3)(NH2OH)Cl2]. Arch. Pharm. Res. 33, 807–811 (2010). https://doi.org/10.1007/s12272-010-0602-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0602-x