Abstract
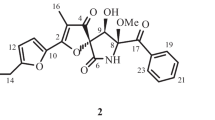
Two new hetero-spirocyclic γ-lactams, azaspirofurans A (1) and B (2), had been isolated from a marine sediment-derived fungus Aspergillus sydowi D2–6. Their structures were determined by the chemical evidence and spectral analyses. Compound 1 exhibited cytotoxicity against A549 cell line with the IC50 value of 10 μM by MTT method.
Similar content being viewed by others
References
Ando, O., Satake, H., Nakajima, M., Sato, A., Nakamura, T., Kinoshita, T., Furuya, and K. Synerazol, A new antifungal antibiotic. J. Antibiot., 44, 382–389 (1991).
Asami, Y., Kakeya, H., Onose, R., Yoshida, A., Matsuzaki, H., and Osada, H., Azaspirene, a novel angiogenesis inhibitor containing a 1-oxa-7-azaspiro [4, 4] non-2-ene-4,6-dione skeleton produced by the fungus Neosartorya sp. Org. Lett., 4, 2845–2848 (2002).
Bloch, P. and Tamm, C., Isolation and structure of pseurotin A, a microbial metabolite of Pseudeurotium ovalis STOLK with an unusual heterospirocyclic system. Helv. Chim. Acta, 64, 304–315 (1981).
Boot, C., Gassner, N. C., Compton, J. E., Tenney, K., Tamble, C. M., Lokey, R. S., Holman, T. R., and Crews, P., Pinpointing pseurotins from a marine-derived Aspergillus as tools for chemical genetics using a synthetic lethality yeast screen. J. Nat. Prod., 70, 1672–1675 (2007).
Hayashi, Y., Shoji, M., Mukaiyama, T., Gotoh, H., Yamaguchi, S., Nakata, H., and Osada, H., First asymmetric total synthesis of synerazol, an antifungal antibiotic, and determination of its absolute stereochemistry. J. Org. Lett., 70, 5643–5654 (2005).
Mossman, T., Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assay. J. Immunol. Methods, 65, 55–63 (1983).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ren, H., Liu, R., Chen, L. et al. Two new hetero-spirocyclic γ-lactam derivatives from marine sediment-derived fungus Aspergillus sydowi D2–6. Arch. Pharm. Res. 33, 499–502 (2010). https://doi.org/10.1007/s12272-010-0401-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0401-4