Abstract
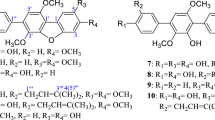
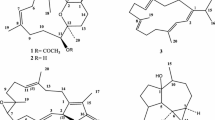
As part of an ongoing search for bioactive metabolites from the fungus Aspergillus versicolor derived from a marine sponge Petrosia sp., an aromatic polyketide derivative (1), two xanthones (2 and 3), and five anthraquinones (4–8) were isolated by bioactivity-guided fractionation. The gross structures were determined based on the NMR and MS spectroscopic data, and the absolute configurations were defined by comparison of optical rotation data with those of reported. Compounds 2, 4, 5, and 7 exhibited significant cytotoxicity against five human solid tumor cell lines (A-549, SK-OV-3, SK-MEL-2, XF-498, and HCT-15) with IC50 values in the range of 0.41–4.61 μg/mL. Compounds 4 and 7 exhibited antibacterial activity against several clinically isolated Gram-positive strains with MIC values of 0.78–6.25 μg/mL.
Similar content being viewed by others
References
Arai, T. and Sano, H., Novel UV-absorbing compounds FK17-P2a, FK-17-P2b1, FK17-P2b2, and FK17-P3 and manufacture of the compounds with Aspergillus sp. Jpn. Kokai Tokkyo Koho, pp. 6, (1994)
Augamp, P. J. and Holzapfel, G. W., Polyhydroxyanthraquinones from Aspergillus versicolor, Aspergillus nidulans and Bipolaris sp. J. S. Afr. Chem. Inst., 23, 40–56 (1970).
Bashyal, B. P., Wijeratne, E. M. K., Faeth, S. H., and Gunatilaka, A. A. L. Globosumones A-C, cytotoxic orsellinic acid esters from the sonoran desert endophytic fungus Chaetomium globosum. J. Nat. Prod., 68, 724–728 (2005).
Belofsky, G. N., Jensen, P. R., Renner, M. K., and Fenical, W., New cytotoxic sesquiterpenoid nitrobenzoyl esters from a marine isolate of the fungus Aspergillus versicolor. Tetrahedron, 54, 1715–1724 (1998).
Bennett, J. W., Lee, L. S., Shoss, S. M., and Boudreaux, G. H., Identification of averantin as aflatoxin B1 precursor: Placement in the biosynthetic pathway. Appl. Environ. Microbiol., 39, 835–839 (1980).
Fredenhagen, A., Hug, P., Sauter, H., and Peter, H. H., Paeciloquinones A, B, C, D, E and F: New potent inhibitors of protein tyrosine kinase produced by Paecilomyces carneus. J. Antibiot., 48, 199–204 (1995).
Fremlin, L. J., Piggott, A. M., Lacey, E., and Capon, R. J., Cottoquinazoline A and cotteslosins A and B, metabolites from an Australian marine-derived strain of Aspergillus versicolor. J. Nat. Prod., 72, 666–670 (2009).
Hyde, K. D., Farrant, C. A., and Jones, E. B. G., Isolation and culture of marine fungi. Botanica Marina, 30, 291–303 (1987).
Lee, Y. M., Mansoor, T. A., Hong, J., Lee, C. O., Bae, K. S., and Jung, J. H., Polyketides from a sponge-derived fungus, Aspergillus versicolor. Nat. Prod. Sci., 13, 90–96 (2007).
Lim, Y. J., Park, H. S., Im, K. S., Lee, C. O., Hong, J., Lee, M. Y., Kim, D. K., and Jung, J. H., Additional cytotoxic polyacetylenes from the marine sponge Petrosia species. J. Nat. Prod., 64, 46–53 (2001).
Radin, N. S., Meta-analysis of anticancer drug structuressignificance of their polar allylic moieties. Anticancer Agents Med. Chem., 7, 209–222 (2007).
Shier, W. T., Lao, Y., Steele, T. W. J., and Abbas, H. K., Yellow pigments used in rapid identification of aflatoxin-producing Aspergillus strains are anthraquinones associated with the aflatoxin biosynthetic pathway. Bioorg. Chem., 33, 426–438 (2005).
Yamaguchi, Y., Masuma, R., Kim, Y. P., Uchida, R., Tomoda, H., and Omura, S., Taxonomy and secondary metabolites of Pseudobotrytis sp. FKA-25. Mycoscience, 45, 9–16 (2004).
Zhu, F. and Lin, Y., Three xanthones from a marine-derived mangrove endophytic fungus. Chem. Nat. Com., 43, 132–135 (2007)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, Y.M., Li, H., Hong, J. et al. Bioactive metabolites from the sponge-derived fungus Aspergillus versicolor . Arch. Pharm. Res. 33, 231–235 (2010). https://doi.org/10.1007/s12272-010-0207-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0207-4