Abstract
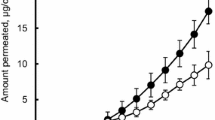
To increase the skin permeation of pranoprofen from the ethylene-vinyl acetate (EVA) matrix, different types of enhancers were added to an EVA matrix containing 2% pranoprofen. The pharmacokinetics and bioavailability of pranoprofen, an anti-inflammatory drug, were examined to determine the feasibility of an enhanced transdermal delivery system for pranoprofen from an EVA matrix containing caprylic acid as the enhancer in rats. The effects of the enhancers on the level of pranoprofen permeation through the skin were evaluated using Franz diffusion cells that were fitted with the intact excised rat skin. Among the enhancers used, including the fatty acids (saturated, unsaturated), the glycols, the glycerides, and the pyrrolidones, caprylic acid showed the best enhancement. A pranoprofen-EVA matrix system was formulated containing caprylic acid as an enhancer. The pranoprofen-EVA matrix system (8 mg/kg) was applied to the abdominal skin of rats. The blood samples were collected through the femoral artery for 24 h and the plasma concentrations of pranoprofen were determined by HPLC. The pharmacokinetic parameters were calculated using the MULTI computer program. The area under the curve (AUC) was significantly higher in the enhancer group (55.49 ± 13.87 ng/mL·h) than in the control group (22.48 ± 5.63 ng/mL·h), which was treated transdermally without the enhancer, showing about 246% increased bioavailability (p<0.05). As the pranoprofen-EVA matrix containing caprylic acid as an enhancer was administered to rats via the transdermal routes, the relative bioavailability increased about 2.46-fold compared to the control group, showing a relatively constant, sustained blood concentration. These results show that a pranoprofen-EVA matrix containing a permeation enhancer could be developed as a transdermal delivery system to provide a sustained plasma concentration.
Similar content being viewed by others
References
Aioi, A., Kuriyama, K., Shimizu, T., Yoshioka, M., and Uenoyama, S., Effects of vitamin E and squalene on skin irritation of an absorption enhancer, laurylsarcosine, Int. J. Pharm., 93, 1–6 (1993).
Amnuaikit, C., Ikeuchi, I., Ogawara, K. I., Higaki, K., and Kimura, T., Skin permeation of propranolol from polymeric film containing terpene enhancers for transdermal use. Int. J. Pharm., 289, 167–178 (2005).
Babu, R. J. and Pandit, J. K., Effect of penetration enhancers on the release and skin permeation of bupranolol from reservoir-type transdermal delivery systems. Int. J. Pharm., 288, 325–334 (2005).
Carelli, V., DiColo, G., Nanoripieri, E., and Serafini, M., Bile acids enhancers of steroid penetration through excised hairless mouse. Int. J. Pharm,. 89, 81–89 (1993).
Cho, C. W., Choi, J. S., and Shin, S. C., Controlled release of pranoprofen from the ethylene-vinyl acetate matrix using plasticizer, Drug Dev. Ind. Pharm., 33, 747–753 (2007).
Gennaro, A. R., Remington: the Science and Practice of Pharmacy 19th ed. Mack Publishing Company, Easton. PA, pp.1207–1218 (1995).
Kanikkannan, N., Kandimalia, K., Lamba, S. S., and Singh, M., Structure-activity relationship of chemical penetration enhancers in transdermal drug delivery. Current Medicinal Chemistry, 7, 593–608 (2000).
Levin J. and Mailbach, H., The correlation between transepidermal water loss and percutaneous absorption: an overview. J. Cont. Rel., 103, 291–299 (2005).
Mura, P., Faucci, M. T., Bramanti, G., and Corti, P., Evaluation transcutol as a clonazepam transdermal permeation enhancer hydrophilic gel formulations. Eur. J. Pharm. Sci., 9, 365–372 (2000).
Nalluri, B. N., Milligan, C., Chen. J.,. Crooks, P. A., and Stinchcomb, A. L., In vitro release studies on matrix type transdermal drug delivery systems. In vitro release studies on matrix type transdermal drug delivery systems of naltroxone and its acetyl prodrug. Drug Dev. Ind. Pham., 31, 871–887 (2005).
Nomura, T., Imai, T., and Otagiri, M., Stereoselective disposition of pranoprofen, a nonsteroidal anti-inflammatory drug, in rabbits. Biol. Pharm. Bull. 6, 298–303 (1993).
Panchagnula, R., Bokalial, R., Sharma, P., and Khandavilli, S., Transdermal delivery of naloxone: skin permeation, pharmacokinetic, irritancy and stability studies, Int. J. Pharm., 293, 213–223 (2005).
Prausnitz, M. R., Microneedles for transdermal drug delivery. Adv. Drug Delivery News. 56, 581–587 (2004).
Roberts, M. S. and Walters, K. A. (Eds.), Dermal Absorption and Assessment, Marcel Dekker, New York, pp. 1–785 (1998).
Sagara, K., Yamada, I., Matsuura, Y., Kawata, M., and Shibata, M., Gastrointestinal physiology-regulated dogs for bioavailability evaluation of an oral controlled-release dosage form composed of pulsatile release granules. Biol. Pharm. Bull., 19, 1184–1188 (1996).
Shinha, V. R. and Kaur, M. P., Permeation enhancers for transdermal drug delivery. Drug Dev. Ind. Pharm., 26, 1131–1140 (2000).
Shin, S. C. and Kim, J. Y., Enhanced permeation of triamcinolone acetonide through the buccal mucosa. Eur. J. Pharm. Biopharm., 50, 217–220 (2000).
Wang, M. Y., Yang, Y. Y., and Heng, P. W. S., Skin permeation of physostigmine from acid-based formulations: evaluating the choice of solvent. Int. J. Pharm., 290, 25–36 (2005).
Warner, K. S., Li, S. K., He, N., Suhonen, T. M., Chantasart, D., Bolikal, D., and Higuchi, W. I., Structure-activity relationship for chemical skin permeation enhancers; probing the chemical microenvironment of the site of action. J. Pharm. Sci., 92, 1305–1322 (2003).
Williams, A. C. and Barry, B. W., Skin absorption enhancers. Crit. Rev. Ther. Drug., 9, 305–353 (1992).
Williams, A. C. and Barry, B. W., Penetration enhancers. Adv. Drug Delivery News., 56, 603–618 (2004).
Yamaoka, K., Tanigawara, Y., Nakagawa, T., and Uno, T., A pharmacokinetic analysis program (multi) for microcomputer. J. Pharmacobiodyn., 4, 879–885 (1981).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, CW., Choi, JS., Yang, KH. et al. Enhanced transdermal absorption and pharmacokinetic evaluation of pranoprofen-ethylene-vinyl acetate matrix containing penetration enhancer in rats. Arch. Pharm. Res. 32, 747–753 (2009). https://doi.org/10.1007/s12272-009-1514-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-009-1514-5