Abstract
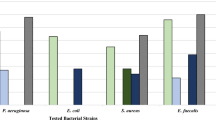
Several 1-benzhydryl-sulfonyl-4-(3-(piperidin-4-yl)propyl)piperidine derivatives 8(a–j) were prepared by the treatment of substituted benzhydryl chlorides with 4-(3-(piperidin-4-yl)propyl)piperidine followed by N-sulfonation with sulfonyl chlorides in the presence of dry methylene dichloride and triethyl amine. The synthesized compounds were characterized by 1H-NMR, IR, and elemental analysis. All the synthesized compounds were evaluated in vitro for their efficacy as antimicrobial agents by artificial inoculation technique against standard strains of two important bacterial viz., Xanthomonas axonopodis pv. vesicatoria and Ralstonia solanacearum as well as and two fungal pathogens namely Alternaria solani and Fusarium solani of tomato plants. We have briefly investigated the structure-activity relation studies and reveal that the nature of substitutions on benzhydryl ring and sulfonamide ring influences the antibacterial activity. Among the synthesized new compounds 8b, 8d, 8g, 8h, 8i, and 8j were showed significant potent antimicrobial activities compared to the standard drugs Chloramphenicol, Mancozeb.
Similar content being viewed by others
References
Akira, K., Masao, Y., and Masuo, M., Effect of chlorsulfuron on shoot sprouting from the stem segment of Torenia fournieri and Nicotiana tabacum. Weed Biol. Manag., 2, 213–234 (2002).
Amat, M., Hidalgo, J., and Bosch, J., Corrigendum. Tetrahedron Asymmetry., 7, 1845–1854 (1996).
Andrea, S. and Calaudin, T. S., Protease inhibitors — part 5. Alkyl/arylsulfonyl- and arylsulfonylureido-/arylureido-glycine hydroxamate inhibitors of Clostridium histolyticum collagenase. Eur. J. Med. Chem., 35, 299–307 (2000).
Andrea, S., Fabrizio, B., Marc, A. I., and Calaudin, T. S., Carbonic anhydrase inhibitors: synthesis of membraneimpermeant low molecular weight sulfonamides possessing in vivo selectivity for the membrane-bound versus cytosolic isozymes. J. Med. Chem., 43(2), 292–300 (2000).
Christopher, A. W., Scott, C. B., Keith, G. R., Silvia, D., Kevin, T. C., Sandra, L. G., Martin, S. S., Lorraine, M., William, A. S., Daria, H., Michael, M., Joseph, K., Renee, D., Karen, H., Janet, L., Anthony, C., Gwen, C., and Emilio, A. E., Combinatorial synthesis of CCR5 antagonists. Bioorg. Med. Chem. Lett., 11, 3137–3141 (2001).
Colapret, J. A., Diamantidis, G., Spencer, H. K., Spaulding, T. C., and Rudo, F. G., Synthesis and pharmacological evaluation of 4,4-disubstituted piperidines. J. Med. Chem., 32, 968–974 (1989).
De Waard, M, A., Synergism and antagonism in fungicides. Longman Scientific and Technical Essex UK, 355 (1987).
Finney, N., Ghosh, S., Hirst, J., Norman, P., and Rotella, D. P., Paper alert. Curr. Opin. Drug Discovery & Devel. 2, 175–181 (1999).
Fletcher, S. R., Burkamp, F., Blurton, P., Cheng, S. K., Clarkson, R., O’Connor, D., Spinks, D., Tudge, M., van Niel, M. B., Patel, S., Chapman, K., Marwood, R., Shepheard, S., Bentley, G., Cook, G. P., Bristow, L. J., Castro, J. L., Hutson, P. H., and MacLeod, A. M., 4-(Phenylsulfonyl)piperidines: novel, selective, and bioavailable 5-HT(2A) receptor antagonists. J. Med. Chem., 45(2), 492–503 (2002).
Ishii, H., Studies on fungicide resistance in phytopathogenic fungi. J. General. Plant. Pathology., 70(6), 379–385 (2004).
Jean-Marie, C., Isabelle, P., Wolfgang, S., Yveline, M. R., and Camille, G. W., Design, synthesis, and structure-activity relationships of a series of 3-[2-(1-benzylpiperidin-4-yl) ethylamino]pyridazine derivatives as acetylcholinesterase inhibitors. J. Med. Chem., 44, 2707–2718 (2001).
Klockgether, T., Wullner, J. P., Steinbach, J. P., Petersen, V., Turski, L., and Loschmann, P. A., Effects of the antiparkinsonian drug budipine on central neurotransmitter systems. Eur. J. Pharmacol., 301, 67–73 (1996).
Laura, G., Marinella, R., and Erik, D. C., Synthesis and antiviral/antiproliferative activity of some N-sulphonylbenzimidazoles. Bioorg. Med. Chem. Lett., 12, 2707–2710 (2002).
Lihu, Y., Greg, M. A., Arthur, P., Kwan, L., Tom, J., Kang, C., Klaus, D. S., William, F., Wanda, W. S. C., Shuet-Hing, L. C., and Roy, G. S., 1-[2(R)-(2-amino-2-methylpropionyl amino)-3-(1H-indol-3-yl)propionyl]-3-benzylpiperidine-3(S)-carboxylic acid ethyl ester (L-163,540): a potent, orally bioavailable, and short-duration growth hormone secretagogue. J. Med. Chem., 41(14), 2439–2441 (1998).
Maniyan, P., Bernard, S., Yuxiang, D., Jacques, C., Hugues, M., Susan, A. C., Darren, J. C., William, N. C., Josefina, S. T., Christian, S., Sergio, W., Reto, B., and Jonathan, L. V., Antimalarial activity of N-alkyl amine, carboxamide, sulfonamide, and urea derivatives of a dispiro-1,2,4-trioxolane piperidine. Bioorg. Med. Chem. Lett., 16, 5542–5545 (2006).
Matthew, W. H., Reddy, P. A., Douglas, F. C., and Steven, M. R., Inhibition of voltage-dependent sodium channels by the anticonvulsant gamma-aminobutyric acid type A receptor modulator, 3-benzyl-3-ethyl-2-piperidinone. J. Pharmacol. Exp. Ther., 285, 1303–1309 (1998).
Medina, J. C., Shan, B., Beckmann, H., Farrell, R. P., Clark, D. L., Learned, R. M., Roche, D., Li, A., Baichwal, V., Case, C., Baeuerle, P. A., Rosen, T., and Jaen, J. C., Novel antineoplastic agents with efficacy against multidrug resistant tumor cells. Bioorg. Med. Chem. Lett., 8, 2653–2656 (1998).
Nanjunda swamy, S., Basapp., Sarala, G., Priya, B. S., Gaonkar, S. L., Prasad, J. S., and Rangappa, K. S., Microwave-assisted synthesis of N-alkylated benzotriazole derivatives: antimicrobial studies. Bioorg. Med. Chem. Lett., 16, 999–1004 (2006).
Ohtaka., Kanazawa, T., Ito, K., and Tsukamoto, G., Benzylpiperazine derivatives. IV. Syntheses and cerebral vasodilating activities of 1-benzyl-4-diphenylmethylpiperazine derivatives. Chem. Pharm. Bull., 35, 3270–3275 (1987).
Padmakar, V. K., Sharma, V., Karmarkar, S., and Claudiu, T. S., QSAR studies on benzene sulfonamide carbonic anhydrase inhibitors: need of hydrophobic parameter for topological modeling of binding constants of sulfonamides to human CA-II. Bioorg. Med. Chem. Lett., 15(4), 923–930 (2005).
Priya, B. S., Basappa., Nanjunda swamy, S., and Rangappa, K. S., Synthesis and characterization of novel 6-fluoro-4-piperidinyl-1,2-benzisoxazole amides and 6-fluoro-chroman-2-carboxamides: antimicrobial studies. Bioorg. Med. Chem., 13, 2623–2628 (2005).
Ramalingan, C., Balasubramanian, S., Kabilan, S., and Vasudevan, M., Synthesis and study of antibacterial and antifungal activities of novel 1-[2-(benzoxazol-2-yl)ethoxy]-2,6-diarylpiperidin-4-ones. Eur. J. Med. Chem., 39(6), 527–533 (2004).
Ramalingan, C., Balasubramanian, S., Kabilan, S., and Vasudevan, M., Synthesis and microbiological evaluation of spiro piperidinyl heterocycles. Med. Chem. Res., 12(1), 41–47 (2003).
Ramalingan, C., Balasubramanian, S., Kabilan, S., and Vasudevan, M., Synthesis and microbiological evaluation of benzimidazolylethoxypiperidones. Med. Chem. Res., 12(1), 26–32 (2003).
Reddy, P. A., Woodward, K. E., McIlheran, S. M., Hsiang, B. C., Latifi, T. N., Hill, M. W., Rothman, S. M., Ferrendelli, J. A., and Covey, D. F., Synthesis and anticonvulsant activities of 3,3-dialkyl- and 3-alkyl-3-benzyl-2-piperidinones (delta-valerolactams) and hexahydro-2H-azepin-2-ones (epsilon-caprolactams). J. Med. Chem., 40, 44–49 (1997).
Sadashiva, M. P., Mallesha, H., Murthy, K. K., and Rangappa, K. S., Enhancement in antimicrobial activity of 2-(phenyl)-3-(2-butyl-4-chloro-1H-imidazolyl)-5-butylate isoxazolidine. Bioorg. Med. Chem. Lett., 15, 1811–1814 (2005).
Singh, S. K., Reddy, P. G., Rao, K. S., Lohray, B. B., Misra, P., Rajjak, S. A., Rao, Y. K., and Venkateswarlu, A., Polar substitutions in the benzenesulfonamide ring of celecoxib afford a potent 1,5-diarylpyrazole class of COX-2 inhibitors. Bioorg. Med. Chem. Lett., 14(2), 499–504 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vinaya, K., Kavitha, R., Ananda Kumar, C.S. et al. Synthesis and antimicrobial activity of 1-benzhydryl-sulfonyl-4-(3-(piperidin-4-yl) propyl)piperidine derivatives against pathogens of Lycopersicon esculentum: A structure-activity evaluation study. Arch. Pharm. Res. 32, 33–41 (2009). https://doi.org/10.1007/s12272-009-1115-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-009-1115-3