Abstract
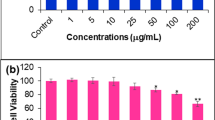
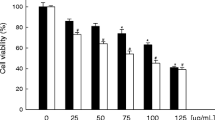
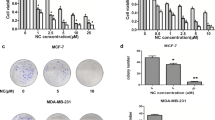
Although isothiocyanates have been shown to inhibit carcinogen-induced tumorigenesis, no studies have been made to determine their therapeutic potential for the treatment of breast cancer. In the present study, we evaluated the apoptotic activities of phenethyl isothiocyanate (PEITC) in human breast cancer MCF-7 cells. Exposure to PEITC potently reduced cell viability. In addition, DNA fragments and TUNEL positive nuclei were detected in PEITC-treated cells. Furthermore, PEITC induced apoptosis via activation of caspases 7 and 9 and the cleavage of PARP, and these effects were reversed by treatment with the caspase inhibitor, Z-VAD-fmk. PEITC also caused a decrease in the levels of Bcl-2 with a concomitant increase in Bax levels, which resulted in the release of cytochrome c. XIAP suppression and Smac translocation also contributed to the PEITC-induced apoptosis. However, PEITC did not increase the expressions of p53 and p21. Taken together, the results of this study demonstrate that PEITC significantly induces apoptosis via a mitochondrial pathway. Specifically, PEITC induced a change in the Bax/Bcl-2 ratios, XIAP levels and Smac translocation that was conjunction with the release of cytochrome c and following caspase activation. Therefore, PEITC has the potential for use as a therapeutic agent for the treatment of breast cancer.
Similar content being viewed by others
References
Almog, N. and Rotter, V., Involvement of p53 in cell differentiation and development. Biochim. Biophys. Acta, 1333, F1–27 (1997).
Baselga, J. and Mendelsohn, J., The epidermal growth factor receptor as a target for therapy in breast carcinoma. Breast Cancer Res. Treat., 29, 127–138 (1994).
Bold, R. J., Termuhlen, P. M., and Mcconkey, D. J., Apoptosis, cancer and cancer therapy. Surg. Oncol., 6, 133–142 (1997).
Cain, K., Brown, D. G., Langlais, C., and Cohen, G. M., Caspase activation involves the formation of the aposome, a large (approximately 700 kDa) caspase-activating complex. J. Biol. Chem., 274, 22686–22692 (1999).
Cho, M. K., Kim, W. D., Ki, S. H., Hwang, J. I., Choi, S., Lee, C. H., and Kim, S. G., Role of Galpha12 and Galpha13 as novel switches for the activity of Nrf2, a key antioxidative transcription factor. Mol. Cell. Biol., 27, 6195–6208 (2007).
Cho, M. K., Sung, M. A., Kim, D. S., Park, H. G., Jew, S. S., and Kim, S. G., 2-Oxo-3,23-isopropylidene-asiatate (AS2006A), a wound-healing asiatate derivative, exerts anti-inflammatory effect by apoptosis of macrophages. Int. Immunopharmacol., 3, 1429–1437 (2003).
Devarajan, E., Sahin, A. A., Chen, J. S., Krishnamurthy, R. R., Aggarwal, N., Brun, A. M., Sapino, A., Zhang, F., Sharma, D., Yang, X. H., Tora, A. D., and Mehta, K., Down-regulation of caspase 3 in breast cancer: a possible mechanism for chemoresistance. Oncogene, 21, 8843–8851 (2002).
Deveraux, Q. L. and Reed, J. C., IAP family proteins—suppressors of apoptosis. Genes Dev., 13, 239–252 (1999).
El-Deiry, W. S., Tokino, T., Velculescu, V. E., Levy, D. B., Parsons, R., Trent, J. M., Lin, D., Mercer, W. E., Kinzler, K. W., and Vogelstein, B., WAF1, a potential mediator of p53 tumor suppression. Cell, 75, 817–825 (1993).
Fahey, J. W., Zhang, Y., and Talalay, P., Broccoli sprouts: an exceptionally rich source of inducers of enzymes that protect against chemical carcinogens. Proc. Natl. Acad. Sci. USA, 94, 10367–10372 (1997).
Gross, A., Mcdonnell, J. M., and Korsmeyer, S. J., BCL-2 family members and the mitochondria in apoptosis. Genes Dev., 13, 1899–1911 (1999).
Hayes, J. D., Kelleher, M. O., and Eggleston, I. M., The cancer chemopreventive actions of phytochemicals derived from glucosinolates. Eur. J. Nutr., 47Suppl 2, 73–88 (2008).
Hecht, S. S., Inhibition of carcinogenesis by isothiocyanates. Drug Metab. Rev., 32, 395–411 (2000).
Hecht, S. S., Kenney, P. M., Wang, M., Trushin, N., and Upadhyaya, P., Effects of phenethyl isothiocyanate and benzyl isothiocyanate, individually and in combination, on lung tumorigenesis induced in A/J mice by benzo[a]pyrene and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone. Cancer Lett., 150, 49–56 (2000).
Hu, R., Kim, B. R., Chen, C., Hebbar, V., and Kong, A. N., The roles of JNK and apoptotic signaling pathways in PEITC-mediated responses in human HT-29 colon adenocarcinoma cells. Carcinogenesis, 24, 1361–1367 (2003).
Ji, Y. and Morris, M. E., Membrane transport of dietary phenethyl isothiocyanate by ABCG2 (breast cancer resistance protein). Mol. Pharm., 2, 414–419 (2005).
Kuang, Y. F. and Chen, Y. H., Induction of apoptosis in a nonsmall cell human lung cancer cell line by isothiocyanates is associated with P53 and P21. Food Chem. Toxicol., 42, 1711–1718 (2004).
Kurebayashi, J., Endocrine-resistant breast cancer: underlying mechanisms and strategies for overcoming resistance. Breast Cancer, 10, 112–119 (2003).
Kurokawa, H., Nishio, K., Fukumoto, H., Tomonari, A., Suzuki, T., and Saijo, N., Alteration of caspase-3 (CPP32/Yama/apopain) in wild-type MCF-7, breast cancer cells. Oncol. Rep., 6, 33–37 (1999).
Satyan, K. S., Swamy, N., Dizon, D. S., Singh, R., Granai, C. O., and Brard, L., Phenethyl isothiocyanate (PEITC) inhibits growth of ovarian cancer cells by inducing apoptosis: role of caspase and MAPK activation. Gynecol. Oncol., 103, 261–270 (2006).
Simstein, R., Burow, M., Parker, A., Weldon, C., and Beckman, B., Apoptosis, chemoresistance, and breast cancer: insights from the MCF-7 cell model system. Exp. Biol. Med. (Maywood), 228, 995–1003 (2003).
Talalay, P. and Fahey, J. W., Phytochemicals from cruciferous plants protect against cancer by modulating carcinogen metabolism. J. Nutr., 131, 3027S–3033S (2001).
Thomadaki, H. and Scorilas, A., BCL2 family of apoptosis-related genes: functions and clinical implications in cancer. Crit. Rev. Clin. Lab. Sci., 43, 1–67 (2006).
Twiddy, D., Brown, D. G., Adrain, C., Jukes, R., Martin, S. J., Cohen, G. M., Macfarlane, M., and Cain, K., Pro-apoptotic proteins released from the mitochondria regulate the protein composition and caspase-processing activity of the native Apaf-1/caspase-9 apoptosome complex. J. Biol. Chem., 279, 19665–19682 (2004).
Twiddy, D., Cohen, G. M., Macfarlane, M., and Cain, K., Caspase-7 is directly activated by the approximately 700- kDa apoptosome complex and is released as a stable XIAP-caspase- 7 approximately 200-kDa complex. J. Biol. Chem., 281, 3876–3888 (2006).
Visanji, J. M., Duthie, S. J., Pirie, L., Thompson, D. G., and Padfield, P. J., Dietary isothiocyanates inhibit Caco-2 cell proliferation and induce G2/M phase cell cycle arrest, DNA damage, and G2/M checkpoint activation. J. Nutr., 134, 3121–3126 (2004).
Xiao, D., Vogel, V., and Singh, S. V., Benzyl isothiocyanate-induced apoptosis in human breast cancer cells is initiated by reactive oxygen species and regulated by Bax and Bak. Mol. Cancer Ther., 5, 2931–2945 (2006).
Xiao, D., Zeng, Y., Choi, S., Lew, K. L., Nelson, J. B., and Singh, S. V., Caspase-dependent apoptosis induction by phenethyl isothiocyanate, a cruciferous vegetable-derived cancer chemopreventive agent, is mediated by Bak and Bax. Clin. Cancer Res., 11, 2670–2679 (2005).
Zhang, Y., Cancer-preventive isothiocyanates: measurement of human exposure and mechanism of action. Mutat. Res., 555, 173–190 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.W., Cho, M.K. Phenethyl isothiocyanate induced apoptosis via down regulation of Bcl-2/XIAP and triggering of the mitochondrial pathway in MCF-7 cells. Arch. Pharm. Res. 31, 1604–1612 (2008). https://doi.org/10.1007/s12272-001-2158-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-001-2158-2