Abstract
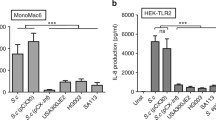
Lipoproteins (Lpp) of Gram-positive bacteria play a crucial role in immune modulation. The degree of lipid acylation determines whether the TLR2/TLR1 or the TLR2/TLR6 signalling pathway is induced in the infected host. Both pathways differ significantly in the extent of innate immune activation. Commensal bacteria on our skin signal via the TLR2/TLR1 pathway thus inducing only a mild immune response while non-commensal bacteria signal via TLR2/TLR6 pathway thereby inducing a fulminant immune response. Consequently, the structure of the lipid residue in Lpp decides over immune tolerance or intolerance.
Similar content being viewed by others
Literatur
Braun V, Rehn K (1969) Chemical characterization, spatial distribution and function of a lipoprotein (murein-lipoprotein) of the E. coli cell wall. The specific effect of trypsin on the membrane structure. Eur J Biochem 10:426–438
Buddelmeijer N (2015) The molecular mechanism of bacterial lipoprotein modification - how, when and why? FEMS Microbiol Rev 39:246–261
Nguyen MT, Götz F (2016) Lipoproteins of Gram-positive bacteria: key players in the immune response and virulence. Microbiol Mol Biol Rev 80:891–903
Schmaler M, Jann NJ, Ferracin F et al. (2009) Lipoproteins in Staphylococcus aureus mediate inflammation by TLR2 and iron-dependent growth in vivo. J Immunol 182:7110–7118
Stoll H, Dengjel J, Nerz C et al. (2005) Staphylococcus aureus deficient in lipidation of prelipoproteins is attenuated in growth and immune activation. Infect Immun 73:2411–2423
Shahmirzadi SV, Nguyen MT, Götz F (2016) Evaluation of Staphylococcus aureus lipoproteins: role in nutritional acquisition and pathogenicity. Front Microbiol 7:1404
Nguyen MT, Kraft B, Yu W et al. (2015) The nuSaα specific lipoprotein like cluster (lpl) of S. aureus USA300 contributes to immune stimulation and invasion in human cells. PLoS Pathog 11:e1004984
Hashimoto M, Tawaratsumida K, Kariya H et al. (2006) Not lipoteichoic acid but lipoproteins appear to be the dominant immunobiologically active compounds in Staphylococcus aureus. J Immunol 177:3162–3169
Schäffler H, Demircioglu DD, Kuhner D et al. (2014) NOD2 stimulation by Staphylococcus aureus-derived peptidoglycan is boosted by Toll-like receptor 2 costimulation with lipoproteins in dendritic cells. Infect Immun 82:4681–4688
Skabytska Y, Wolbing F, Gunther C et al. (2014) Cutaneous innate immune sensing of Toll-like receptor 2-6 ligands suppresses T cell immunity by inducing myeloid-derived suppressor cells. Immunity 41:762–775
Nguyen MT, Uebele J, Kumari N et al. (2017) How commensal staphylococci adapt to our immune system. Nat Commun (accepted)
Nguyen MT, Hanzelmann D, Hartner T et al. (2016) Skin-specific unsaturated fatty acids boost the Staphylococcus aureus innate immune response. Infect Immun 84:205–215
Author information
Authors and Affiliations
Corresponding author
Additional information
Minh-Thu Nguyen B. Sc. an der Vietnam National University (Hanoi) am College of Natural Sciences. M. Sc. an der Sungkyunkwan University, Südkorea. 2010 mit Stipendium der Regierung Vietnam an die Universität Tübingen (seit Juli 2014 DFG-finanziert). Seit 2016 Postdoc der AG Mikrobielle Genetik an der Universität Tübingen.
Friedrich Götz Biologie- und Chemiestudium an der LMU München; 1978 Promotion bei Prof. Dr. K.-H. Schleifer. 1980–1981 EMBO Long-Term Fellowship für Postdoc-Aufenthalt an der Universität Uppsala, Schweden.1985 Habilitation an der TU München. Seit 1987 Lehrstuhl Mikrobielle Genetik an der Universität Tübingen. Seit 2015 Seniorpro fessor an der Universität Tübingen.
Rights and permissions
About this article
Cite this article
Nguyen, M.T., Götz, F. Wie bakterielle Moleküle unser Immunsystem manipulieren. Biospektrum 23, 759–761 (2017). https://doi.org/10.1007/s12268-017-0867-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12268-017-0867-4