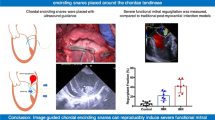
Abstract
Several interventional therapies are in development to treat functional tricuspid regurgitation. Most have failed to achieve adequate efficacy, as animal models of this lesion are lacking. We developed a new image-guided technique in swine, by tethering the tricuspid valve chordae using echo-guided chordal encircling snares. Five swine underwent baseline echocardiographic assessment of tricuspid valve function, followed by echo-guided placement of snares that encircle the chordae inserting into the anterior and posterior tricuspid valve leaflets. Tethering these snares and stabilizing them on the right ventricle caused the regurgitant fraction to increase from 8.48±5.38% to 48.76±12.5%, and the valve tenting area to increase from 60.26±52.19 to 160.9±86.92 mm2. Image-guided chordal encircling snares could reproducibly induce clinically significant levels of functional tricuspid regurgitation and create a valve geometry like that seen in patients, providing a new animal model for use to study novel interventional devices.
Graphical Abstract







Similar content being viewed by others
Abbreviations
- FTR:
-
Functional tricuspid regurgitation
- SL:
-
Septal leaflet
- AL:
-
Anterior leaflet
- PL:
-
Posterior leaflet
- SMPM:
-
Septal/medial papillary muscle
- APM:
-
Anterior papillary muscle
- IPM:
-
Inferior papillary muscle
- TA:
-
Tricuspid annulus
- RA:
-
Right atrium
- RV:
-
Right ventricle
References
Zhan Y, Debs D, Khan MA, Nguyen DT, Graviss EA, Khalaf S, Little SH, Reardon MJ, Nagueh S, Quinones MA, Kleiman N, Zoghbi WA, Shah DJ. Natural history of functional tricuspid regurgitation quantified by cardiovascular magnetic resonance. J Am Coll Cardiol. 2020;76:1291–301.
Mutlak D, Khalil J, Lessick J, Kehat I, Agmon Y, Aronson D. Risk factors for the development of functional tricuspid regurgitation and their population-attributable fractions. JACC Cardiovasc Imaging. 2020;13:1643–51.
Sala A, Lorusso R, Bargagna M, Ascione G, Ruggeri S, Meneghin R, Schiavi D, Buzzatti N, Trumello C, Monaco F, Agricola E, Alfieri O, Castiglioni A, De Bonis M. Isolated tricuspid valve surgery: first outcomes report according to a novel clinical and functional staging of tricuspid regurgitation. Eur J Cardiothorac Surg. 2021;60:1124–30.
Pahwa S, Saran N, Pochettino A, Schaff H, Stulak J, Greason K, Daly R, Crestanello J, King K, Dearani J. Outcomes of tricuspid valve surgery in patients with functional tricuspid regurgitation. Eur J Cardiothorac Surg. 2021;59:577–85.
Besler C, Meduri CU, Lurz P. Transcatheter treatment of functional tricuspid regurgitation using the Trialign device. Interv Cardiol. 2018;13:8–13.
Schofer J. Transcatheter interventions for tricuspid regurgitation: Trialign and Mitralign. EuroIntervention. 2016;12:Y119–20.
Lurz P, Besler C, Kiefer P, Ender J, Seeburger J. Early experience of the Trialign system for catheter-based treatment of severe tricuspid regurgitation. Eur Heart J. 2016;37:3543.
Pardo Sanz A, Gomez JLZ, Tahoces LS, Ruiz JMM, Martin AG, Gomez AG, Baydes RH, Sequeiros MA, Fernandez MS, Barcelo JLM, Ferrer EG, Santos SF, Loban CF, Recalde AS. Long-term outcomes of percutaneous tricuspid annuloplasty with Cardioband device. Eur Heart J Cardiovasc Imaging. 2022;23:979–88.
Kuwata S, Taramasso M, Nietlispach F, Maisano F. Transcatheter tricuspid valve repair toward a surgical standard: first-in-man report of direct annuloplasty with a cardioband device to treat severe functional tricuspid regurgitation. Eur Heart J. 2017;38:1261.
Reddy VY, Abbo AR, Ruiz CE, Kerner A, Kreidel F, Topilsky Y, Kipshidze N, Avisar N, Petru J, Neuzil P. First-in-human percutaneous circumferential annuloplasty for secondary tricuspid regurgitation. JACC Case Rep. 2020;2:2176–82.
Fam NP, Ali FM, Hassanin M, Ong G. Transcatheter tricuspid valve repair with the modified TriClip/MitraClip G4 system. EuroIntervention. 2021;17:e441–2.
Aurich M, Volz MJ, Mereles D, Geis NA, Frey N, Konstandin MH, Raake PW. Initial Experience with the PASCAL ace implant system for treatment of severe tricuspid regurgitation. Circ Cardiovasc Interv. 2021;14:e010770.
Asmarats L, Perlman G, Praz F, Hensey M, Chrissoheris MP, Philippon F, Ofek H, Ye J, Puri R, Pibarot P, Attinger A, Moss R, Bedard E, Moschovitis A, Reineke D, Lauck S, Blanke P, Leipsic J, Spargias K, et al. Long-term outcomes of the FORMA transcatheter tricuspid valve repair system for the treatment of severe tricuspid regurgitation: insights from the first-in-human experience. JACC Cardiovasc Interv. 2019;12:1438–47.
Fam NP, von Bardeleben RS, Hensey M, Kodali SK, Smith RL, Hausleiter J, Ong G, Boone R, Ruf T, George I, Szerlip M, Nabauer M, Ali FM, Moss R, Bapat V, Schnitzler K, Kreidel F, Ye J, Deva DP, et al. Transfemoral transcatheter tricuspid valve replacement with the EVOQUE System: a multicenter, observational, first-in-human experience. JACC Cardiovasc Interv. 2021;14:501–11.
Otaki M, Lust RM. Modification of De Vega’s tricuspid annuloplasty for experimental tricuspid regurgitation. J Card Surg. 1994;9:399–404.
Walter EM, Vasilyev NV, Sill B, Padala M, Jimenez J, Yoganathan AP, Hetzer R, del Nido PJ. Creation of a tricuspid valve regurgitation model from tricuspid annular dilatation using the cardioport video-assisted imaging system. J Heart Valve Dis. 2011;20:184–8.
Buffington CW, Nystrom EUM. Neither the accuracy nor the precision of thermal dilution cardiac output measurements is altered by acute tricuspid regurgitation in pigs. Anesth Analg. 2004;98:884–90.
Boerboom LE, Kinney TE, Olinger GN, Hoffmann RG. Validity of cardiac output measurement by the thermodilution method in the presence of acute tricuspid regurgitation. J Thorac Cardiovasc Surg. 1993;106:636–42.
Xie XJ, Liao SJ, Wu YH, Lu C, Zhu P, Fei HW, Xiao XJ, Huang HL. Tricuspid leaflet resection in an open beating heart for the creation of a canine tricuspid regurgitation model. Interact Cardiovasc Thorac Surg. 2016;22:149–54.
Ishibashi Y, Rembert JC, Carabello BA, Nemoto S, Hamawaki M, Zile MR, Greenfield JC Jr, Gt C. Normal myocardial function in severe right ventricular volume overload hypertrophy. Am J Physiol Heart Circ Physiol. 2001;280:H11–6.
Bai Y, Chen HY, Zong GJ, Jiang HB, Li WP, Wu H, Zhao XX, Qin YW. Percutaneous establishment of tricuspid regurgitation: an experimental model for transcatheter tricuspid valve replacement. Chin Med J (Engl). 2010;123:806–9.
Malinowski M, Proudfoot AG, Langholz D, Eberhart L, Brown M, Schubert H, Wodarek J, Timek TA. Large animal model of functional tricuspid regurgitation in pacing induced end-stage heart failure. Interact Cardiovasc Thorac Surg. 2017;24:905–10.
Gaweda B, Iwasieczko A, Gaddam M, Bush JD, MacDougal B, Timek TA. Chronic ovine model of right ventricular failure and functional tricuspid regurgitation. J Vis Exp. 2023;17(193). https://doi.org/10.3791/64529
Spinner EM, Shannon P, Buice D, Jimenez JH, Veledar E, Del Nido PJ, Adams DH, Yoganathan AP. In vitro characterization of the mechanisms responsible for functional tricuspid regurgitation. Circulation. 2011;124:920–9.
Maisano F, Reser D, Pavicevic J, Guidotti A, Denti P, Taramasso M, Addis A, Cesarovic N, Emmert MY, Nietlispach F, Swain J, Falk V, Leon M. A translational "humanised" porcine model for transcatheter mitral valve interventions: the neo inferior vena cava approach. EuroIntervention. 2015;11:92–5.
Acknowledgements
The authors acknowledge veterinary technicians Amanda Maddamma and Trish Bruce for providing anesthesia support for the animal procedures. The authors also acknowledge infrastructure support from the Carlyle Fraser Heart Center at Emory University Hospital Midtown for their generous support of our lab.
Funding
This work was funded by grants from the National Institutes of Health HL135145, HL140325, HL133667, and infrastructure support from the Carlyle Fraser Heart Center at Emory University Hospital Midtown to M. Padala, when he was employed at Emory University. As of April 2023, Dr. Padala has changed his employment to be the founder and chief scientific officer of Nyra Medical. D. Onohara was supported by a grant 23CDA1053806, D. Xu was supported by grant 834086, and K. Suresh was supported by grant 915799 from the American Heart Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
This study did not include any human subject research. Animals were used in this research, as there was no relevant alternative available. The animal study protocol, including the procedures, animal care, and endpoints, was reviewed and approved by the Institutional Animal Use and Care Committee at Emory University, and the procedures were performed according to the guidelines for the use of animals in research by the National Institutes of Health. The studies were performed at an AAALAC-accredited facility, which at all times was overseen by an attending veterinarian.
Human and Animal Rights
This study did not include any human subject’s research.
Conflict of Interest
None of the authors has any direct conflicts of interest to report that are relevant to this work. Outside this, M.Padala reports owning significant stock, employment, and board directorship at Nyra Medical, Inc, which has neither sponsored, reviewed nor had any input in this work. K. Suresh reports owning minor stock in Nyra Medical. D. Xu is currently an employee of Peijia Medical, which did not have any role in this work.
Additional information
Associate Editor Laura Iop oversaw the review of this article
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Onohara, D., Silverman, M., Suresh, K.S. et al. An Animal Model of Functional Tricuspid Regurgitation by Leaflet Tethering Using Image-Guided Chordal Encircling Snares. J. of Cardiovasc. Trans. Res. 17, 417–425 (2024). https://doi.org/10.1007/s12265-023-10424-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-023-10424-3