Abstract
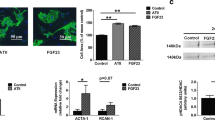
The β2-selective adrenoreceptor agonist clenbuterol promotes both skeletal and cardiac muscle hypertrophy and is undergoing clinical trials in the treatment of muscle wasting and heart failure. We have previously demonstrated that clenbuterol induces a mild physiological ventricular hypertrophy in vivo with normal contractile function and without induction of α-skeletal muscle actin (αSkA), a marker of pathological hypertrophy. The mechanisms of this response remain poorly defined. In this study, we examine the direct action of clenbuterol on cardiocyte cultures in vitro. Clenbuterol treatment resulted in increased cell size of cardiac myocytes with increased protein accumulation and myofibrillar organisation characteristic of hypertrophic growth. Real-time quantitative reverse transcription-polymerase chain reaction (RT-PCR) revealed elevated mRNA expression of ANP and brain natriuretic peptide (BNP) but without change in αSkA, consistent with physiological hypertrophic growth. Clenbuterol-treated cultures also showed elevated insulin-like growth factor I (IGF-1) mRNA and activation of the protein kinase Akt. Addition of either IGF-1 receptor-blocking antibodies or LY294002 in order to inhibit phosphatidylinositol 3-kinase, a downstream effector of the IGF-1 receptor, inhibited the hypertrophic response indicating that IGF-1 signalling is required. IGF-1 expression localised primarily to the minor population of cardiac fibroblasts present in the cardiocyte cultures. Together these data show that clenbuterol acts to induce mild cardiac hypertrophy in cardiac myocytes via paracrine signalling involving fibroblast-derived IGF-1.






Similar content being viewed by others
References
Yacoub, M. H. (2001). A novel strategy to maximize the efficacy of left ventricular assist devices as a bridge to recovery. European Heart Journal, 22(7), 534–540.
Birks, E. J., Tansley, P. D., Hardy, J., George, R. S., Bowles, C. T., Burke, M., et al. (2006). Left ventricular assist device and drug therapy for the reversal of heart failure. The New England Journal of Medicine, 355(18), 1873–1884.
Porter, K. E., & Turner, N. A. (2009). Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacology & Therapeutics, 123(2), 255–278.
Souders, C. A., Bowers, S. L., & Baudino, T. A. (2009). Cardiac fibroblast: the renaissance cell. Circulation Research, 105(12), 1164–1176.
Kakkar, R., & Lee, R. T. (2010). Intramyocardial fibroblast myocyte communication. Circulation Research, 106(1), 47–57.
Takeda, N., Manabe, I., Uchino, Y., Eguchi, K., Matsumoto, S., Nishimura, S., et al. (2010). Cardiac fibroblasts are essential for the adaptive response of the murine heart to pressure overload. Journal of Clinical Investigation, 120(1), 254–265.
Petrou, M., Wynne, D. G., Boheler, K. R., & Yacoub, M. H. (1995). Clenbuterol induces hypertrophy of the latissimus dorsi muscle and heart in the rat with molecular and phenotypic changes. Circulation, 92(suppl II), 483–489.
Dorn, G. W., Robbins, J., & Sugden, P. H. (2003). Phenotyping hypertrophy: eschew obfuscation. Circulation Research, 92(11), 1171–1175.
Soppa, G. K., Lee, J., Stagg, M. A., Felkin, L. E., Barton, P. J., Siedlecka, U., et al. (2008). Role and possible mechanisms of clenbuterol in enhancing reverse remodelling during mechanical unloading in murine heart failure. Cardiovascular Research, 77(4), 695–706.
Frey, N., & Olson, E. N. (2003). Cardiac hypertrophy: the good, the bad, and the ugly. Annual Review of Physiology, 65, 45–79.
Duerr, R. L., Huang, S., Miraliakbar, H. R., Clark, R., Chien, K. R., & Ross, J., Jr. (1995). Insulin-like growth factor-1 enhances ventricular hypertrophy and function during the onset of experimental cardiac failure. Journal of Clinical Investigation, 95(2), 619–627.
Fuller, S. J., Mynett, J. R., & Sugden, P. H. (1992). Stimulation of cardiac protein synthesis by insulin-like growth factors. The Biochemical Journal, 282(Pt 1), 85–90.
Delaughter, M. C., Taffet, G. E., Fiorotto, M. L., Entman, M. L., & Schwartz, R. J. (1999). Local insulin-like growth factor I expression induces physiologic, then pathologic, cardiac hypertrophy in transgenic mice. The FASEB Journal, 13(14), 1923–1929.
McMullen, J. R., Shioi, T., Huang, W. Y., Zhang, L., Tarnavski, O., Bisping, E., et al. (2004). The insulin-like growth factor 1 receptor induces physiological heart growth via the phosphoinositide 3-kinase(p110alpha) pathway. The Journal of Biological Chemistry, 279(6), 4782–4793.
Santini, M. P., Tsao, L., Monassier, L., Theodoropoulos, C., Carter, J., Lara-Pezzi, E., et al. (2007). Enhancing repair of the mammalian heart. Circulation Research, 100(12), 1732–1740.
Awede, B. L., Thissen, J. P., & Lebacq, J. (2002). Role of IGF-I and IGFBPs in the changes of mass and phenotype induced in rat soleus muscle by clenbuterol. American Journal of Physiology. Endocrinology and Metabolism, 282(1), E31–E37.
Bhavsar, P. K., Brand, N. J., Yacoub, M. H., & Barton, P. J. R. (1996). Isolation and characterisation of the human cardiac troponin I gene (TNNI3). Genomics, 35, 11–23.
Chomczynski, P. (1987). Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Analytical Biochemistry, 162, 156–159.
Hall, L. J., Kajimoto, Y., Bichell, D., Kim, S. W., James, P. L., Counts, D., et al. (1992). Functional analysis of the rat insulin-like growth factor I gene and identification of an IGF-I gene promoter. DNA and Cell Biology, 11(4), 301–313.
Wong, K., Boheler, K. R., Bishop, J., Petrou, M., & Yacoub, M. (1998). Clenbuterol induces cardiac hypertrophy with normal function, morphological and molecular features. Cardiovascular Research, 37, 115–122.
Sharma, S., Ying, J., Razeghi, P., Stepkowski, S., & Taegtmeyer, H. (2006). Atrophic remodeling of the transplanted rat heart. Cardiology, 105(2), 128–136.
Barton, P. J. R., Felkin, L. E., Birks, E. J., Cullen, M. E., Banner, N. R., Grindle, S., et al. (2005). Myocardial IGF-I gene expression during recovery from heart failure following combined LVAD and clenbuterol therapy. Circulation, 112(Supple 9), 146–150.
Shioi, T., McMullen, J. R., Kang, P. M., Douglas, P. S., Obata, T., Franke, T. F., et al. (2002). Akt/protein kinase B promotes organ growth in transgenic mice. Molecular and Cellular Biology, 22(8), 2799–2809.
Matsui, T., Li, L., Wu, J. C., Cook, S. A., Nagoshi, T., Picard, M. H., et al. (2002). Phenotypic spectrum caused by transgenic overexpression of activated Akt in the heart. The Journal of Biological Chemistry, 277(25), 22896–22901.
Condorelli, G., Drusco, A., Stassi, G., Bellacosa, A., Roncarati, R., Iaccarino, G., et al. (2002). Akt induces enhanced myocardial contractility and cell size in vivo in transgenic mice. Proceedings of the National Academy of Sciences of the United States of America, 99(19), 12333–12338.
Hardt, S. E., & Sadoshima, J. (2002). Glycogen synthase kinase-3beta: a novel regulator of cardiac hypertrophy and development. Circulation Research, 90(10), 1055–1063.
Shioi, T., McMullen, J. R., Tarnavski, O., Converso, K., Sherwood, M. C., Manning, W. J., et al. (2003). Rapamycin attenuates load-induced cardiac hypertrophy in mice. Circulation, 107(12), 1664–1670.
Musaro, A., McCullagh, K., Paul, A., Houghton, L., Dobrowolny, G., Molinaro, M., et al. (2001). Localized Igf-1 transgene expression sustains hypertrophy and regeneration in senescent skeletal muscle. Nature Genetics, 27(2), 195–200.
Musaro, A., McCullagh, K. J., Naya, F. J., Olson, E. N., & Rosenthal, N. (1999). IGF-1 induces skeletal myocyte hypertrophy through calcineurin in association with GATA-2 and NF-ATc1. Nature, 400(6744), 581–585.
Harada, M., Itoh, H., Nakagawa, O., Ogawa, Y., Miyamoto, Y., Kuwahara, K., et al. (1997). Significance of ventricular myocytes and nonmyocytes interaction during cardiocyte hypertrophy: evidence for endothelin-1 as a paracrine hypertrophic factor from cardiac nonmyocytes. Circulation, 96(10), 3737–3744.
King, K. L., Winer, J., Phillips, D. M., Quach, J., Williams, P. M., & Mather, J. P. (1998). Phenylephrine, endothelin, prostaglandin F2alpha' and leukemia inhibitory factor induce different cardiac hypertrophy phenotypes in vitro. Endocrine, 9(1), 45–55.
Kuwahara, K., Saito, Y., Harada, M., Ishikawa, M., Ogawa, E., Miyamoto, Y., et al. (1999). Involvement of cardiotrophin-1 in cardiac myocyte-nonmyocyte interactions during hypertrophy of rat cardiac myocytes in vitro. Circulation, 100(10), 1116–1124.
Sano, M., Fukuda, K., Kodama, H., Pan, J., Saito, M., Matsuzaki, J., et al. (2000). Interleukin-6 family of cytokines mediate angiotensin II-induced cardiac hypertrophy in rodent cardiomyocytes. The Journal of Biological Chemistry, 275(38), 29717–29723.
Oka, T., Xu, J., Kaiser, R. A., Melendez, J., Hambleton, M., Sargent, M. A., et al. (2007). Genetic manipulation of periostin expression reveals a role in cardiac hypertrophy and ventricular remodeling. Circulation Research, 101(3), 313–321.
Rockman, H. A., Koch, W. J., & Lefkowitz, R. J. (2002). Seven transmembrane-spanning receptors and heart function. Nature, 415(6868), 206–212.
Ahmet, I., Krawczyk, M., Heller, P., Moon, C., Lakatta, E. G., & Talan, M. I. (2004). Beneficial effects of chronic pharmacological manipulation of beta-adrenoreceptor subtype signaling in rodent dilated ischemic cardiomyopathy. Circulation, 110(9), 1083–1090.
Bristow, M. R. (2000). beta-adrenergic receptor blockade in chronic heart failure. Circulation, 101(5), 558–569.
Zaugg, M., Xu, W., Lucchinetti, E., Shafiq, S. A., Jamali, N. Z., & Siddiqui, M. A. Q. (2000). B-adrenergic receptor subtypes differentially affect apoptosis in adult rat ventricular myocytes. Circulation, 102, 344–350.
Engelhardt, S., Hein, L., Wiesmann, F., & Lohse, M. J. (1999). Progressive hypertrophy and heart failure in beta1-adrenergic receptor transgenic mice. Proceedings of the National Academy of Sciences of the United States of America, 96(12), 7059–7064.
Milano, C. A., Allen, L. F., Rockman, H. A., Dolber, P. C., McMinn, T. R., Chien, K. R., et al. (1994). Enhanced myocardial function in transgenic mice overexpressing the beta 2-adrenergic receptor. Science, 264(5158), 582–586.
Liggett, S. B., Tepe, N. M., Lorenz, J. N., Canning, A. M., Jantz, T. D., Mitarai, S., et al. (2000). Early and delayed consequences of beta(2)-adrenergic receptor overexpression in mouse hearts: critical role for expression level. Circulation, 101(14), 1707–1714.
Dorn, G. W., Tepe, N. M., Lorenz, J. N., Koch, W. J., & Liggett, S. B. (1999). Low- and high-level transgenic expression of beta2-adrenergic receptors differentially affect cardiac hypertrophy and function in Gαq-overexpressing mice. Proceedings of the National Academy of Sciences of the United States of America, 96(11), 6400–6405.
Chruscinski, A. J., Rohrer, D. K., Schauble, E., Desai, K. H., Bernstein, D., & Kobilka, B. K. (1999). Targeted disruption of the beta2 adrenergic receptor gene. The Journal of Biological Chemistry, 274(24), 16694–16700.
Burniston, J. G., Ng, Y., Clark, W. A., Colyer, J., Tan, L. B., & Goldspink, D. F. (2002). Myotoxic effects of clenbuterol in the rat heart and soleus muscle. Journal of Applied Physiology, 93(5), 1824–1832.
Burniston, J. G., Tan, L. B., & Goldspink, D. F. (2005). beta2-Adrenergic receptor stimulation in vivo induces apoptosis in the rat heart and soleus muscle. Journal of Applied Physiology, 98(4), 1379–1386.
Acknowledgements
This work was supported by the British Heart Foundation and by the Magdi Yacoub Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Pankaj K. Bhavsar and Nigel J. Brand contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Bhavsar, P.K., Brand, N.J., Felkin, L.E. et al. Clenbuterol Induces Cardiac Myocyte Hypertrophy via Paracrine Signalling and Fibroblast-derived IGF-1. J. of Cardiovasc. Trans. Res. 3, 688–695 (2010). https://doi.org/10.1007/s12265-010-9199-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-010-9199-1