Abstract
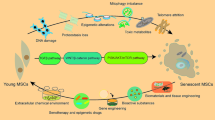
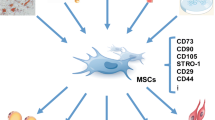
Mesenchymal stem cells (MSCs) are cluster of differentiation 34 (CD34)–CD45-negative nonhematopoietic progenitors derived typically from the stromal fraction of the bone marrow. These stem cells display multipotent properties with a demonstrable differentiation capacity along multiple mesodermal lineages. In the setting of myocardial injury, preclinical studies indicate benefit of both autologous and allogeneic transplantation in line with a recognized immunotolerant profile. Initial clinical experience supports the value of mesenchymal stem-cell-based therapy in ischemic cardiomyopathy. Experience is however limited to naïve mesenchymal stem cells, with efforts underway to identify optimal means of enhancing the cardiogenic potential of transplanted cells through guided cardiopoiesis with the ultimate aim of achieving standardized therapy of the ischemic myocardium.
Similar content being viewed by others
References
Chamberlain, G., Fox, J., Ashton, B., & Middleton, J. (2007). Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells, 25, 2739–2749.
Prockop, D. J. (2007). “Stemness” does not explain the repair of many tissues by mesenchymal stem/multipotent stromal cells (MSCs). Clinical Pharmacology and Therapeutics, 82, 241–243.
Dazzi, F., & Horwood, N. (2007). Potential of mesenchymal stem cell therapy. Current Opinion in Oncology, 19, 650–655.
Mimeault, M., Hauke, R., & Batra, S. K. (2007). Stem cells: A revolution in therapeutics—Recent advances in stem cell biology and their therapeutic applications in regenerative medicine and cancer therapies. Clinical Pharmacology and Therapeutics, 82, 252–264.
Caplan, A. I. (2007). Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. Journal of Cellular Physiology, 213, 341–347.
Martinez, C., Hofmann, T. J., Marino, R., Dominici, M., & Horwitz, E. M. (2007). Human bone marrow mesenchymal stromal cells express the neural ganglioside GD2: A novel surface marker for the identification of MSCs. Blood, 109, 4245–4248.
Toma, C., Pittenger, M. F., Cahill, K. S., Byrne, B. J., & Kessler, P. D. (2002). Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation, 105, 93–38.
Dai, W., Hale, S. L., Martin, B. J., Kuang, J. Q., Dow, J. S., Wold, L. E., & Kloner, R. A. (2005). Allogeneic mesenchymal stem cell transplantation in postinfarcted rat myocardium: short- and long-term effects. Circulation, 112, 214–223.
Martin, B., Senechal, G., Pittenger, M., & Byrne, B. (1999). Human mesenchymal stem cells (hMSCs) exhibit myogenic differentiation when implanted in infarcted rat myocardium. Circulation, 100, 1–54.
Shake, J. G., Gruber, P. J., Baumgartner, W. A., Senechal, G., Meyers, J., Redmond, J. M., et al. (2002). Mesenchymal stem cell implantation in a swine myocardial infarct model: engraftment and functional effects. Annals of Thoracic Surgery, 73, 1919–1926.
Silva, G. V., Litovsky, S., Assad, J. A., Sousa, A. L., Martin, B. J., Vela, D., et al. (2005). Mesenchymal stem cells differentiate into an endothelial phenotype, enhance vascular density, and improve heart function in a canine chronic ischemia model. Circulation, 111, 150–156.
Amado, L. C., Salaris, A. P., Schuleri, K. H., St John, M., Xie, J. S., Cattaneo, S., et al. (2005). Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proceedings of the National Academy of Sciences of the United States of America, 102, 11474–11479.
Chen, S. L., Fang, W. W., Ye, F., Liu, Y. H., Shan, S. J., Zhang, J. J., et al. (2004). Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cells in patients with acute myocardial infarction. American Journal of Cardiology, 94, 92–95.
Hare, J. (2007). Late breaking clinical trials. In Annual meeting of the American College of Cardiology, March 2007.
Le Blanc, K., & Ringdén, O. (2007). Immunomodulation by mesenchymal stem cells and clinical experience. Journal of Internal Medicine, 262, 509–525.
Wang, J. S., Shum-Tim, D., Chedrawy, E., & Chiu, R. C. (2001). The coronary delivery of marrow stromal cells for myocardial regeneration: pathophysiologic and therapeutic implications. Journal of Thoracic and Cardiovascular Surgery, 122, 699–1005.
Yoon, Y. S., Park, J. S., Tkebuchava, T., Luedeman, C., & Losordo, D. W. (2004). Unexpected severe calcification after transplantation of bone marrow cells in acute myocardial infarction. Circulation, 109, 3154–3157.
Bartunek, J., Croissant, J. D., Wijns, W., Gofflot, S., de Lavareille, A., Vanderheyden, M., et al. (2007). Pretreatment of adult bone marrow mesenchymal stem cells with cardiomyogenic growth factors and repair of the chronically infracted myocardium. American Journal of Physiology Heart and Circulatory Physiology, 292, H1095–H1104.
Behfar, A., Zingman, L., Hodgson, D., Rauzier, J., Kane, G., & Terzic, A. (2002). Stem cell differentiation requires a paracrine pathway in the heart. FASEB Journal, 16, 1558–1602.
Olson, E. (2004). A decade of discoveries in cardiac biology. Nature Medicine, 10, 467–474.
Behfar, A., & Terzic, A. (2007). Optimizing adult mesenchymal stem cell for heart repair. Journal of Molecular and Cellular Cardiology, 42, 283–284.
Nakamura, T., & Schneider, M. (2003). The way to a human’s heart is through the stomach. Visceral endoderm-like cells drive human embryonic stem cells to a cardiac fate. Circulation, 107, 2638–2639.
Tomita, S., Li, R.-K., Weisel, R. D., Micjke, D. A. G., & Jia, Z. Q. (1999). Autologous transplantation of bone marrow cells improves damaged heart function. Circulation, 100(Suppl), II247–II256.
Tomita, S., Mickle, D. A. G., Weissel, R. D., et al. (2002). Improved heart function with myogenesis and angiogenesis after autologous porcine bone marrow stromal cell transplantation. Journal of Thoracic and Cardiovascular Surgery, 123, 1132–1140.
Li, X. H., Yu, X. Y., Lin, Q. X., Deng, C. Y., Shan, Z. X., Yang, M., et al. (2007). Bone marrow mesenchymal stem cells differentiate into functional cardiac phenotypes by cardiac microenvironment. Journal of Molecular and Cellular Cardiology, 42, 295–303.
Behfar, A., Perez-Terzic, C., Faustino, R. S., Arrell, D. K., Hodgson, D. M., Yamada, S., et al. (2007). Cardiopoietic programming of embryonic stem cells for tumor-free heart repair. Journal of Experimental Medicine, 204, 405–420.
Behfar, A., & Terzic, A. (2006). Derivation of a cardiogenic population from human mesenchymal stem cells yields cardiac progeny. Nature Clinical Practice Cardiovascular Medicine, 3, S78–S82.
Faustino, R. S., Behfar, A., Perez-Terzic, C., & Terzic, A. (2008). Genomic chart guiding embryonic stem cell cardiopoiesis. Genome Biology, 9, R6.
Arrell, D. K., Niederlander, N. J., Faustino, R. S., Behafr, A., & Terzic, A. (2008). Cardioinductive network guiding stem cell differentiation revealed by proteomic cartography of TNF{alpha}-primed endodermal secretome. Stem Cells, 26, 387–400.
Mazhari, R., & Hare, J. M. (2007). Mechanisms of action of mesenchymal stem cells in cardiac repair: Potential influences on the cardiac stem cell niche. Nature Clinical Practice Cardiovascular Medicine, 4(Suppl1), S21–S26.
Acknowledgements
Drs. J Bartunek, M. Vanderheyden, and W. Wijns are members of a nonprofit organization which is a founding member of Cardio3Biosciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bartunek, J., Behfar, A., Vanderheyden, M. et al. Mesenchymal Stem Cells and Cardiac Repair: Principles and Practice. J. of Cardiovasc. Trans. Res. 1, 115–119 (2008). https://doi.org/10.1007/s12265-008-9021-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-008-9021-5