Abstract
Objective
To construct recombinant lentiviral vectors for gene delivery of the glial cell line-derived neurotrophic factor (GDNF), and evaluate the neuroprotective effect of GDNF on lactacystin-damaged PC12 cells by transfecting it into bone marrow stromal cells (BMSCs).
Methods
PLenti6/V5-GDNF plasmid was set up by double restriction enzyme digestion and ligation, and then the plasmid was transformed into Top10 cells. Purified pLenti6/V5-GDNF plasmids from the positive clones and the packaging mixture were cotransfected to the 293FT packaging cell line by Lipofectamine2000 to produce lentivirus, then the concentrated virus was transduced to BMSCs. Overexpression of GDNF in BMSCs was tested by RT-PCR, ELISA and immunocytochemistry, and its neuroprotection for lactacystin-damaged PC12 cells was evaluated by MTT assay.
Results
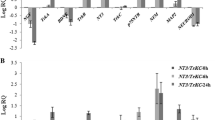
Virus stock of GDNF was harvested with the titer of 5.6×105 TU/mL. After transduction, GDNF-BMSCs successfully secreted GDNF to supernatant with higher concentration (800 pg/mL) than BMSCs did (less than 100 pg/mL). The supernatant of GDNF-BMSCs could significantly alleviate the damage of PC12 cells induced by lactacystin (10 μmol/L).
Conclusion
Overexpression of lentivirus-mediated GDNF in the BMSCs cells can effectively protect PC12 cells from the injury by the proteasome inhibitor.
摘要
目的
建立慢病毒介导的胶质细胞系源性神经营养因子(glial cell line-derived neurotrophic factor, GDNF)表达系统, 体外感染骨髓基质细胞, 检测过表达GDNF对蛋白酶抑制剂引起的PC12细胞损伤的神经保护作用。
方法
经双酶切和T4连接酶构建pLenti6/V5-GDNF表达质粒, 经293FT细胞包装产生高滴度病毒。 用RT-PCR、 ELISA和免疫细胞化学方法检测感染骨髓基质细胞(bone marrow stromal cells, BMSCs)后GDNF的表达, 并检测过表达GDNF对蛋白酶抑制剂乳胞素(lactacystin)引起的PC12细胞损伤的保护作用。
结果
成功构建pLenti6/V5-GDNF表达质粒, 获得高滴度具有感染能力的病毒储存液(5.6 × 105 TU/mL)。 BMSCs体外被感染后能大量分泌GDNF(接近800 pg/mL), 过表达GDNF能减轻乳胞素(10 μmol/L)引起的PC12细胞损伤。
结论
慢病毒介导的GDNF转染骨髓基质细胞后能分泌具有生物学活性的GDNF, 对蛋白酶体抑制剂引起的PC12细胞损伤有保护作用。
Similar content being viewed by others
References
Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F. GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science 1993, 260: 1130–1132.
Costa S, Iravani MM, Pearce RK, Jenner P. Glial cell linederived neurotrophic factor concentration dependently improves disability and motor activity in MPTP-treated common marmosets. Eur J Pharmacol 2001, 412: 45–50.
Bowenkamp KE, Lapchak PA, Hoffer BJ, Miller PJ, Bickford PC. Intracerebroventricular glial cell line-derived neurotrophic factor improves motor function and supports nigrostriatal dopamine neurons in bilaterally 6-hydroxydopamine lesioned rats. Exp Neurol 1997, 145: 104–117.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D, et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998, 72: 8463–8471.
Chen Q, Long Y, Yuan X, Zou L, Sun J, Chen S, et al. Protective effects of bone marrow stromal cell transplantation in injured rodent brain: synthesis of neurotrophic factors. J Neurosci Res 2005, 80: 611–619.
Leroy E, Boyer R, Auburger G, Leube B, Ulm G, Mezey E, et al. The ubiquitin pathway in Parkinson’s disease. Nature 1998, 395: 451–452.
Kitada T, Asakawa S, Hattori N, Matsumine H, Yamamura Y, Minoshima S, et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 1998, 392: 605–608.
McNaught KS, Jenner P. Proteasomal function is impaired in substantia nigra in Parkinson’s disease. Neurosci Lett 2001, 297: 191–194.
Wu S, Suzuki Y, Ejiri Y, Noda T, Bai H, Kitada M, et al. Bone marrow stromal cells enhance differentiation of cocultured neurosphere cells and promote regeneration of injured spinal cord. J Neurosci Res 2003, 72: 343–351.
Lou S, Gu P, Chen F, He C, Wang M, Lu C. The effect of bone marrow stromal cells on neuronal differentiation of mesencephalic neural stem cells in Sprague-Dawley rats. Brain Res 2003, 968: 114–121.
Ye M, Chensheng D, Qi C, Guoqiang L, Liang L, Xujie Y. Expression of mRNAs and prote in for glial cell line2der ived neurotrophic factors in bone marrow stromal cells of SD rat. Neurosci Bull 2005, 21: 23–27.
Park KW, Eglitis MA, Mouradian MM. Protection of nigral neurons by GDNF-engineered marrow cell transplantation. Neurosci Res 2001, 40: 315–323.
Elkon H, Melamed E, Offen D. Oxidative stress, induced by 6-hydroxydopamine, reduces proteasome activities in PC12 cells: implications for the pathogenesis of Parkinson’s disease. J Mol Neurosci 2004, 24: 387–400.
Moser SS, Besler-Panos W. Pharmacotherapy for Parkinson’s disease: current options, promising future therapies. JAAPA 2006, 19: 38–44.
Wirth T, Yla-Herttuala S. Gene technology based therapies in the brain. Adv Tech Stand Neurosurg 2006, 31: 3–32.
Limberis MP, Wilson JM. Adeno-associated virus serotype 9 vectors transduce murine alveolar and nasal epithelia and can be readministered. Proc Natl Acad Sci USA 2006, 103: 12993–12998.
Bankiewicz KS, Forsayeth J, Eberling JL, Sanchez-Pernaute R, Pivirotto P, Bringas J, et al. Long-term clinical improvement in MPTP-lesioned primates after gene therapy with AAV-hAADC. Mol Ther 2006, 14: 564–570.
Zufferey R, Dull T, Mandel RJ, Bukovsky A, Quiroz D, Naldini L, et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J Virol 1998, 72: 9873–9880.
Kang UJ, Fisher LJ, Joh TH, O’Malley KL, Gage FH. Regulation of dopamine production by genetically modified primary fibroblasts. J Neurosci 1993, 13: 5203–5211.
Dass B, Olanow CW, Kordower JH. Gene transfer of trophic factors and stem cell grafting as treatments for Parkinson’s disease. Neurology 2006, 66(10 Suppl 4): S89–S103.
Lu J, Moochhala S, Moore XL, Ng KC, Tan MH, Lee LK, et al. Adult bone marrow cells differentiate into neural phenotypes and improve functional recovery in rats following traumatic brain injury. Neurosci Lett 2006, 398: 12–17.
Garcia R, Aguiar J, Alberti E, de la Cuetara K, Pavon N. Bone marrow stromal cells produce nerve growth factor and glial cell line-derived neurotrophic factors. Biochem Biophys Res Commun 2004, 316: 753–754.
Qiu JH, Asai A, Chi S, Saito N, Hamada H, Kirino T. Proteasome inhibitors induce cytochrome c-caspase-3-like protease-mediated apoptosis in cultured cortical neurons. J Neurosci 2000, 20: 259–265.
Hyun DH, Lee M, Halliwell B, Jenner P. Proteasomal inhibition causes the formation of protein aggregates containing a wide range of proteins, including nitrated proteins. J Neurochem 2003, 86: 363–373.
Lev N, Melamed E, Offen D. Proteasomal inhibition hypersensitizes differentiated neuroblastoma cells to oxidative damage. Neurosci Lett 2006, 399: 27–32.
Omura S, Fujimoto T, Otoguro K, Matsuzaki K, Moriguchi R, Tanaka H, et al. Lactacystin, a novel microbial metabolite, induces neuritogenesis of neuroblastoma cells. J Antibiot 1991, 44: 113–116.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Su, YR., Wang, J., Wu, JJ. et al. Overexpression of lentivirus-mediated glial cell line-derived neurotrophic factor in bone marrow stromal cells and its neuroprotection for the PC12 cells damaged by lactacystin. Neurosci. Bull. 23, 67–74 (2007). https://doi.org/10.1007/s12264-007-0010-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-007-0010-5
Keywords
- Parkinson’s disease
- proteasome inhibitor
- glial cell line-derived neurotrophic factor
- lentivirus
- gene therapy
- bone marrow stromal cells