Abstract
Background
There is ambiguity about the use of antibiotic prophylaxis in inguinal mesh hernioplasty. We have tried to assess the efficacy of antibiotic prophylaxis in this procedure.
Materials and methods
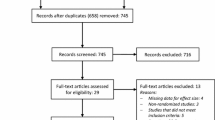
A randomized double blind placebo controlled study was conducted which included 55 patients who underwent an inguinal mesh hernioplasty over a 2 year period. The patients were evaluated for the status of the suture line as well as the presence of wound infection.
Results
Out of 55 patients 29 were randomized to the antibiotic arm and 26 to the placebo group. The groups were well matched for all variables studied excluding wound infections, which occurred at a rate of 10.34% (n = 3) in the antibiotic group and 15.38% (n = 4) in the placebo arm, (p > 0.01).
Conclusion
This study did not document any statistically significant difference observed between those who received antibiotics and those receiving placebo in terms of any of the prognostic end points evaluated for Lichtenstein mesh hernioplasty.
Similar content being viewed by others
References
Burger JW, Luijendijk RW, Hop WC, Halm JA, Verdaasdonk EG, Jeekel J (2004) Long term follow-up of a randomized controlled trial of suture versus mesh repair of incisional hernia. Ann Surg 240:578–585
Stephensen BM (2003) Complications of open groin hernia repair. Surg Clin North Am 83:1255–1278
Gristina AG, Costerton JW (1984) Bacterial adherence and the glycocalyx and their role in musculoskeletal infections Orthop Clin North Am 15:517–535
Aufenacker TJ, Koelemay MJW, Goulma DJ, Simons MP (2005) Systematic review and meta analysis of the effectiveness of antibiotic prophylaxis in the prevention of wound infection after mesh repair of abdominal wall hernia. Br J Surg 93:5–10
Perez AR, Roxas MF, Hilvano SS (2005) A randomized double blind, placebo controlled trial to determine effectiveness of antibiotic prophylaxis for tension free mesh hernioraphy. J Am Coll Surg 200:393–398
Teresa HC, Robert GP, William MJ, William JR, Grace TE (1992) CDC Definitions of Nosocomial Surgical Site Infections, 1992: A Modification of CDC Definitions of Surgical Wound Infections
Patino JF, Garcia-Herreros LG, Zundel N (1998) Inguinal hernia repair: The Nyhus posterior peritoneal operation. Surg Cil North Am 78:1063–1074
Yerdel MA, Akin EB, Dolalan S, et al. (2001) Effect of single dose prophylactic ampicillin and sulbactam on wound infection after tension-free inguinal hernia repair with polypropylene mesh: the randomized, double blind, prospective trial. Ann Surg 233:26–33
Sanabria A, Dominguez LC, Valdivieso E, Gomez G (2007) Prophylactic antibiotic for mesh hernioplasty. A meta analysis. Ann Surg 245:392–396
Sanchez-Manuel FJ, Lozano-García J, Seco-Gil JL (2007) Antibiotic prophylaxis for hernia repair. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD003769. DOI: 10.1002/14651858.CD003769.pub3.
Tzovaras G, Delikoukos S, Christodoulides G, Spyridakis M, Mantzos F, Tepetes K, et al. (2006) Role of Antibiotic Prophylaxis in Tension-Free Mesh Inguinal Hernia Repair: Results of a single-centre prospective randomised trial 6:236–239
Platt R, Zaleznik DF, Hopkins CC, Dellinger EP, Karchmer AW, Bryan CS, et al. (1990) Perioperative antibiotic prophylaxis for hernioraphy and breast surgery. N Engl J Med 322:153–160
Taylor SG, Dwyer PJO (1999) Chronic groin sepsis following tension free inguinal hernioplasty. Br J Surg 86:562–565
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thakur, L., Upadhyay, S., Peters, N.J. et al. Prophylactic antibiotic usage in patients undergoing inguinal mesh hernioplasty — A clinical study. Indian J Surg 72, 240–242 (2010). https://doi.org/10.1007/s12262-010-0051-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-010-0051-2