Abstract
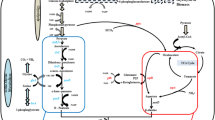
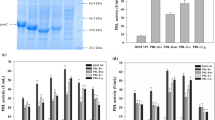
The safe production of β-alanine (BA) has attracted significant attention by its multifaceted applications in pharmaceutical, polymer, and nutrition. The extant high-yielding chemical and enzymatic methods of BA synthesis are handicapped by raw materials derived from petroleum resources, harsh reaction conditions, and catalyst instability. Consequently, this study explored a safe and alternative route via microbial fermentation, utilizing metabolic engineering of Bacillus megaterium to produce BA. The Bacillus subtilis panD gene (encoding L-aspartate-α-decarboxylase) was codon-optimized and overexpressed, which yielded 0.13 ± 0.05 g/L BA. Aspartate ammonia-lyase (AspA) and aspartate aminotransferase (AspB) based pathways were examined for BA production from glucose. NADH-dependent glutamate dehydrogenase (gdh) was used to regenerate the cofactor NAD+ in the pathway with AspB. Dosing of the rate liming panD showed a positive effect on BA production. The BA titer was further increased to 1.4 ± 0.06 g/L by over-expression of phosphoenolpyruvate carboxylase (PPC). Optimizing (NH4)2SO4 Pyridoxine, and NaHCO3 allowed the production of 2.41 ± 0.15 g/L BA. Fed-batch fermentation of the final strain allowed 17.60 ± 0.13 g/L BA production in 22 h. The present study has effectively unlocked the potential of engineering the B. megaterium for the sustainable production of the other ASP (L-aspartic acid) and BA-derived products at a large scale.
Similar content being viewed by others
References
Spierling, S., E. Knüpffer, H. Behnsen, M. Mudersbach, H. Krieg, S. Springer, S. Albrecht, C. Herrmann, and H.-J. Endres (2018) Bio-based plastics — a review of environmental, social and economic impact assessments. J. Clean. Prod. 185: 476–491.
Francois, J. M., C. Alkim, and N. Morin (2020) Engineering microbial pathways for production of bio-based chemicals from lignocellulosic sugars: current status and perspectives. Biotechnol. Biofuels 13: 118.
Choi, K. R., S. Jiao, and S. Y. Lee (2020) Metabolic engineering strategies toward production of biofuels. Curr. Opin. Chem. Biol. 59: 1–14.
Wendisch, V. F. (2020) Metabolic engineering advances and prospects for amino acid production. Metab. Eng. 58: 17–34.
Ko, Y.-S., J. W. Kim, J. A. Lee, T. Han, G. B. Kim, J. E. Park, and S. Y. Lee (2020) Tools and strategies of systems metabolic engineering for the development of microbial cell factories for chemical production. Chem. Soc. Rev. 49: 4615–4636.
Wang, L., Y. Mao, Z. Wang, H. Ma, and T. Chen (2021) Advances in biotechnological production of β-alanine. World J. Microbiol. Biotechnol. 37: 79.
Song, C. W., J. Lee, Y.-S. Ko, and S. Y. Lee (2015) Metabolic engineering of Escherichia coli for the production of 3-aminopropionic acid. Metab. Eng. 30: 121–129.
Shen, Y., L. Zhao, Y. Li, L. Zhang, and G. Shi (2014) Synthesis of β-alanine from L-aspartate using L-aspartate-α-decarboxylase from Corynebacterium glutamicum. Biotechnol. Lett. 36: 1681–1686.
Li, H., X. Lu, K. Chen, J. Yang, A. Zhang, X. Wang, and P. Ouyang (2018) β-alanine production using whole-cell biocatalysts in recombinant Escherichia coli. Mol. Catal. 449: 93–98.
Piao, X., L. Wang, B. Lin, H. Chen, W. Liu, and Y. Tao (2019) Metabolic engineering of Escherichia coli for production of L-aspartate and its derivative β-alanine with high stoichiometric yield. Metab. Eng. 54: 244–254.
Pei, W., J. Zhang, S. Deng, F. Tigu, Y. Li, Q. Li, Z. Cai, and Y. Li (2017) Molecular engineering of L-aspartate-α-decarboxylase for improved activity and catalytic stability. Appl. Microbiol. Biotechnol. 101: 6015–6021.
Gao, Y., Z. Liu, K. Liu, Z. Zhou, and W. Cui (2017) [Biocatalytic access to β-alanine by a two-enzyme cascade synthesis]. Sheng Wu Gong Cheng Xue Bao 33: 875–879.
Zhang, T., R. Zhang, M. Xu, X. Zhang, T. Yang, F. Liu, S. Yang, and Z. Rao (2018) Glu56Ser mutation improves the enzymatic activity and catalytic stability of Bacillus subtilis L-aspartate α-decarboxylase for an efficient β-alanine production. Process Biochem. 70: 117–123.
Mo, Q., A. Mao, Y. Li, and G. Shi (2019) Substrate inactivation of bacterial L-aspartate α-decarboxylase from Corynebacterium jeikeium K411 and improvement of molecular stability by saturation mutagenesis. World J. Microbiol. Biotechnol. 35: 62.
Yu, X.-J., C.-Y. Huang, X.-D. Xu, H. Chen, M.-J. Liang, Z.-X. Xu, H.-X. Xu, and Z. Wang (2020) Protein engineering of a pyridoxal-5′-phosphate-dependent l-aspartate-α-decarboxylase from Tribolium castaneum for β-alanine production. Molecules 25: 1280.
Feng, Z., J. Zhang, G. Chen, Y. Ge, X. Zhang, and H. Zhu (2019) Extracellular expression of L-aspartate-α-decarboxylase from Bacillus tequilensis and its application in the biosynthesis of β-alanine. Appl. Biochem. Biotechnol. 189: 273–283.
Qian, Y., J. Liu, W. Song, X. Chen, Q. Luo, and L. Liu (2018) Production of β-alanine from fumaric acid using a dual-enzyme cascade. ChemCatChem 10: 4984–4991.
Tadi, S. R. R., G. Nehru, and S. Sivaprakasam (2021) Combinatorial approach for improved production of whole-cell 3-aminopropionic acid in recombinant Bacillus megaterium: codon optimization, gene duplication and process optimization. 3 Biotech 11: 333.
Liu, Z., W. Zheng, W. Ye, C. Wang, Y. Gao, W. Cui, and Z. Zhou (2019) Characterization of cysteine sulfinic acid decarboxylase from Tribolium castaneum and its application in the production of β-alanine. Appl. Microbiol. Biotechnol. 103: 9443–9453.
Wang, L., X. Piao, S. Cui, M. Hu, and Y. Tao (2020) Enhanced production of β-alanine through co-expressing two different subtypes of l-aspartate-α-decarboxylase. J. Ind. Microbiol. Biotechnol. 47: 465–474.
Zou, X., L. Guo, L. Huang, M. Li, S. Zhang, A. Yang, Y. Zhang, L. Zhu, H. Zhang, J. Zhang, and Z. Feng (2020) Pathway construction and metabolic engineering for fermentative production of β-alanine in Escherichia coli. Appl. Microbiol. Biotechnol. 104: 2545–2559.
Xu, J., Y. Zhu, and Z. Zhou (2021) Systematic engineering of the rate-limiting step of β-alanine biosynthesis in Escherichia coli. Electron. J. Biotechnol. 51: 88–94.
Xu, J., L. Zhou, M. Yin, and Z. Zhou (2021) Novel mode engineering for β-alanine production in Escherichia coli with the guide of adaptive laboratory evolution. Microorganisms 9: 600.
Miao, L., Y. Li, and T. Zhu (2021) Metabolic engineering of methylotrophic Pichia pastoris for the production of β-alanine. Bioresour. Bioprocess. 8: 89.
Biedendieck, R., C. Borgmeier, B. Bunk, S. Stammen, C. Scherling, F. Meinhardt, C. Wittmann, and D. Jahn (2011) Systems biology of recombinant protein production using Bacillus megaterium. Methods Enzymol. 500: 165–195.
Bunk, B., A. Schulz, S. Stammen, R. Münch, M. J. Warren, M. Rohde, D. Jahn, and R. Biedendieck (2010) A short story about a big magic bug. Bioeng. Bugs 1: 85–91.
Kalbarczyk, K. Z., E. J. Mazeau, K. M. Rapp, N. Marchand, M. A. Koffas, and C. H. Collins (2018) Engineering Bacillus megaterium strains to secrete cellulases for synergistic cellulose degradation in a microbial community. ACS Synth. Biol. 7: 2413–2422.
Gädke, J., J.-W. Thies, L. Kleinfeldt, A. Kalinin, G. Starke, A. Lakowitz, R. Biedendieck, G. Garnweitner, A. Dietzel, and R. Krull (2017) Integrated in situ-purification of recombinant proteins from Bacillus megaterium cultivation using SPION in stirred tank reactors. Biochem. Eng. J. 126: 58–67.
Tadi, S. R. R., G. Nehru, A. M. Limaye, and S. Sivaprakasam (2022) High-level expression and optimization of pantoate-β-alanine ligase in Bacillus megaterium for the enhanced biocatalytic production of D-pantothenic acid. J. Food Sci. Technol. 59: 917–926.
Moore, S. J., M. J. Mayer, R. Biedendieck, E. Deery, and M. J. Warren (2014) Towards a cell factory for vitamin B12 production in Bacillus megaterium: bypassing of the cobalamin riboswitch control elements. N. Biotechnol. 31: 553–561.
Gerber, A., M. Milhim, P. Hartz, J. Zapp, and R. Bernhardt (2016) Genetic engineering of Bacillus megaterium for high-yield production of the major teleost progestogens 17α,20β-di-and 17α,20β,21α-trihydroxy-4-pregnen-3-one. Metab. Eng. 36: 19–27.
Tadi, S. R. R., S. D. Ravindran, R. Balakrishnan, and S. Sivaprakasam (2021) Recombinant production of poly-(3-hydroxybutyrate) by Bacillus megaterium utilizing millet bran and rapeseed meal hydrolysates. Bioresour. Technol. 326: 124800.
Grage, K., P. McDermott, and B. H. Rehm (2017) Engineering Bacillus megaterium for production of functional intracellular materials. Microb. Cell Fact. 16: 211.
Nehru, G., S. R. R. Tadi, A. M. Limaye, and S. Sivaprakasam (2020) Production and characterization of low molecular weight heparosan in Bacillus megaterium using Escherichia coli K5 glycosyltransferases. Int. J. Biol. Macromol. 160: 69–76.
Corsa, V., A. Negro, and S. Bisicchia (2017) Process for the production of hyaluronic acid in Escherichia coli or Bacillus megaterium. US Patent 9,695,453.
Williams, A., K. S. Gedeon, D. Vaidyanathan, Y. Yu, C. H. Collins, J. S. Dordick, R. J. Linhardt, and M. A. Koffas (2019) Metabolic engineering of Bacillus megaterium for heparosan biosynthesis using Pasteurella multocida heparosan synthase, PmHS2. Microb. Cell Fact. 18: 132.
Ortiz, A. and E. Sansinenea (2020) Succinic acid production as secondary metabolite from Bacillus megaterium ELI24. Nat. Prod. J. 10: 153–157.
Ghosh, S., U. Mohan, and U. C. Banerjee (2016) Studies on the production of shikimic acid using the aroK knockout strain of Bacillus megaterium. World J. Microbiol. Biotechnol. 32: 127.
Gibson, D. G., L. Young, R.-Y. Chuang, J. C. Venter, C. A. Hutchison 3rd, and H. O. Smith (2009) Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 6: 343–345.
Borodina, I., K. R. Kildegaard, N. B. Jensen, T. H. Blicher, J. Maury, S. Sherstyk, K. Schneider, P. Lamosa, M. J. Herrgård, I. Rosenstand, F. Öberg, J. Forster, and J. Nielsen (2015) Establishing a synthetic pathway for high-level production of 3-hydroxypropionic acid in Saccharomyces cerevisiae via β-alanine. Metab. Eng. 27: 57–64.
Ko, Y.-S., J. W. Kim, T. U. Chae, C. W. Song, and S. Y. Lee (2020) A novel biosynthetic pathway for the production of acrylic acid through β-alanine route in Escherichia coli. ACS Synth. Biol. 9: 1150–1159.
Song, C. W., J. W. Kim, I. J. Cho, and S. Y. Lee (2016) Metabolic engineering of Escherichia coli for the production of 3-hydroxypropionic acid and malonic acid through β-alanine route. ACS Synth. Biol. 5: 1256–1263.
Qian, Y., C. Lu, J. Liu, W. Song, X. Chen, Q. Luo, L. Liu, and J. Wu (2020) Engineering protonation conformation of l-aspartate-α-decarboxylase to relieve mechanism-based inactivation. Biotechnol. Bioeng. 117: 1607–1614.
Stammen, S., B. K. Müller, C. Korneli, R. Biedendieck, M. Gamer, E. Franco-Lara, and D. Jahn (2010) High-yield intra- and extracellular protein production using Bacillus megaterium. Appl. Environ. Microbiol. 76: 4037–4046.
Su, L., Y. Huang, and J. Wu (2015) Enhanced production of recombinant Escherichia coli glutamate decarboxylase through optimization of induction strategy and addition of pyridoxine. Bioresour. Technol. 198: 63–69.
Huang, Y., L. Su, and J. Wu (2016) Pyridoxine supplementation improves the activity of recombinant glutamate decarboxylase and the enzymatic production of gama-aminobutyric acid. PLoS One 11: e0157466.
Acknowledgements
The Department of Biotechnology, Govt. of India financially supported this research (Grant No — BT/PR15946/NER/95/485/2016). Authors acknowledge the Ministry of Human Resource and Development, New Delhi, India, for fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Tadi, S.R.R., Nehru, G. & Sivaprakasam, S. Metabolic Engineering of Bacillus megaterium for the Production of β-alanine. Biotechnol Bioproc E 27, 909–920 (2022). https://doi.org/10.1007/s12257-022-0077-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0077-x