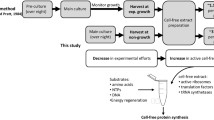
Abstract
The open nature of cell-free synthesis allows customization of the reaction conditions for given target molecules using diverse biological and non-biological substances. This study demonstrates that non-ionic detergents can be used to enhance translation during the synthesis of peptides in a cell-free system derived from an Escherichia coli extract. The yield of the antimicrobial peptide Cecropin P1 was markedly increased in the presence of detergents. The stimulatory effect of detergents was not limited to the Cecropin P1 peptide, but the detergent also enhanced the translation of other antimicrobial peptides. Furthermore, the enhanced translation rate by detergents was maintained for extended periods by a continuous exchange cell-free synthesis reaction, leading to production of antimicrobial peptides with markedly improved yields.
Similar content being viewed by others
References
Albericio, F. (2004) Developments in peptide and amide synthesis. Curr. Opin. Chem. Biol. 8: 211–221.
Park, J. C., D. H. Kim, C. S. Kim, and J. H. Seo (2018) R5 peptide–based biosilicification using methyltrimethoxysilane. Biotechnol. Bioprocess Eng. 23: 11–15.
Munk, J. K., C. Ritz, F. P. Fliedner, N. Frimodt–Møller, and P. R. Hansen (2014) Novel method to identify the optimal antimicrobial peptide in a combination matrix, using anoplin as an example. Antimicrob. Agents Chemother. 58: 1063–1070.
Feng, X., W. Xu, P. Qu, X. Li, L. Xing, D. Liu, J. Jiao, J. Wang, Z. Li, and C. Liu (2015) High–yield recombinant expression of the chicken antimicrobial peptide fowlicidin–2 in Escherichia coli. Biotechnol. Prog. 31: 369–374.
Bray, B. L. (2003) Large–scale manufacture of peptide therapeutics by chemical synthesis. Nat. Rev. Drug Discov. 2: 587–593.
Martemyanov, K. A., V. A. Shirokov, O. V. Kurnasov, A. T. Gudkov, and A. S. Spirin (2001) Cell–free production of biologically active polypeptides: application to the synthesis of antibacterial peptide Cecropin. Protein Expr. Purif. 21: 456–461.
Cui, H., M. J. Webber, and S. I. Stupp (2010) Self–assembly of peptide amphiphiles: from molecules to nanostructures to biomaterials. Biopolymers 94: 1–18.
Loose, C. R., R. S. Langer, and G. N. Stephanopoulos (2007) Optimization of protein fusion partner length for maximizing in vitro translation of peptides. Biotechnol. Prog. 23: 444–451.
Lee, K. H., Y. C. Kwon, S. J. Yoo, and D. M. Kim (2010) Ribosomal synthesis and in situ isolation of peptide molecules in a cell–free translation system. Protein Exp. Purif. 71: 16–20.
Yin, G. and J. R. Swartz (2004) Enhancing multiple disulfide bonded protein folding in a cell–free system. Biotechnol. Bioeng. 86: 188–195.
Lee, K. Y., K. H. Lee, J. W. Park, and D. M. Kim (2012) Flexible programming of cell–free protein synthesis using magnetic beadimmobilized plasmids. PLoS One 7: e34429.
Klammt, C., D. Schwarz, K. Fendler, W. Haase, V. Dotsch, and F. Bernhard (2005) Evaluation of detergents for the soluble expression of alpha–helical and beta–barrel–type integral membrane proteins by a preparative scale individual cell–free expression system. FEBS J. 272: 6024–6038.
Kim, T. W., J. W. Keum, I. S. Oh, C. Y. Choi, C. G. Park, and D. M. Kim (2006) Simple procedures for the construction of a robust and cost–effective cell–free protein synthesis system. J. Biotechnol. 126: 554–561.
Sipos, D., M. Andersson, and A. Ehrenberg (1992) The structure of the mammalian antibacterial peptide cecropin–P1 in solution, determined by proton–NMR. Eur. J. Biochem. 209: 163–169.
Richardson, S. M., S. J. Wheelan, R. M. Yarrington, and J. D. Boeke (2006) GeneDesign: rapid, automated design of multikilobase synthetic genes. Genome Res. 16: 550–556.
Ahn, J. H., K. H. Lee, J. W. Shim, E. Y. Lee, and D. M. Kim (2013) Streamlined cell–free protein synthesis from sequence information. Biotechnol. Bioproc. Eng. 18: 1101–1108.
Moerman, L., S. Bosteels, W. Noppe, J. Willems, E. Clynen, L. Schoofs, K. Thevissen, J. Tytgat, J. V. Eldere, J. V. D. Walt, and F. Verdonck (2002) Antibacterial and antifungal properties of alpha–helical, cationic peptides in the venom of scorpions from southern Africa. Eur. J. Biochem. 19: 4799–4810.
Reed, W. A., K. L. White, F. M. Enright, J. Holck, G. W. Jeffers, and J. M. Jaynes (1992) Enhanced in vitro growth of murine fibroblast cells and preimplantation embryos cultured in medium supplemented with an amphipathic peptide. Mol. Reprod. Dev. 31: 106–113.
Son, J. M., J. H. Ahn, M. Y. Hwang, C. G. Park, C. Y. Choi, and D. M. Kim (2006) Enhancing the efficiency of cell–free protein synthesis through the polymerase–chain–reaction–based addition of a translation enhancer sequence and the in situ removal of the extra amino acid residues. Anal. Biochem. 351: 187–192.
Schagger, H. (2006) Tricine–SDS–PAGE. Nat. Protoc. 1: 16–22.
Kim, D. M. and C. Y. Choi (1996) A semicontinuous prokaryotic coupled transcription/ translation system using a dialysis membrane. Biotechnol. Prog. 12: 645–649.
Hahn, G. H. and D. M. Kim (2006) Production of milligram quantities of recombinant proteins from PCR–amplified DNAs in a continuous–exchange cell–free protein synthesis system. Anal. Biochem. 355: 151–153.
Ahn, J. H., H. S. Chu, T. W. Kim, I. S. Oh, C. Y. Choi, G. H. Hahn, C. G. Park, and D. M. Kim (2005) Cell–free synthesis of recombinant proteins from PCR–amplified genesat a comparable productivity to that of plasmid–based reactions. Biochem. Biophys. Res. Commun. 338: 1346–1352.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Go, SY., Lee, KH. & Kim, DM. Detergent-assisted Enhancement of the Translation Rate during Cell-free Synthesis of Peptides in an Escherichia coli Extract. Biotechnol Bioproc E 23, 679–685 (2018). https://doi.org/10.1007/s12257-018-0418-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-018-0418-y