Abstract
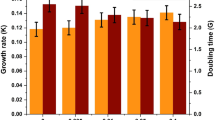
The effects of iron sources with different speciation and anionic moieties (ferric chloride, ferrous chloride, ferric EDTA, ferrous EDTA, ferric ammonium sulfate, and ferrous ammonium sulfate) on the cell growth and the production of energy storage (lipid and carbohydrate) from Dunaliella tertiolecta were investigated. The influence of iron dosage was also compared in the range from 0.65 mg/L (1X) to 6.5 mg/L (10X) as Fe concentration. Best cell growth rate was achieved when ferrous ammonium sulfate was used. Ferric EDTA resulted in higher lipid content than other iron sources, while ferrous ammonium sulfate favored the accumulation of carbohydrate among six iron sources. The accumulations of lipid and carbohydrate as energy storage competed each other and thus both contents did not increase together. In the presence of ferric EDTA, lipid content is increasing, while carbohydrate content is decreasing. On the contrary, lipid content is decreasing while carbohydrate is increasing in the presence of ferric ammonium sulfate. Because the overall carbohydrate content was larger than that of lipid, bioethanol production would be more advantageous than biodiesel production with the present D. tertiolecta strain if the carbohydrate in D. tertiolecta contains a high fraction of glucose with a good saccharification yield.
Similar content being viewed by others
References
Wang, B., Y. Li, N. Wu, and C. Q. Lan (2008) CO2 bio-mitigation using microalgae. Appl. Microbiol. Biotechnol. 79: 707–718.
O. K. Lee, D. H. Sung, C. G. Lee, and E. Y. Lee (2015) Sustainable production of liquid biofuels from renewable microalgae biomass. J. Ind. Eng. Chem. 29: 24–31.
Kwon, M. H. and S. H. Yeom (2015) Optimization of one-step extraction and transesterification process for biodiesel production from the marine microalga Nannochloropsis sp. KMMCC 290 cultivated in a raceway pond. Bioetchnol. Bioproc. Eng. 20: 276–283.
Kim, J. and J.-Y. Lee (2016) Enhanced autotrophic growth of Nannochloris sp. with trona buffer for sustainable carbon recycle. Bioetchnol. Bioproc. Eng. 10: 422–429.
Siaut, M., S. Cuiné, C. Cagnon, B. Fessler, M. Nguyen, P. Carrier, A. Beyly, F. Beisson, C. Triantaphylidès, Y. Li-Beisson, and G. Peltier (2011) Oil accumulation in the model green alga Chlamydomonas reinhardtii: Characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol. 11: 7.
Kim, G., G. Mujtaba, M. Rizwan, and K. Lee (2014) Environmental stress strategies for stimulating lipid production from microlagae for biodiesel. Appl. Chem. Eng. 25: 553–558.
Mujtaba, G., W. Choi, C. G. Lee, and K. Lee (2012) Lipid production by Chlorella vulgaris after a shift from nutrient-rich to nitrogen starvation conditions. Bioresour. Technol. 123: 279–283.
Kim, G., C. H. Lee, and K. Lee (2016) Enhancement of lipid production in marine microalga Tetraselmis sp. through salinity variation. Kor. J. Chem. Eng. 33: 230–237.
Liu, Z. Y., G. C. Wang, and B. C. Zhou (2008) Effect of iron on growth and lipid accumulation in Chlorella vulgaris. Bioresour. Technol. 99: 4717–4722.
Kim, G., J. Bae, and K. Lee (2016) Nitrate repletion strategy for enhancing lipid production from marine microalga Tetraselmis sp. Bioresour. Technol. 205: 274–279.
Naito, K., M. Matsui, and I. Imai (2005) Ability of marine eukaryotic red tide microalgae to utilize insoluble iron. Harmful Algae 4: 1021–1032.
Chiu, S. Y., C. Y. Kao, M. T. Tsai, S. C. Ong, C, H, Chen, and C. S. Lin (2009) Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour. Technol. 100: 833–838.
Li, Y., Y. F. Chen, P. Chen, M. Min, W. Zhou, B. Martinez, J. Zhu, and R. Ruan (2011) Characterization of a microalga Chlorella sp. well adapted to highly concentrated municipal wastewater for nutrient removal and biodiesel production. Bioresour. Technol. 102: 5138–5144.
Oijen, T. V., M. Van Leeuwe, W. W. C. Gieskes, and H. J. W. De Baar (2004) Effects of iron limitation on photosynthesis and carbohydrate metabolism in the Antarctic diatom Chaetoceros brevis (Bacillariophyceae). Eur. J. Phycol. 39: 161–171.
Yeesang, C. and B. Cheirsilp (2011) Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresour. Technol. 102: 3034–3040.
Mata, T. M., R. Almeida, and N. S. Caetano (2013) Effect of the culture nutrients on the biomass and lipid productivities of microalgae Dunaliella tertiolecta. Chem. Eng. Trans. 32: 973–978.
Lee, O. K., A. L. Kim, D. H. Seong, C. G. Lee, Y. T. Jung, J. W. Lee, and E. Y. Lee (2013) Chemoenzymatic saccharification and bioethanol fermentation of lipid-extracted residual biomass of the microalga, Dunaliella tertiolecta. Bioresour. Technol. 132: 197–201.
Guillard, R. R. L (1975) Culture of phytoplankton for feeding marine invertebrates. pp. 26-60. In Smith, W. L. and M. H. Chanley (eds.) Culture of Marine Invertebrate Animals. Plenum Press, NY, USA.
Dubois, M., K. A. Gilles, J. K. Hamilton, P. Reberts, and F. Smith (1956) Colorimetric method for determination of sugars and related substances. Anal. Chem. 28: 350–356.
Van Wychen, S., K. Ramirez, and L. M. Laurens (2013) Determination of total lipids as fatty acid methyl esters (FAME) by in situ transesterification. Laboratory Analytical Procedure. p. 12. National Renewable Energy Laboratory, Golden, CO, USA.
Terauchi, A. M., G. Peers, M. C. Kobayashi, K. K. Niyogi, and S. S. Merchant (2010) Trophic status of Chlamydomonas reinhardtii influences the impact of iron deficiency on photosynthesis. Photosyn. Res. 105: 39–49.
Concas, A., A. Steriti, M. Pisu, and G. Cao (2014) Comprehensive modeling and investigation of the effect of iron on the growth rate and lipid accumulation of Chlorella vulgaris cultured in batch photobioreactors. Bioresour. Technol. 153: 340–350.
Ren, H. Y., B. F. Liu, F. Kong, L. Zhao, G. J. Xie, and N. Q. Ren (2014) Enhanced lipid accumulation of green microalga Scenedesmus sp. by metal ions and EDTA addition. Bioresour. Technol. 169: 763–767.
Sun, X., Y. Cao, H. Xu, Y. Liu, J. Sun, D. Qiao, and Y. Cao (2014) Effect of nitrogen-starvation, light intensity and iron on triacylglyceride/carbohydrate production and fatty acid profile of Neochloris oleoabundans HK-129 by a two-stage process. Bioresour. Technol. 155: 204–212.
Baky, H. H. A. E., G. S. El-Baroty, A. Bouaid, M. Martinez, and J. Aracil (2012) Enhancement of lipid accumulation in Scenedesmus obliquus by optimizing CO2 and Fe3+ levels for biodiesel production. Bioresour. Technol. 119: 429–432.
Ruangsomboon, S., M. Ganmanee, and S. Choochote (2013) Effects of different nitrogen, phosphorus, and iron concentrations and salinity on lipid production in newly isolated strain of the tropical green microalga, Scenedesmus dimorphus KMITL. J. Appl. Phycol. 25: 867–874.
Sakthivel, R., S. Elumalai, and M. M. Arif (2011) Microalgae lipid research, past, present: A critical review for biodiesel production, in the future. J. Exp. Sci. 2: 29–49.
Singh, B., A. Guldhe, I. Rawat, and F. Bux (2014) Towards a sustainable approach for development of biodiesel from plant and microalgae. Renew. Sustain. Energy Rev. 29: 216–245.
Kim, G., G. Mujtaba, and K. Lee (2016) Effects of nitrogen sources on cell growth and biochemical composition of marine chlorophyte Tetraselmis sp. for lipid production. Algae 31: 257–266.
Radakovits, R., R. E. Jinkerson, A. Darzins, and M. C. Posewitz (2010) Genetic engineering of algae for enhanced biofuel production. Eukaryotic Cell 9: 486–501.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rizwan, M., Mujtaba, G. & Lee, K. Effects of iron sources on the growth and lipid/carbohydrate production of marine microalga Dunaliella tertiolecta . Biotechnol Bioproc E 22, 68–75 (2017). https://doi.org/10.1007/s12257-016-0628-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-016-0628-0