Abstract
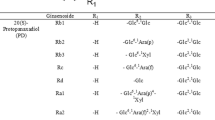
Lactic acid bacteria (LAB) were screened for ginsenoside transforming activity using crude ginseng extract. Thin-layer chromatography analysis of fermented ginseng extract showed that LAB strain MJM60396 possessed higher ginsenoside transformation ability than other strains. It converted major ginsenosides into minor ginsenosides such as Rg3 and Rh2. MJM60396 also showed high β-glucosidase activity. Strain MJM60396 was identified as Lactobacillus paracasei subsp. tolerans based on 16S rRNA gene sequence. To delineate the pathway involved in the production of the minor ginsenosides Rg3 and Rh2, strain MJM60396 was incubated with pure ginsenoside Rb1. HPLC analysis revealed the appearance of Rg3 and Rh2 peak from the incubation mixture containing Rb1 and strain MJM60396. Furthermore, β-glucosidase enzyme was prepared from strain MJM60396. To achieve its maximum activity, we optimized the pH and temperature conditions. Cell-free β-glucosidase enzyme hydrolyzed ginsenoside Rb1 through the following pathway: ginsenoside Rb1 → Rd → Rg3 → Rh2. This is the first report on the transformation of ginsenosides Rb1 to Rg3 and Rh2 by a Lac. paracasei subsp. tolerans strain. Our results indicate that Lac. paracasei subsp. tolerans MJM60396 has the potential to be used for preparing ginsenosides Rg3 and Rh2 as nutraceuticals.
Similar content being viewed by others
References
Qi, L. W., C. Z. Wang, and C. S. Yuan (2011) Ginsenosides from American ginseng: Chemical and pharmacological diversity. Phytochem. 72: 689–699.
Ye, L., C. Q. Zhou, W. Zhou, P. Zhou, D. F. Chen, X. H. Liu, X. L. Shi, and M. Q. Feng (2010) Biotransformation of ginsenoside Rb1 to ginsenoside Rd by highly substrate-tolerant Paecilomyces bainier 229-7. Bioresour. Technol. 101 7872–7876.
Kim, D. -H. (2012) Chemical Diversity of Panax ginseng, Panax quinquifolium, and Panax notoginseng. J. Ginseng Res. 36: 1–15.
Kim, S. -H., J. -W. Min, L. -H. Quan, S. -Y. Lee, D. -U. Yang, and D. -C. Yang (2012) Enzymatic transformation of ginsenoside Rb1 by Lactobacillus pentosus strain 6105 from kimchi. J. Ginseng Res. 36: 291–297.
Nag, S. A., J. J. Qin, W. Wang, M. H. Wang, H. Wang, and R. Zhang (2012) Ginsenosides as anticancer agents: In vitro and in vivo activities, structure-activity relationships, and molecular mechanisms of action. Front. Pharmacol. 3:25.
Zhang, Q., X. Kang, and W. Zhao (2006) Antiangiogenic effect of low-dose cyclophosphamide combined with ginsenoside Rg3 on Lewis lung carcinoma. Biochem. Biophys. Res. Commun. 342: 824–828.
Liu, T. G., Y. Huang, D. D. Cui, X. B. Huang, S. H. Mao, L. L. Ji, H. B. Song, and C. Yi (2009) Inhibitory effect of ginsenoside Rg3 combined with gemcitabine on angiogenesis and growth of lung cancer in mice. BMC Cancer 9:250.
Iishi, H., M. Tatsuta, M. Baba, H. Uehara, A. Nakaizumi, K. Shinkai, H. Akedo, H. Funai, S. Ishiguro, and I. Kitagawa (1997) Inhibition by ginsenoside Rg3 of bombesin-enhanced peritoneal metastasis of intestinal adenocarcinomas induced by azoxymeth-ane in Wistar rats. Clin. Exp. Metastasis. 15: 603–611.
Chen, J., H. Peng, X. Ou-Yang, and X. He (2008) Research on the antitumor effect of ginsenoside Rg3 in B16 melanoma cells. Melanoma Res. 18: 322–329.
Kim, S. M., S. Y. Lee, J. S. Cho, S. M. Son, S. S. Choi, Y. P. Yun, H. S. Yoo, Y. Yoon do, K. W. Oh, S. B. Han, and J. T. Hong (2010) Combination of ginsenoside Rg3 with docetaxel enhances the susceptibility of prostate cancer cells via inhibition of NFkappaB. Eur. J. Pharmacol. 631: 1–9.
Kim, S. W., H. Y. Kwon, D. W. Chi, J. H. Shim, J. D. Park, Y. H. Lee, S. Pyo, and D. K. Rhee (2003) Reversal of P-glycoproteinmediated multidrug resistance by ginsenoside Rg3. Biochem. Pharmacol. 65: 75–82.
Lee, C. K., K. K. Park, A. S. Chung, and W. Y. Chung (2012) Ginsenoside Rg3 enhances the chemosensitivity of tumors to cisplatin by reducing the basal level of nuclear factor erythroid 2-related factor 2-mediated heme oxygenase-1/NAD(P)H quinone oxidoreductase-1 and prevents normal tissue damage by scavenging cisplatin-induced intracellular reactive oxygen species. Food Chem. Toxicol. 50: 2565–2574.
Huang, J. Y., Y. Sun, and Q. -X. Fan (2009) Efficacy of Shenyi capsule combined with gemcitabine plus cisplatin in treatment of advanced esophageal cancer-a randomized controlled trial. J. Chin. Integr. Med. 7: 1047–1051.
Lu, P., W. Su, Z. Miao, H. Niu, J. Liu, and Q. Hua (2008) Effect and mechanism of ginsenoside Rg3 on postoperative life span of patients with non-small cell lung cancer. Chin. J. Integr. Med. 14: 33–36.
Chen, Z., J. Cheng, Y. Huang, S. Han, N. Liu, G. Zhu, and J. Yao (2007) Effect of adjuvant chemotherapy of ginsenoside Rg3 combined with mitomycin C and tegafur in advanced gastric cancer. Chin. J. Gastrointes. Surg. 10: 64–66.
Park, C. S., M. H. Yoo, K. H. Noh, and D. K. Oh (2010) Biotransformation of ginsenosides by hydrolyzing the sugar moieties of ginsenosides using microbial glycosidases. Appl. Microbiol. Biotechnol. 87: 9–19.
Wang, W., Y. Zhao, E. R. Rayburn, D. L. Hill, H. Wang, and R. Zhang (2007) In vitro anti-cancer activity and structure-activity relationships of natural products isolated from fruits of Panax ginseng. Cancer Chemother. Pharmacol. 59: 589–601.
Zhang, H., J. Gong, H. Zhang, and D. Kong (2015) Induction of apoptosis and reversal of permeability glycoprotein-mediated multidrug resistance of MCF-7-ADM by ginsenoside Rh2. Int. J. Clin. Exp. Pathol. 8: 4444–4456.
Li, S., Y. Gao, W. Ma, T. Cheng, and Y. Liu (2015) Ginsenoside Rh2 inhibits invasiveness of glioblastoma through modulation of VEGF-A. Tumour Biol. 1–6.
Her, Y., Y. -C. Lee, J. H. Oh, Y. -E. Choi, C. -W. Lee, J. -S. Kim, H. M. Kim, and J. -W. Yang (2012) An application of β-glycosidase to transformation of ginsenosides for the effective production of specific ginsenosides with biological efficacy. Biotechnol. Bioproc. Eng. 17: 538–546.
Chi, H., B. H. Lee, H. J. You, M. S. Park, and G. E. Ji (2006) Differential transformation of ginsenosides from Panax ginseng by lactic acid bacteria. J. Microbiol. Biotechnol. 16: 1629–1633.
Kim, M. K., J. W. Lee, K. Y. Lee, and D. -C. Yang (2005) Microbial conversion of major ginsenoside Rb1 to pharmaceutically active minor ginsenoside Rd. J. Microbiol. 43: 456–462.
Saqib, A. A. N. and P. J. Whitney (2006) Esculin gel diffusion assay (EGDA): A simple and sensitive method for screening β-glucosidases. Enz. Microb. Technol. 39: 182–184.
Tamura, K., G. Stecher, D. peterson, A. Filipski, and S. Kumar (2013) MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30: 2725–2729.
Palaniyandi, S. A., K. Damodharan, K. W. Lee, S. H. Yang, and J. -W. Suh (2015) Enrichment of ginsenoside Rd in Panax ginseng extract with combination of enzyme treatment and high hydrostatic pressure. Biotechnol. Bioproc. Eng. 20: 608–613.
Liu, L., X. M. Zhu, Q. J. Wang, D. L. Zhang, Z. M. Fang, C. Y. Wang, Z. Wang, B. S. Sun, H. Wu, and C. K. Sung (2010) Enzymatic preparation of 20(S, R)-protopanaxadiol by transformation of 20(S, R)-Rg3 from black ginseng. Phytochem. 71: 1514–1520.
Desai, A. S., V. M. Chauhan, A. P. Johnston, T. Esler, and J. W. Aylott (2013) Fluorescent nanosensors for intracellular measurements: Synthesis, characterization, calibration, and measurement. Front. Physiol. 4:401.
Lee, B. H., H. J. You, M. S. Park, B. Kwon, and G. E. Ji (2006) Transformation of the Glycosides from food materials by probiotics and food microorganisms. J. Microbiol. Biotechnol. 16: 497–504.
Lee, Y., J. Oh, H. Lee, N. K. Lee, D. -Y. Jeong, and Y. -S. Jeong (2015) Lactic acid bacteria-mediated fermentation of Cudrania tricuspidata leaf extract improves its antioxidative activity, osteogenic effects, and anti-adipogenic effects. Biotechnol. Bioproc. Eng. 20: 861–870.
Kim, S. H., S. Y. Kim, H. Lee, K. S. Ra, H. J. Suh, S. Y. Kim, and K. -S. Shin (2011) Transformation of ginsenoside-rich fraction isolated from ginseng (Panax ginseng) leaves induces compound K. Food Sci. Biotechnol. 20: 1179–1186.
Quan, K., Q. Liu, J. Y. Wan, Y. J. Zhao, R. Z. Guo, R. N. Alolga, P. Li, and L. W. Qi (2015) Rapid preparation of rare ginsenosides by acid transformation and their structure-activity relationships against cancer cells. Sci. Rep. 5: 8598.
Su, J. -H., J. -H. Xu, H. -L. Yu, Y. -C. He, W. -Y. Lu, and G. -Q. Lin (2009) Properties of a novel β-glucosidase from Fusarium proliferatum ECU2042 that converts ginsenoside Rg3 into Rh2. J. Mol. Catal. B: Enz. 57: 278–283.
Zhao, X., L. Gao, J. Wang, H. Bi, J. Gao, X. Du, Y. Zhou, and G. Tai (2009) A novel ginsenoside Rb1-hydrolyzing β-D-glucosidase from Cladosporium fulvum. Proc. Biochem. 44: 612–618.
Gao, J., X. Zhao, H. Liu, Y. Fan, H. Cheng, F. Liang, X. Chen, N. Wang, Y. Zhou, and G. Tai (2010) A highly selective ginsenoside Rb1-hydrolyzing β-D-glucosidase from Cladosporium fulvum. Proc. Biochem. 45: 897–903.
Ruan, C. C., H. Zhang, L. X. Zhang, Z. Liu, G. Z. Sun, J. Lei, Y. X. Qin, Y. N. Zheng, X. Li, and H. Y. Pan (2009) Biotransformation of ginsenoside Rf to Rh1 by recombinant beta-glucosidase. Molecules 14: 2043–2048.
Quan, L. H., J. W. Min, S. Sathiyamoorthy, D. U. Yang, Y. J. Kim, and D. C. Yang (2012) Biotransformation of ginsenosides Re and Rg1 into ginsenosides Rg2 and Rh1 by recombinant betaglucosidase. Biotechnol. Lett. 34: 913–917.
Chi, H. and G. E. Ji (2005) Transformation of ginsenosides Rb1 and Re from Panax ginseng by food microorganisms. Biotechnol. Lett. 27: 765–771.
Jung, H. M., Q. M. Liu, J. K. Kim, S. T. Lee, S. C. Kim, and W. T. Im (2012) Lactobacillus ginsenosidimutans sp. nov., isolated from kimchi with the ability to transform ginsenosides. Antonie Van Leeuwenhoek. 103: 867–876.
Kim, B. -G., S. -Y. Choi, M. -R. Kim, H. J. Suh, and H. J. Park (2010) Changes of ginsenosides in Korean red ginseng (Panax ginseng) fermented by Lactobacillus plantarum M1. Proc. Biochem. 45: 1319–1324.
Quan, L. -H., L. -Q. Cheng, H. -B. Kim, J. -H. Kim, N. -R. Son, S. -Y. Kim, H. -O. Jin, and D. -C. Yang (2010) Bioconversion of ginsenoside Rd into Compound K by Lactobacillus pentosus DC101 isolated from Kimchi. J. Ginseng Res. 34: 288–295.
Quan, L. H., Y. J. Kim, G. H. Li, K. T. Choi, and D. C. Yang (2013) Microbial transformation of ginsenoside Rb1 to compound K by Lactobacillus paralimentarius. World J. Microbiol. Biotechnol. 29: 1001–1007.
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Palaniyandi, S.A., Son, B.M., Damodharan, K. et al. Fermentative transformation of ginsenoside Rb1 from Panax ginseng C. A. Meyer to Rg3 and Rh2 by Lactobacillus paracasei subsp. tolerans MJM60396. Biotechnol Bioproc E 21, 587–594 (2016). https://doi.org/10.1007/s12257-016-0281-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-016-0281-7