Abstract
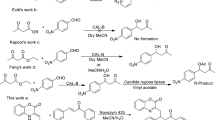
Previous studies on the lipase-mediated acylation of 6-azauridine with vinyl stearate in organic solvents revealed that while preparing a potential prodrug, 3′-O-stearoyl-6-azauridine, a lipase from Burkholderia cepacia showed high regioselectivity toward the second hydroxyl group. The most suitable reaction solvent, molar ratio of vinyl stearate to 6-azauridine, and reaction temperature were anhydrous acetone, 15:1, and 45°C, respectively. Under these conditions, the initial reaction rate, 3′-regioselectivity, and maximum substrate conversion were as high as 10.4 mM/h, 86.0, and 99.0%, respectively.
Similar content being viewed by others
References
Pejanović, V., V. Piperski, D. Ugljeşœić-Kilibarda, J. Tasić, M. Dačević, L. Medić-Mijačević, E. Gunić, M. Popsavin, and V. Popsavin (2006) Synthesis and biological activity of some new 5′-O-acyl tiazofurin derivatives. Eur. J. Med. Chem. 41: 503–512.
Ferrero, M. and V. Gotor (2000) Biocatalytic selective modifications of conventional nucleosides, carbocyclic nucleosides, and C-nucleosides. Chem. Rev. 100: 4319–4348.
Li, X. F., M. H. Zong, and G. L. Zhao (2010) Highly regioselective enzymatic synthesis of 5′-O-stearate of 1-β-D-arabinofuranosylcytosine in binary organic solvent mixtures. Appl. Microbiol. Biotechnol. 88: 1–7.
Wang, H., M. H. Zong, H. Wu, and W. Y. Lou (2007) Novel and highly regioselective route for synthesis of 5-fluorouridine lipophilic ester derivatives by lipozyme TL IM. J. Biotechnol. 129: 689–695.
Li, N., M. H. Zong, and D. Ma (2008) Regioselective acylation of nucleosides catalyzed by Candida Antarctica lipase B: Enzyme substrate recognition. Eur. J. Org. Chem. 2008: 5375–5378.
Li, N., M. H. Zong, and D. Ma (2009) Regioselective acylation of nucleosides and their analogs catalyzed by Pseudomonas cepacia lipase: Enzyme substrate recognition. Tetrahed. 65: 1063–1068.
Clercq, E. De (2011) A 40-year journey in the antiviral drug field. Ann. R. Pharm. 51: 1–24.
Lum, P. Y., L. Y. Ngo, A. H. Bakken, and J. D. Unadkat (2000) Human intestinal es nucleoside transporter: Molecular characterization and nucleoside inhibitory profiles. Canc. Chemot. 45: 273–278.
Zeng, H., Z. P. Lin, and A. C. Sartorelli (2004) Resistance to purine and pyrimidine nucleoside and nucleobase analogs by the human MDR1 transfected murine leukemia cell line L1210/ VMDRC. 06. Biochem. Pharmacol. 68: 911–921.
Bello, A. M., D. Konforte, E. Poduch, C. Furlonger, L. Wei, Y. Liu, M. Lewis, E. F. Pai, C. J. Paige, and L. P. Kotra (2009) Structure-activity relationships of orotidine-5′-monophosphate decarboxylase inhibitors as anticancer agents. J. Med. Chem. 52: 1648–1658.
Plevová, J., F. H. Mohamed, and I. Jank (1971) Elimination of 2′,3′,5′-tri-O-acetyl-6-azauridine in the rat and in man. Biochem. Pharmacol. 20: 2079–2083.
Zinni, M. A., L. E. Iglesias, and A. M. Iribarren (2007) Preparation of potential 3-deazauridine and 6-azauridine prodrugs through an enzymatic alcoholysis. J. Mol. Catal. B-Enz. 47: 86–90.
Díaz-Rodríguez, A., S. Fernández, Y. S. Sanghvi, M. Ferrero, and V. Gotor (2006) Novel chemoenzymatic protocol for the synthesis of 3-O-dimethoxytrityl-2-deoxynucleoside derivatives as building blocks for oligonucleotide synthesis. Org. Process Res. Dev. 10: 581–587.
Wang, Z. Y., N. Li, and M. H. Zong (2009) A simple procedure for the synthesis of potential 6-azauridine prodrugs by Thermomyces lanuginosus lipase. J. Mol. Catal. B-Enz. 59: 212–219.
Wang, Z. Y. and M. H. Zong (2009) Recognition of acyl donors by lipase CAL-B in the acylation of 6-azauridine. Biotechnol. Prog. 25: 784–791.
Hamamura, E. K., M. Prystasz, J. P. H. Verheyden, J. G. Moffatt, K. Yamaguchi, N. Uchida, K. Sato, A. Nomura, and O. Shiratori (1976) Reactions of 2-acyloxyisobutyryl halides with nucleosides. 6. Synthesis and biological evaluation of some 3′-acyl derivatives of 2,2′-anhydro-1-(β-D-arabinofuranosyl) cytosine hydrochloride. J. Med. Chem. 19: 654–662.
Halling, P. J. (1994) Thermodynamic predictions for biocatalysis in nonconventional media: theory, tests, and recommendations for experimental design and analysis. Enz. Microb. Technol. 16: 178–206.
Bell, G., P. J. Halling, B. D. Moore, J. Partridge, and D. G. Rees (1995) Biocatalyst behaviour in low-water systems. Trends Biotechnol. 13: 468–473.
Kumar, V., V. S. Parmar, and S. V. Malhotra (2010) Structural modifications of nucleosides in ionic liquids. Biochimie. 92: 1260–1265.
Almarsson, O. and A. M. Klibanov (1996) Remarkable activation of enzymes in nonaqueous media by denaturing organic cosolvents. Biotechnol. Bioeng. 49: 87–92.
Tsikaris, V., M. Sakarellos-Daitsiotis, N. Theophanidis, C. Sakarellos, M. T. Cung, and M. Marraud (1991) Significance of bound residual water in the DMSO solution structure of stable peptide hydrates. J. Chem. Soc., Perkin Trans. 2. 9: 1353–1357.
Anthonsen, T. and B. H. Hoff (1998) Resolution of derivatives of 1, 2-propanediol with lipase B from Candida antarctica: Effect of substrate structure, medium, water activity and acyl donor on enantiomeric ratio. Chem. Phys. Lipids 93: 199–207.
Chamouleau, F., D. Coulon, M. Girardin, and M. Ghoul (2001) Influence of water activity and water content on sugar esters lipase-catalyzed synthesis in organic media. J. Mol. Catal. B-Enz. 11: 949–954.
Klibanov, A. M. (2001) Improving enzymes by using them in organic solvents. Nature 409: 241–246.
Soares, C. M., V. H. Teixeira, and A. M. Baptista (2003) Protein structure and dynamics in nonaqueous solvents: INSIGHTS from molecular dynamics simulation studies. Biophys. J. 84: 1628–1641.
Weber, H. K., H. Weber, and R. J. Kazlauskas (1999) Watching lipase-catalyzed acylations using 1H NMR: Competing hydrolysis of vinyl acetate in dry organic solvents. Tetrahed. Asymmetr. 10: 2635–2638.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, ZY., Bi, YH. & Zong, MH. Regioselective enzymatic procedure for preparing 3′-O-stearoyl-6-azauridine by using Burkholderia cepacia lipase. Biotechnol Bioproc E 17, 393–397 (2012). https://doi.org/10.1007/s12257-011-0483-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-011-0483-y