Abstract
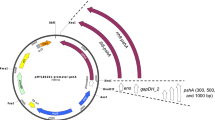
Overexpression in Escherichia coli and functional characterization of the group II chaperonin from the hyperthermophilic archaeum Pyrococcus horikoshii OT3 were investigated in this study. PhCpn, the chaperonin gene from the P. horikoshii OT3, was amplified by PCR from the P. horikoshii genomic DNA, subcloned into pET21a vector, and expressed in three E. coli host cells such as BL21, Rosetta, and Codonplus (DE3). Among these host cells, E. coli Rosetta showed the highest expression level of recombinant PhCpn at induction with 1 mM IPTG. The recombinant PhCpn was purified to 91% by heat-shock treatment and anion-exchange chromatography. The ATPase activity of the purified PhCpn increased in a PhCpn concentration-dependent manner. Also, PhCpn protected the inorganic phosphatase from thermal inactivation at 85 and 110°C, speculating that PhCpn is effective in in vitro holding of the protein. The holding efficiency was enhanced by the addition of Mg2+ ion. Through the coexpression of pro-carboxypeptidase B (pro-CPB) and PhCpn in E. coli Rosetta, pro-CPB was produced as a soluble and active form with a marked yield. This result indicated that PhCpn facilitated the in vivo correct folding of pro-CPB and could be used as powerful and novel molecular machinery for the production of recombinant proteins as soluble and active forms in E. coli.
Similar content being viewed by others
References
Kawarabayasi, Y., M. Sawada, H. Horikawa, Y. Haikawa, Y. Hino, S. Yamamoto, M. Sekine, S. Baba, H. Kosugi, A. Hosoyama, Y. Nagai, M. Sakai, K. Ogura, R. Otsuka, H. Nakazawa, M. Takamiya, Y. Ohfuku, T. Funahashi, T. Tanaka, Y. Kudoh, J. Yamazaki, N. Kushida, A. Oguchi, K. Aoki, and H. Kikuchi (1998) Complete sequence and gene organization of the genome of a hyper-thermophilic archaebacterium, Pyrococcus horikoshii OT3 (supplement). DNA Res. 5: 147–155.
Bukau, B., E. Deuerling, C. Pfund, and E. A. Craig (2000) Getting newly synthesized proteins into shape. Cell 101: 119–122.
Ellis, R. J. and F. U. Hartl (1996) Protein folding in the cell: competing models of chaperonin function. FASEB J. 10: 20–26.
Ellis, R. J. (1996) The chaperonins. pp. 2–25. Academic Press, San Diego, USA
Gething, M. J. and J. Sambrook (1992) Protein folding in the cell. Nature 355: 33–45.
Laksanalamai, P., T. A. Whitehead, and F. T. Robb (2004) Minimal protein-folding systems in hyperthermophilic archaea. Nat. Rev. Microbiol. 2: 315–324.
Mogk, A., B. Bukau, and E. Deuerling (2001) Cellular functions of cytosolic E. coli chaperones. pp. 1–34. In: P. Lund (ed.). Molecular chaperones in the cell. Oxford University Press, Oxford, UK.
Hartl, F. U. and M. Hayer-Hartl (2002) Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295: 1852–1858.
Valpuesta, J. M., J. Martin-Benito, P. Gomez-Puertas, J. L. Carrascosa, and K. R. Willison (2002) Structure and function of a protein folding machine: the eukaryotic cytosolic chaperonin CCT. FEBS Lett. 529: 11–16.
Ranson, N. A., H. E. White, and H. R. Saibil (1998) Chaperonins. Biochem. J. 333: 233–242.
Kim, S., K. R. Willison, and A. L. Horwich (1994) Cystosolic chaperonin subunits have a conserved ATPase domain but diverged polypeptide-binding domains. Trends Biochem. Sci. 19: 543–548.
Kubota, H., G. Hynes, and K. Willison (1995) The chaperonin containing t-complex polypeptide 1 (TCP-1). Multisubunit machinery assisting in protein folding and assembly in the eukaryotic cytosol. Eur. J. Biochem. 230: 3–16.
Bukau, B. and A. L. Horwich (1998) The Hsp70 and Hsp60 chaperone machines. Cell 92: 351–366.
Yoshida, T., R. Kawaguchi, H. Taguchi, M. Yoshida, T. Yasunaga, T. Wakabayashi, M. Yohda, and T. Maruyama (2002) Archaeal group II chaperonin mediates protein folding in the cis-cavity without a detachable GroES-like co-chaperonin. J. Mol. Biol. 315: 73–85.
Phipps, B. M., D. Typke, R. Heger, S. Volker, A. Hoffmann, K. O. Stetter, and W. Baumeister (1993) Structure of a molecular chaperone from a thermophilic archaebacterium. Nature 361: 475–477.
Hartl, F. U. (1996) Molecular chaperones in cellular protein folding. Nature 381: 571–579.
Fenton, W. A. and A. L. Horwich (1997) GroEL-mediated protein folding. Protein Sci. 6: 743–760.
Sigler, P. B., Z. Xu, H. S. Rye, S. G. Burston, W. A. Fenton, and A. L. Horwich (1998) Structure and function in GroEL-mediated protein folding. Annu. Rev. Biochem. 67: 581–608.
Archibald, J. M., J. M. Logsdon, and W. F. Doolittle (1999) Recurrent paralogy in the evolution of archaeal chaperonins. Curr. Biol. 9: 1053–1056.
Gutsche, I., L. O. Essen, and W. Baumeister (1999) Group II chaperonins: new TRiC(k)s and turns of a protein folding machine. J. Mol. Biol. 293: 295–312.
Yan, Z., S. Fujiwara, K. Kohda, M. Takagi, and T. Imanaka (1997) In vitro stabilization and in vivo solubilization of foreign proteins by the subunit of a chaperonin from the hyperthermophilic archaeon Pyrococcus sp. strain KOD1. Appl. Environ. Microbiol. 63: 785–789.
Kohda, J., Y. Endo, N. Okumura, Y. Kurokawa, K. Nishihara, H. Yanagi, T. Yura, H. Fukuda, and A. Kondo (2002) Improvement of productivity of active form of glutamate racemase in Escherichia coli by coexpression of folding accessory proteins. Biochem. Eng. J. 10: 39–45.
Kwon, M. J., S. L. Park, S. K. Kim, and S. W. Nam (2002) Overproduction of Bacillus macerans cyclodextrin glucanotransferase in E. Coli by coexpression of GroEL/ES chaperone. J. Microbiol. Biotechnol. 12: 1002–1005.
Guagliardi, A., L. Cerchia, S. Bartolucci, and M. Rossi (1994) The chaperonin from the archaeon Sulfolobus solfataricus promotes correct refolding and prevents thermal denaturation in vitro. Protein Sci. 3: 1436–1443.
Guagliardi, A., L. Cerchia, and M. Rossi (1995) Prevention of in vitro protein thermal aggregation by the Sulfolobus solfataricus chaperonin. Evidence for nonequivalent binding surfaces on the chaperonin molecule. J. Biol. Chem. 270: 28126–28132.
Furutani, M., T. Iida, T. Yoshida, and T. Maruyama (1998) Group II chaperonin in a thermophilic methanogen, Methanococcus thermolithotrophicus. Chaperone activity and filament-forming ability. J. Biol. Chem. 273: 28399–28407.
Kusmierczyk, A. R. and J. Martin (2003) Nucleotidedependent protein folding in the type II chaperonin from the mesophilic archaeon Methanococcus maripaludis. Biochem. J. 371: 669–673.
Yoshida, T., M. Yohda, T. Iida, T. Maruyama, H. Taguchi, K. Yazaki, T. Ohta, M. Odaka, I. Endo, and Y. Kagawa (1997) Structural and functional characterization of homo-oligomeric complexes of alpha and beta chaperonin subunits from the hyperthermophilic archaeum Thermococcus strain KS-1. J. Mol. Biol. 273: 635–645.
Yoshida, T., A. Ideno, S. Hiyamuta, M. Yohda, and T. Maruyama (2001) Natural chaperonin of the hyperthermophilic archaeum, Thermococcus strain KS-1: a heterooligomeric chaperonin with variable subunit composition. Mol. Microbiol. 39: 1406–1413.
Yoshida, T., A. Ideno, R. Suzuki, M. Yohda, and T. Maruyama (2002) Two kinds of archaeal group II chaperonin subunits with different thermostability in Thermococcus strain KS-1. Mol. Microbiol. 44: 761–769.
Izumi, M., S. Fujiwara, M. Takagi, S. Kanaya, and T. Imanaka (1999) Isolation and characterization of a second subunit of molecular chaperonin from Pyrococcus kodakaraensis KOD1: analysis of an ATPase-deficient mutant enzyme. Appl. Environ. Microbiol. 65: 1801–1805.
Okochi, M., H. Matsuzaki, T. Nomura, N. Ishii N, and M. Yohda (2005) Molecular characterization of the group II chaperonin from the hyperthermophilic archaeum Pyrococcus horikoshii OT3. Extremophiles 9: 127–134.
Chen, H. Y., Z. M. Chu, Y. H. Ma, Y. Zhang, and S. L. Yang (2007) Expression and characterization of the chaperonin molecular machine from the hyperthermophilic archaeon Pyrococcus furiosus. J. Basic Microbiol. 47: 132–137.
Geladopoulos, T. P., T. G. Sotiroudis, and A. E. Evangelopoulos (1991) A malachite green colorimetric assay for protein phosphatase activity. Anal. Biochem. 192: 112–116.
Kim, M. J., S. H. Kim, J. H. Lee, J. H. Seo, J. H. Lee, J. H. Kim, Y. H. Kim, and S. W. Nam (2008) High-level secretory expression of human procarboxypeptidase B by fedbatch cultivation of Pichia pastoris and its partial characterization. J. Microbiol. Biotechnol. 18: 1938–1944.
Jeon, S. J. and K. Ishikawa (2005) Characterization of the family I inorganic pyrophosphatase from Pyrococcus horikoshii OT3. Archaea 1: 385–389.
Hongo, K., H. Hirai, C. Uemura, S. Ono, J. Tsunemi, T. Higurashi, T. Mizobata, and Y. Kawata (2006) A novel ATP/ADP hydrolysis activity of hyperthermostable group II chaperonin in the presence of cobalt or manganese ion. FEBS Lett. 580: 34–40.
Park, S. L., E. J. Shin, S. P. Hong, S. J. Jeon, and S. W. Nam (2005) Production of soluble human granulocyte colony stimulating factor in E. coli by molecular chaperones. J. Microbiol. Biotechnol. 15: 1267–1272.
Shin, E. J., S. L. Park, S. J. Jeon, J. W. Lee, Y. T. Kim, Y. H. Kim, and S. W. Nam (2006) Effect of molecular chaperones on the soluble expression of alginate lyase in E. coli. Biotechnol. Bioprocess Eng. 11: 414–419.
Oh, I. S., T. W. Kim, J. H. Ahn, J. W. Keum, C. Y. Choi, and D. M. Kim (2007) Use of L-buthionine sulfoximine for the efficient expression of disulfide-containing proteins in cell-free extracts of Escherichia coli. Biotechnol. Bioprocess Eng. 5: 574–578.
Kondo, A., J. Kohda, Y. Endo, T. Shiromizu, Y. Kurokawa, K. Nishihara, H. Yanagi, T. Yura, and H. Fukuda (2000) Improvement of productivity of active horseradish peroxidase in Escherichia coli by coexpression of Dsb proteins. J. Biosci. Bioeng. 90: 600–606.
Thomas, J. G., A. Ayling, and F. Baneyx (1997) Molecular chaperones, folding catalysts, and the recovery of active recombinant proteins from Escherichia coil. Appl. Biochem. Biotechnol. 66: 197–238.
Kang, C. S., C. W. Park, and I. S. Bang (2008) Production and purification of a cecropin family antibacterial peptide, hinnavin II, in Escherichia coli. Biotechnol. Bioprocess Eng. 13: 377–382.
Okochi, M., K. Kanie, M. Kurimoto, M. Yohda, and H. Honda (2008) Overexpression of prefoldin from the hyperthermophilic archaeum Pyrococcus horikoshii OT3 endowed Escherichia coli with organic solvent tolerance. Appl. Biochem. Biotechnol. 79: 443–449.
Jung, S. M., A. K. Lim, and K. M. Park (2008) Construction of Candida antarctica lipase B expression system in E. coli coexpressing chaperones. Korean J. Biotechnol. Bioeng. 23: 403–407.
Gribaldo, S., V. Lumia, R. Creti, E. C. de Macario, A. Sanangelantoni, and P. Cammarano (1999) Discontinuous occurrence of the hsp70 (DnaK) gene among archaea and sequence features of HSP70 suggest a novel outlook on phylogenies inferred from this protein. J. Bacteriol. 181: 434–443.
Laksanalamai, P., T. A. Whitehead, and F. T. Robb (2004) Minimal protein-folding systems in hyperthermophilic archaea. Nat. Rev. Microbiol. 2: 315–324.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, JH., Shin, EJ., Jeon, SJ. et al. Overexpression, purification, and functional characterization of the group II chaperonin from the hyperthermophilic archaeum Pyrococcus horikoshii OT3. Biotechnol Bioproc E 14, 551–558 (2009). https://doi.org/10.1007/s12257-009-0008-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-009-0008-0