Abstract
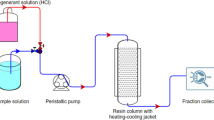
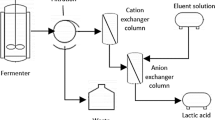
Ion exchange chromatography (IEC) and desalting electrodialysis (DSED) processes were developed for the recovery and purification of potassium clavulanate (KCA) from fermentation broth. A strong anion exchanger, Amberlite IRA 400 resin, a potassium acetate solution as equilibrium buffer, and a potassium chloride (KCl) solution as elution buffer were used for the recovery of KCA in IEC. In order to determine optimal operating conditions, the effects of various operating parameters such as equilibrium buffer pH and concentration, elution buffer concentration, gradient length, and volumetric flow rate on KCA recovery and by-product removal were investigated using a simulated fermentation broth. In the subsequent step of DSED, employing cation (Neocepta CMS, Tokuyama, Japan) and anion (Neocepta ACS, Tokuyama, Japan) exchange membranes were carried out to remove KCl that existed in a large amount in the ion exchanged solution. The effects of operation voltage and feed composition on the performance of DSED were investigated. Based on the operating conditions determined above, IEC and DSED were applied in sequence to an ultrafiltered fermentation broth. Almost complete removal of KCl was possible with no significant loss of KCA, although the KCA recovery was slightly lower than that with the simulated fermentation broth. Based on this observation, it was concluded that IEC and DESD could be an effective process combination for the recovery of KCA from fermentation broth.
Similar content being viewed by others
References
Brown, A. G., D. Butterworth, M. Cole, G. Hanscomb, J. D. Hood, C. Reading, and G. N. Rolinson (1976) Naturally occurring β-lactamase inhibitors with antibacterial activity. J. Antibiot. 29: 668–669.
Mayer, A. F. and W. D. Deckwer (1996) Simultaneous production and decomposition of clavulanic acid during Streptomyces clavuligerus cultivation. Appl. Microbiol. Biotechnol. 45: 41–46.
Butterworth, D. (1984) Clavulanic acid: Properties, Biosynthesis, and Fermentation. pp. 225–235. In: E. J. Vandamme (ed.). Biotechnology of Industrial Antibiotics. Marcel Dekker, NY, USA.
Mayer, A. F., F. B. Anspach, and W. D. Deckwer (1996) Purification of clavulanic acid by ion-pairing systems. Bioseparation 6: 25–39.
Mayer, A. F. and W. D. Deckwer (1996) Ion pair adsorption chromatography for process purposes: basic equilibrium studies for the recovery of clavulanic acid by using quaternary ammonium salts. J. Chromatogr. A 741: 185–203.
Barboza, M., R. M. R. G. Almeida, and C. O. Hokka (2002) Intrinsic kinetic parameters of clavulanic acid adsorption by ion-exchange chromatography. Ind. Eng. Chem. Res. 41: 5789–5793.
Barboza, M., R. M. R. G. Almeida, and C.O. Hokka (2003) Influence of temperature on the kinetics of adsorption and desorption of clavulanic acid by ion exchange. Biochem. Eng. J. 14: 19–26.
Yamamoto, S., K. Nakanishi, and R. Matsuno (1988) Ion-Exchange Chromatography of Proteins. Marcel Dekker, NY, USA.
Haginaka, J., T. Nakagawa, and T. Uno (1981) Stability of clavulanic acid in aqueous solution. Chem. Pharm. Bull. 29: 3334–3341.
Bersanetti, P. A., R. M. R. G. Almeida, M. Barboza, M. L. G. C. Araújo, and C. O. Hokka (2005) Kinetic studies on clavulanic acid degradation. Biochem. Eng. J. 23: 31–36.
Vahdat, L. (2000) Factors influencing the rates of degradation of amoxicillin sodium and potassium clavulanate in the liquid and frozen states. Ph.D. Thesis. Curtin University of Technology, Australia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H.H., Kang, S.H. & Chang, Y.K. Recovery of potassium clavulanate from fermentation broth by ion exchange chromatography and desalting electrodialysis. Biotechnol Bioproc E 14, 803–810 (2009). https://doi.org/10.1007/s12257-008-0161-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0161-x