Abstract
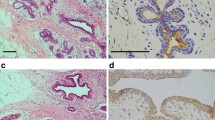
Focal adhesion kinase (FAK) is involved in progression of various cancers, and FAK overexpression has been associated with cancer invasion and metastasis. However, the involvement of FAK expression in the clinicopathological malignancy of oral squamous cell carcinoma (OSCC) remains unknown. In addition, there is no consensus regarding the role of p16 expression in OSCC. In this study, the immunohistochemically measured expression of FAK, phosphorylated FAK (FAKpY397) and p16 expressions and their associations with clinicopathological features and 5-year survival rates were examined in surgical samples from 70 patients with primary OSCC. FAK and FAKpY397 were expressed at high levels in 42 cases (60.0%) and 34 cases (48.6%), respectively, and 9 cases (12.9%) were positive for p16. FAK expression was significantly correlated with local recurrence, subsequent metastasis, and the mode of invasion. FAKpY397 expression was significantly correlated with both N classification and the mode of invasion. p16 expression was significantly correlated with clinical stage only. Patients having high expression of FAK, FAKpY397, or both showed significantly worse prognosis, but p16 expression showed no significant relation to prognosis. The results suggested that overexpression and phosphorylation of FAK in OSCC may affect cancer progression, such as local invasion and lymph node metastasis, and thereby contribute to life prognosis.



Similar content being viewed by others
References
Simard EP, Torre LA, Jemal A (2014) International trends in head and neck cancer incidence rates: differences by country, sex and anatomic site. Oral Oncol 50:387–403
Schaller MD (2010) Cellular functions of FAK kinases: insight into molecular mechanisms and novel functions. J Cell Sci 123:1007–1013
McLean GW, Carragher NO, Avizienyte E, Evans J, Brunton VG, Frame MC (2005) The role of focal-adhesion kinase in cancer - a new therapeutic opportunity. Nat Rev Cancer 5:505–515
Lee BY, Timpson P, Horvath LG, Daly RJ (2015) FAK signaling in human cancer as a target for therapeutics. Pharmacol Ther 146:132–149
Owens LV, Xu L, Dent GA, Yang X, Sturge GC, Craven RJ, Cance WG (1996) Focal adhesion kinase as a marker of invasive potential in differentiated human thyroid cancer. Ann Surg Oncol 3:100–105
Tremblay L, Hauck W, Aprikian AG, Begin LR, Chapdelaine A, Chevalier S (1996) Focal adhesion kinase (pp125FAK) expression, activation and association with paxillin and p50CSK in human metastatic prostate carcinoma. Int J Cancer 68:164–171
McCormack SJ, Brazinski SE, Moore JL Jr, Werness BA, Goldstein DJ (1997) Activation of the focal adhesion kinase signal transduction pathway in cervical carcinoma cell lines and human genital epithelial cells immortalized with human papillomavirus type 18. Oncogene 15:265–274
Judson PL, He X, Cance WG, Van Le L (1999) Overexpression of focal adhesion kinase, a protein tyrosine kinase, in ovarian carcinoma. Cancer 86:1551–1556
Lark AL, Livasy CA, Calvo B, Caskey L, Moore DT, Yang X, Cance WG (2003) Overexpression of focal adhesion kinase in primary colorectal carcinomas and colorectal liver metastases: immunohistochemistry and real-time PCR analyses. Clin Cancer Res 9:215–222
Owens LV, Xu L, Craven RJ, Dent GA, Weiner TM, Kornberg L, Liu ET, Cance WG (1995) Overexpression of the focal adhesion kinase (p125FAK) in invasive human tumors. Cancer Res 55:2752–2755
Recher C, Ysebaert L, Beyne-Rauzy O, Mansat-De Mas V, Ruidavets JB, Cariven P, Demur C, Payrastre B, Laurent G, Racaud-Sultan C (2004) Expression of focal adhesion kinase in acute myeloid leukemia is associated with enhanced blast migration, increased cellularity, and poor prognosis. Cancer Res 64:3191–3197
Schlaepfer DD, Mitra SK, Ilic D (2004) Control of motile and invasive cell phenotypes by focal adhesion kinase. Biochim Biophys Acta 1692:77–102
Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tân PF, Westra WH, Chung CH, Jordan RC, Lu C, Kim H, Axelrod R, Silverman CC, Redmond KP, Gillison ML (2010) Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 363:24–35
Allen CT, Lewis JS Jr, El-Mofty SK, Haughey BH, Nussenbaum B (2010) Human papillomavirus and oropharynx cancer: biology, detection and clinical implications. Laryngoscope 120:1756–1772
Marur S, D'Souza G, Westra WH, Forastiere AA (2010) HPV-associated head and neck cancer: a virus-related cancer epidemic. Lancet Oncol 11:781–789
Wang MB, Liu IY, Gornbein JA, Nguyen CT (2015) HPV-positive oropharyngeal carcinoma: a systematic review of treatment and prognosis. Otolaryngol Head Neck Surg 153:758–769
In (2009) International Union against Cancer, LH Sobin, MK Gospodrowicz, CH Wittekind (eds.). TNM Classification of Malignant Tumours. 7th Ed. Wiley-Liss, New York, NY
Yamamoto E, Kohama G, Sunakawa H, Iwai M, Hiratsuka H (1983) Mode of invasion, bleomycin sensitivity, and clinical course in squamous cell carcinoma of the oral cavity. Cancer 51:2175–2180
Theocharis S, Klijanienko J, Giaginis C, Alexandrou P, Patsouris E, Sastre-Garau X (2012) FAK and Src expression in mobile tongue squamous cell carcinoma: associations with clinicopathological parameters and patients survival. J Cancer Res Clin Oncol 138:1369–1377
Heiduschka G, Grah A, Oberndorfer F, Kadletz L, Altorjai G, Kornek G, Wrba F, Thurnher D, Selzer E (2015) Improved survival in HPV/p16-positive oropharyngeal cancer patients treated with postoperative radiotherapy. Strahlenther Onkol 191:209–216
Yamamoto E (1994) Role of chemotherapy for oral cancer from the viewpoint of mode of invasion. Asian J Oral Maxillofac Surg 6:113–121
Kurokawa H, Yamashita Y, Takeda S, Zhang M, Fukuyama H, Takahashi T (2002) Risk factors for late cervical lymph node metastases in patients with stage I or II carcinoma of the tongue. Head Neck 24:731–736
Pereira CH, Morais MO, Martins AF, Soares MQ, Alencar Rde C, Batista AC, Leles CR, Mendonça EF (2016) Expression of adhesion proteins (E-cadherin and β-catenin) and cell proliferation (Ki-67) at the invasive tumor front in conventional oral squamous cell and basaloid squamous cell carcinomas. Arch Oral Biol 61:8–15
Sen S, Carnelio S (2016) Expression of epithelial cell adhesion molecule (EpCAM) in oral squamous cell carcinoma. Histopathology 68:897–904
Richardson A, Parsons T (1996) A mechanism for regulation of the adhesion-associated proteintyrosine kinase pp125FAK. Nature 380:538–540
Webb DJ, Donais K, Whitmore LA, Thomas SM, Turner CE, Parsons JT, Horwitz AF (2004) FAK-Src signalling through paxillin, ERK and MLCK regulates adhesion disassembly. Nat Cell Biol 6:154–161
Avizienyte E, Wyke AW, Jones RJ, McLean GW, Westhoff MA, Brunton VG, Frame MC (2002) Src-induced de-regulation of E-cadherin in colon cancer cells requires integrin signalling. Nat Cell Biol 4:632–638
Eliceiri BP, Puente XS, Hood JD, Stupack DG, Schlaepfer DD, Huang XZ, Sheppard D, Cheresh DA (2002) Src-mediated coupling of focal adhesion kinase to integrin alpha(v)beta5 in vascular endothelial growth factor signaling. J Cell Biol 157:149–160
Aronsohn MS, Brown HM, Hauptman G, Kornberg LJ (2003) Expression of focal adhesion kinase and phosphorylated focal adhesion kinase in squamous cell carcinoma of the larynx. Laryngoscope 113:1944–1948
Moon HS, Park WI, Choi EA, Chung HW, Kim SC (2003) The expression and tyrosine phosphorylation of E-cadherin/catenin adhesion complex, and focal adhesion kinase in invasive cervical carcinomas. Int J Gynecol Cancer 13:640–646
Brunton VG, Avizienyte E, Fincham VJ, Serrels B, Metcalf CA 3rd, Sawyer TK, Frame MC (2005) Identification of Src-specific phosphorylation site on focal adhesion kinase: dissection of the role of Src SH2 and catalytic functions and their consequences for tumor cell behavior. Cancer Res 65:1335–1342
Kornberg LJ (1998) Focal adhesion kinase expression in oral cancers 20:634–9
Kornberg LJ (1998) Focal adhesion kinase and its potential involvement in tumor invasion and metastasis. Head Neck 20:745–752
Grisaru-Granovsky S, Salah Z, Maoz M, Pruss D, Beller U, Bar-Shavit R (2005) Differential expression of protease activated receptor 1 (Par1) and pY397FAK in benign and malignant human ovarian tissue samples. Int J Cancer 113:372–378
Chiu YW, Liou LY, Chen PT, Huang CM, Luo FJ, Hsu YK, Yuan TC (2016) Tyrosine 397 phosphorylation is critical for FAK-promoted Rac1 activation and invasive properties in oral squamous cell carcinoma cells. Lab Investig 96:296–306
Han X, Xue L, Zhou L, Gong L, Zhu S, Yao L, Wang S, Lan M, Li Y, Zhang W (2013) The role of PTPN13 in invasion and metastasis of lung squamous cell carcinoma. Exp Mol Pathol 95:270–275
Aust S, Auer K, Bachmayr-Heyda A, Denkert C, Sehouli J, Braicu I, Mahner S, Lambrechts S, Vergote I, Grimm C, Horvat R, Castillo-Tong DC, Zeillinger R, Pils D (2014) Ambivalent role of pFAK-Y397 in serous ovarian cancer--a study of the OVCAD consortium. Mol Cancer 13:67
Lai IR, Chu PY, Lin HS, Liou JY, Jan YJ, Lee JC, Shen TL (2010) Phosphorylation of focal adhesion kinase at Tyr397 in gastric carcinomas and its clinical significance. Am J Pathol 177:1629–1637
Albasri A, Fadhil W, Scholefield JH, Durrant LG, Ilyas M (2014) Nuclear expression of phosphorylated focal adhesion kinase is associated with poor prognosis in human colorectal cancer. Anticancer Res 34:3969–3974
Ding L, Sun X, You Y, Liu N, Fu Z (2010) Expression of focal adhesion kinase and phosphorylated focal adhesion kinase in human gliomas is associated with unfavorable overall survival. Transl Res 156:45–52
Duxbury MS, Ito H, Benoit E, Zinner MJ, Ashley SW, Whang EE (2003) RNA interference targeting focal adhesion kinase enhances pancreatic adenocarcinoma gemcitabine chemosensitivity. Biochem Biophys Res Commun 311:786–792
Sonoda Y, Matsumoto Y, Funakoshi M, Yamamoto D, Hanks SK, Kasahara T (2000) Anti-apoptotic role of focal adhesion kinase (FAK). Induction of inhibitor-of-apoptosis proteins and apoptosis suppression by the overexpression of FAK in a human leukemic cell line, HL-60. J Biol Chem 275:16309–16315
Munoz N, Bosch FX, Castellsague X, Diaz M, de Sanjose S, Hammouda D, Shah KV, Meijer CJ (2004) Against which human papillomavirus types shall we vaccinate and screen? The international perspective. Int J Cancer 111:278–285
Khleif SN, DeGregori J, Yee CL, Otterson GA, Kaye FJ, Nevins JR, Howley PM (1996) Inhibition of cyclin D-CDK4/CDK 6 activity is associated with an E2F-mediated induction of cyclin kinase inhibitor activity. Proc Natl Acad Sci U S A 93:424–429
Tong X, Howley PM (1997) The bovine papillomavirus E6 oncoprotein interacts with paxillin and disrupts the actin cytoskeleton. Proc Natl Acad Sci U S A 94:4412–4417
Shintani S, Mihara M, Nakahara Y, Kiyota A, Yoshihama Y, Ueyama Y, Matsumura T (2000) Infrequent alternations of RB pathway(Rb-p16INK4A-cyclinD1) in adenoid cystic carcinoma of salivary glands. Anticancer Res 20:2169–2175
Takeuchi H, Ozawa S, Ando N, Shih CH, Koyanagi K, Ueda M, Kitajima M (1997) Altered p16/MTS1/CDKN2 and cyclin D1/PRAD-1 gene expression is associated with the prognosis of squamous cell carcinoma of the esophagus. Clin Cancer Res 3:2229–2236
Namazie A, Alavi S, Olopade OI, Pauletti G, Aghamohammadi N, Aghamohammadi M, Gornbein JA, Calcaterra TC, Slamon DJ, Wang MB, Srivatsan ES (2002) Cyclin D1 amplification and p16(MTS1/CDK4I)deletion correlate with poor prognosis in head and neck tumors. Laryngoscope 112:472–481
Zafereo ME, Xu L, Dahlstrom KR, Viamonte CA, El-Naggar AK, Wei Q, Li G, Sturgis EM (2016) Squamous cell carcinoma of the oral cavity often overexpresses p16 but is rarely driven by human papillomavirus. Oral Oncol 56:47–53
Kouketsu A, Sato I, Abe S, Oikawa M, Shimizu Y, Takahashi T, Kumamoto H (2016) Detection of human papillomavirus infection in oral squamous cell carcinoma: a cohort study of Japanese patients. J Oral Pathol Med 45:565–572
Götz C, Drecoll E, Straub M, Bissinger O, Wolff KD, Kolk A (2016) Impact of HPV infection on oral squamous cell carcinoma. Oncotarget 7:76704–76712
Ohara T, Kawashiri S, Tanaka A, Noguchi N, Kitahara H, Okamune A, Kato K, Hase T, Nakaya H, Yoshizawa K (2009) Integrin expression levels correlate with invasion, metastasis and prognosis of oral squamous cell carcinoma. Pathol Oncol Res 15:429–436
Raguse JD, Gath HJ, Bier J, Riess H, Oettle H (2004) Cilengitide (EMD 121974) arrests the growth of a heavily pretreated highly vascularised head and neck tumour. Oral Oncol 40:228–230
Brown NF, Williams M, Arkenau HT, Fleming RA, Tolson J, Yan L, Zhang J, Swartz L, Singh R, Auger KR, Lenox L, Cox D, Lewis Y, Plisson C, Searle G, Saleem A, Blagden S, Mulholland P (2018) A study of the focal adhesion kinase inhibitor GSK2256098 in patients with recurrent glioblastoma with evaluation of tumor penetration of [11C]GSK2256098. Neuro-Oncology 20:1634–1642
Jeong K, Murphy JM, Rodriguez YAR, Kim JS, Ahn EE, Lim SS (2019) FAK inhibition reduces metastasis of α4 integrin-expressing melanoma to lymph nodes by targeting lymphatic VCAM-1 expression. Biochem Biophys Res Commun 509:1034–1040
Hu C, Chen X, Wen J, Gong L, Liu Z, Wang J, Liang J, Hu F, Zhou Q, Wei L, Shen Y, Zhang W (2017) Antitumor effect of focal adhesion kinase inhibitor PF562271 against human osteosarcoma in vitro and in vivo. Cancer Sci 108:1347–1356
Shimizu T, Fukuoka K, Takeda M, Iwasa T, Yoshida T, Horobin J, Keegan M, Vaickus L, Chavan A, Padval M, Nakagawa K (2016) A first-in-Asian phase 1 study to evaluate safety, pharmacokinetics and clinical activity of VS-6063, a focal adhesion kinase (FAK) inhibitor in Japanese patients with advanced solid tumors. Cancer Chemother Pharmacol 77:997–1003
Kurio N, Shimo T, Fukazawa T, Okui T, Hassan NM, Honami T, Horikiri Y, Hatakeyama S, Takaoka M, Naomoto Y, Sasaki A (2012) Anti-tumor effect of a novel FAK inhibitor TAE226 against human oral squamous cell carcinoma. Oral Oncol 48:1159–1170
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kato, A., Kato, K., Miyazawa, H. et al. Focal Adhesion Kinase (FAK) Overexpression and Phosphorylation in Oral Squamous Cell Carcinoma and their Clinicopathological Significance. Pathol. Oncol. Res. 26, 1659–1667 (2020). https://doi.org/10.1007/s12253-019-00732-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-019-00732-y