Abstract
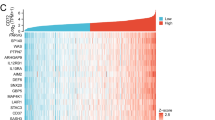
Immunotherapy base on immune checkpoint inhibitor had obtained significant progress in extending the survival of clear cell renal carcinoma (ccRCC) patients. In order to further improve the efficiency of immunotherapy, novel immune checkpoint inhibitors needed to be developed. Differentially expressed genes (DEGs) between healthy kidney tissues and ccRCC tissues had been found from GSE68417 by GEO2R online analysis tool. Correlation analysis and Kaplan–Meier survival analyses were based on UALCAN database. Analyses of the outcome of anti-PD1 treatment had been found from GSE67501 dataset. At first, 9 genes with higher expression were associated with shorter overall survival time. More importantly, higher expression of LGALS1 was correlated with a profitable outcome of anti-PD1 treatment and the combined the expression level of PD-L1 and LGALS1 together could more efficiently predict the outcome of anti-PD1 treatment than using PD-L1 alone. At last, the genes which correlated with LGALS1 expression in ccRCC patients were enriched in TNF alpha Signaling Pathway which is mainly correlated with T cell apoptosis and survival. Together, these suggest LGALS1 could be a potential immune checkpoint, which could promote tumor progression through affecting T cell survival.




Similar content being viewed by others
References
Frew IJ, Moch H (2015) A clearer view of the molecular complexity of clear cell renal cell carcinoma. Annu Rev Pathol 10:263–289
Hecht JR. Current and emerging therapies for metastatic colorectal cancer: applying research findings to clinical practice. American journal of health-system pharmacy : AJHP : official journal of the American Society of Health-System Pharmacists. 2008;65:S15–21; quiz S2–4
Buczek M, Escudier B, Bartnik E, Szczylik C, Czarnecka A (1845) Resistance to tyrosine kinase inhibitors in clear cell renal cell carcinoma: from the patient's bed to molecular mechanisms. Biochim Biophys Acta 2014:31–41
Ciccarese C, Massari F, Santoni M, Heng DY, Sotte V, Brunelli M et al (2015) New molecular targets in non clear renal cell carcinoma: an overview of ongoing clinical trials. Cancer Treat Rev 41:614–622
Vermassen T, De Meulenaere A, Van de Walle M, Rottey S (2017) Therapeutic approaches in clear cell and non-clear cell renal cell carcinoma. Acta Clin Belg 72:12–18
Miao D, Margolis CA, Gao W, Voss MH, Li W, Martini DJ, Norton C, Bossé D, Wankowicz SM, Cullen D, Horak C, Wind-Rotolo M, Tracy A, Giannakis M, Hodi FS, Drake CG, Ball MW, Allaf ME, Snyder A, Hellmann MD, Ho T, Motzer RJ, Signoretti S, Kaelin WG Jr, Choueiri TK, van Allen EM (2018) Genomic correlates of response to immune checkpoint therapies in clear cell renal cell carcinoma. Science. 359:801–806
Choueiri TK, Fishman MN, Escudier B, McDermott DF, Drake CG, Kluger H et al (2016) Immunomodulatory activity of Nivolumab in metastatic renal cell carcinoma. Clinical cancer research : an Official Journal of the American Association for Cancer Research 22:5461–5471
Motzer RJ, Tannir NM, McDermott DF, Aren Frontera O, Melichar B, Choueiri TK et al (2018) Nivolumab plus Ipilimumab versus Sunitinib in advanced renal-cell carcinoma. N Engl J Med 378:1277–1290
Sharpe AH, Pauken KE (2018) The diverse functions of the PD1 inhibitory pathway. Nat Rev Immunol 18:153–167
Thibodeau BJ, Fulton M, Fortier LE, Geddes TJ, Pruetz BL, Ahmed S, et al. Characterization of clear cell renal cell carcinoma by gene expression profiling. Urologic oncology. 2016;34:168.e1–9
Ascierto ML, McMiller TL, Berger AE, Danilova L, Anders RA, Netto GJ et al (2016) The Intratumoral balance between metabolic and immunologic gene expression is associated with anti-PD-1 response in patients with renal cell carcinoma. Cancer immunology research 4:726–733
Yang W, Ma J, Zhou W, Li Z, Zhou X, Cao B et al (2019) Identification of hub genes and outcome in colon cancer based on bioinformatics analysis. Cancer Manag Res 11:323–338
Jha PK, Vijay A, Sahu A, Ashraf MZ (2016) Comprehensive gene expression meta-analysis and integrated bioinformatic approaches reveal shared signatures between thrombosis and myeloproliferative disorders. Sci Rep 6:37099
Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, Koplev S, Jenkins SL, Jagodnik KM, Lachmann A, McDermott MG, Monteiro CD, Gundersen GW, Ma'ayan A (2016) Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res 44:W90–W97
Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, et al. UALCAN: A Portal for Facilitating Tumor Subgroup Gene Expression and Survival Analyses. Neoplasia (New York, NY). 2017;19:649–58
Lou W, Chen J, Ding B, Chen D, Zheng H, Jiang D, Xu L, Bao C, Cao G, Fan W (2018) Identification of invasion-metastasis-associated microRNAs in hepatocellular carcinoma based on bioinformatic analysis and experimental validation. J Transl Med 16:266
Pomer S, Schirrmacher V, Thiele R, Lohrke H, Brkovic D, Staehler G (1995) Tumor response and 4 year survival-data of patients with advanced renal-cell carcinoma treated with autologous tumor vaccine and subcutaneous R-IL-2 and IFN-alpha(2b). Int J Oncol 6:947–954
Picarda E, Ohaegbulam KC, Zang X (2016) Molecular pathways: targeting B7-H3 (CD276) for human Cancer immunotherapy. Clinical Cancer Research : an Official Journal of the American Association for Cancer Research 22:3425–3431
He J, Baum LG (2004) Presentation of galectin-1 by extracellular matrix triggers T cell death. J Biol Chem 279:4705–4712
Ramanan VK, Risacher SL, Nho K, Kim S, Shen L, McDonald BC et al (2015) GWAS of longitudinal amyloid accumulation on 18F-florbetapir PET in Alzheimer's disease implicates microglial activation gene IL1RAP. Brain J Neurol 138:3076–3088
Smith NL, Hankinson J, Simpson A, Denning DW, Bowyer P (2014) Reduced expression of TLR3, TLR10 and TREM1 by human macrophages in chronic cavitary pulmonary aspergillosis, and novel associations of VEGFA, DENND1B and PLAT. Clinical Microbiology and Infection : Official Publication of the European Society of Clinical Microbiology and Infectious Diseases 20:O960–O968
Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, Miller ML, Rekhtman N, Moreira AL, Ibrahim F, Bruggeman C, Gasmi B, Zappasodi R, Maeda Y, Sander C, Garon EB, Merghoub T, Wolchok JD, Schumacher TN, Chan TA (2015) Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 348:124–128
Castro MP, Goldstein N (2015) Mismatch repair deficiency associated with complete remission to combination programmed cell death ligand immune therapy in a patient with sporadic urothelial carcinoma: immunotheranostic considerations. Journal for immunotherapy of cancer 3:58
Astorgues-Xerri L, Riveiro ME, Tijeras-Raballand A, Serova M, Neuzillet C, Albert S, Raymond E, Faivre S (2014) Unraveling galectin-1 as a novel therapeutic target for cancer. Cancer Treat Rev 40:307–319
Cedeno-Laurent F, Dimitroff CJ. Galectin-1 research in T cell immunity: past, present and future. Clinical immunology (Orlando, Fla). 2012;142:107–16
Perillo NL, Pace KE, Seilhamer JJ, Baum LG (1995) Apoptosis of T cells mediated by galectin-1. Nature. 378:736–739
Ishitani T, Takaesu G, Ninomiya-Tsuji J, Shibuya H, Gaynor RB, Matsumoto K (2003) Role of the TAB2-related protein TAB3 in IL-1 and TNF signaling. EMBO J 22:6277–6288
Wang L, Feng W, Yang X, Yang F, Wang R, Ren Q, Zhu X, Zheng G (2018) Fbxw11 promotes the proliferation of lymphocytic leukemia cells through the concomitant activation of NF-kappaB and beta-catenin/TCF signaling pathways. Cell Death Dis 9:427
Acknowledgments
This study was supported by research grants from, the National Natural Science Foundation of China (No. 81500544, 81873605, 81700379, 81500561), the Research on The Basis and Frontier of Chongqing (grant no. cstc2016jcyjA0049).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Y., Yang, S., Yue, H. et al. Unraveling LGALS1 as a Potential Immune Checkpoint and a Predictor of the Response to Anti-PD1 Therapy in Clear Cell Renal Carcinoma. Pathol. Oncol. Res. 26, 1451–1458 (2020). https://doi.org/10.1007/s12253-019-00710-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-019-00710-4