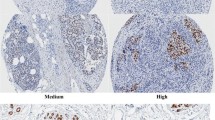
Abstract
Metastasis represents a major problem in the treatment of patients with advanced primary breast cancer. Both Transforming Growth Factor-Beta (TGF-β) signaling and Plasminogen Activator (PA) components, urokinase-type Plasminogen Activator (uPA) and Plasminogen Activator Inhibitor-1 (PAI-1) represent a complex network crucial for such enhanced invasiveness of tumors and imply high prognostic/predictive and promising therapeutic potential. Therefore, protein expression of specific effector molecules comprising the main parts of the TGF-β signaling pathway were determined in HOPE-fixed human tumor tissues through IHC (Scoring) using tissue microarray (TMA) technique and correlated with respective uPA and PAI-1 levels determined earlier in the same TMAs through optimized IHC and semi-quantitative image analysis. TGF-β signaling was active in vast majority (96 %) of the tumor samples and 88 % of all cases were significantly correlated with established metastasis markers uPA and PAI-1. In addition, TGF-β was also closely associated with tumor size, nodal status and two steroid hormone receptors. Consistent interrelationships between TGF-β, PA components and additional tumor characteristics underline the superiority of such more comprising data with regards to confirming TGF-β signaling as a promising target system to inhibit metastasis in advanced breast cancer.

Similar content being viewed by others
References
Welch DR, Steeg PS, Rinker-Schaeffer CW (2000) Molecular biology of breast cancer metastasis. Genetic regulation of human breast carcinoma metastasis. Breast Cancer Res 2:408–412
Lyer S, Wang Z-G, Akhtari M, Zhao W, Seth P (2005) Targeting TGFβ signaling for cancer therapy. Cancer Biol Ther 4(3):261–266
Malinowsky K, Böllner C, Hipp S, Berg D, Schmitt M, Becker KF (2010) UPA and PAI-1 analysis from fixed tissues—new perspectives for a known set of predictive markers. Curr Med Chem 17:4370–4377 (Review)
Jo M, Eastman BM, Webb DL, Stoletov K, Klemke R, Gonias SL (2010) Cell signaling by urokinase-type plasminogen activator receptor induces stem cell-like properties in breast cancer cells. Cancer Res 70(21):8948–8958
Harbeck N, Schmitt M, Vetter M, Krol J, Paepke D, Uhlig M, Paepke S, Jänicke F, Geurts-Moespot A, von Minckwitz G, Sweep F, Thomssen C (2008) Prospective biomarker trials Chemo NO and NNBC-3 Europe validate the clinical utility of invasion markers uPA and PAI-1 in node-negative breast cancer. Breast Care 3(suppl 2):11–15
Kreienberg R, Albert U-S, Follmann M, Kopp I, Kühn T, Wöckel A, Zemmler T (2012) Interdisziplinäre S-3 Leitlinie für Diagnostik, Therapie und Nachsorge des Mammakarzinoms. Published by German Cancer Society e.V
Shiou S-R, Datta PK, Dhawan P, Law BK, Yingling JM, Dixon DA, Beauchamp RD (2006) Smad4-dependent regulation of urokinase plasminogen activator secretion and RNA stability associated with invasiveness by autocrine and paracrine transforming growth factor-β. J Biol Chem 281:33971–33981
Song X, Thalacker FW, Nilsen-Hamilton M (2012) Synergistic and multidimensional regulation of plasminogen activator inhibitor type 1 expression by transforming growth factor type β and epidermal growth factor. J Biol Chem 287:12520–12528
Kohn EA, Yang YA, Du Z, Nagano Y, Van Schyndle CM, Herrmann MA, Heldman M, Chen J-Q, Stuelten CH, Flanders KC (2012) Wakefield LM (2012) Biological responses of TGF-β in the mammary epithelium show a complex dependency on Smad3 gene dosage with important implications for tumor progression. Mol Cancer Res 10(10):1389–1399
Kocic J, Bugarski D, Santibanez JF (2012) Smad3 is essential for transforming growth factor-β1-induced urokinase type plasminogen activator expression and migration in transformed keratinocytes. Eur J Cancer 48:1550–1557
Onichtchouk D, Chen YG, Dosch R, Gawantka V, Delius H, Massaque J, Niehrs C (1999) Silencing of TGF-beta signaling by the pseudoreceptor BAMBI. Nature 401:480–485
Drömann D, Rupp J, Rohmann K, Osbahr S, Ulmer A, Marwitz S, Röschmann K, Abdullah M, Schultz H, Vollmer E, Zabel P, Dalhoff K, Goldmann T (2010) The TGF-beta- pseudoreceptor BAMBI is strongly expressed in COPD lungs and regulated by nontypeable Haemophilus influenzae. Respir Res 11:67
Ito I, Hanyu A, Wayama M, Goto N, Katsuno N, Kawasaki S, Nakajima Y, Kajiro M, Komatsu Y, Fujimura A, Hirota R, Murayama A, Kimura K, Imamura T, Yanagisawa J (2010) Estrogen inhibits transforming growth factor β signaling by promoting Smad2/3 degradation. J Biol Chem 285:14747–14755
Goto N, Hiyoshi H, Ito I, Tsuchiya M, Nakajima Y, Yanagisawa J (2011) Estrogen and antiestrogens alter breast cancer invasiveness by modulating the transforming growth factor-β signaling pathway. Cancer Sci 102:1501–1508
Lang DS, Heilenkötter U, Schumm W, Behrens O, Simon R, Vollmer E, Goldmann T (2013) Optimized immunohistochemistry in combination with image analysis: a reliable alternative to quantitative ELISA determination of uPA and PAI-1 for routine risk group discrimination in breast cancer. Breast. doi:10.1016/j.breast.2012.12.011
Schulz H, Kähler D, Branscheid D, Vollmer E, Zabel P, Goldmann T (2008) TKTL1 is overexpressed in a large portion of non-small cell lung cancer specimens. Diagn Pathol 3:35
Vollmer E, Galle J, Lang DS, Loeschke S, Schultz H, Goldmann T (2006) The HOPE technique opens up a multitude of new possibilities in pathology. Rom J Morphol Embryol 47(1):15–19 (Review)
Marwitz S, Abdullah M, Vock C, Fine JS, Visvanathan S, Gaede KI, Hauber HP, Zabel P, Goldmann T (2011) HOPE-BAL: improved molecular diagnostics by application of a novel technique for fixation and paraffin embedding. J Histochem Cytochem 59(6):601–614
Tobar N, Villar V, Santibanez JF (2010) ROS-NFkB mediates TGF-β1-induced expression of urokinase-type plasminogen activator, matrix metalloproteinase-9 and cell invasion. Mol Cell Biochem 340:195–202
Giehl K, Imamichi Y, Menke A (2007) Smad4-independent TGF–β signaling in tumor cell migration. Cells Tissues Organs 185:123–130
Wakefield LM, Roberts AB (2002) TGF-b signaling: positive and negative effects on tumorigenesis. Curr Opin Genet Dev 12:22–29 (Review)
Wolff C, Malinoswky K, Berg D, Schragner K, Schuster T, Walch A, Bronger H, Höfler H, Becker K-F (2011) Signalling networks associated with urokinase-type plasminogen activator (uPA) and ist inhibitor PAI-1 in breast cancer tissues: new insights from protein microarray analysis. J Pathol 223:54–63
Malinowsky K, Wolff C, Berg D, Schuster T, Walch A, Bronger H, Mannsperger H, Schmidt C, Korf U, Höfler H, Becker K-F (2012) uPA and PAI-1-related signaling pathways differ between primary breast cancers and lymph node metastases. Trans Oncol 5:98–104
Czekay R-P, Wilkins-Port CE, Higgins SP, Freytag J, Overstreet JM, Klein RM, Higgins CE, Samarakoon R, Higgins PJ (2011) PAI-1: an integrator of cell signaling and migration. Int J Cell Biol 2011:1–9 (Review)
Huang SS, Ling T-Y, Tseng W-F, Huang Y-W, Tang F-M, Leal SM, Huang JS (2003) FASEB J 17:2068–2080
Khin SS, Kitazawa R, Win N, Aye TT, Mori K, Kondo T, Kitazawa S (2009) BAMBI gene is epigenetically silenced in subset of high-grade bladder. Int J Cancer 125:328–338
Ganapathy V, Rongrong G, Grazioli A, Xie W, Banach-Petrosky W, Kang Y, Lonning S, McPherson J, Yingling JM, Biswas S, Mundy GR, Reiss M (2010) Targeting the transforming growth factor–β pathway inhibits human basal-like breast cancer metastasis. Mol Cancer 9:122
Liu J, Liao S, Diop-Frimpong B, Chen W, Goel S, Naxerova K, Ancukiewicz M, Boucher Y, Jain RK, Xu L (2012) TGF-β blockage improves the distribution and efficiacy of therapeutics in breast carcinoma by normalizing the tumor stroma. PNAS 109:16618–16623
Matise LA, Palmer TD, Ashby WJ, Nashabi A, Chytil A, Aakre M, Pickup MW, Gorska AE, Zijlstra A, Moses HL (2012) Lack of transforming growth factor-β signaling promotes collective cancer cell invasion through tumor-stromal crosstalk. Breast Cancer Res 14:R98
Acknowledgments
The authors would like to thank Jasmin Tiebach, Maria Lammers and Stefanie Fox for their excellent technical support.
Conflict of Interest
There is no conflict of interest for any of the authors.
Ethics
The research protocol has been approved by the Ethics committees of Bad Segeberg (39/09) and of the University of Lübeck (07–157).
Authors’ Contributions
DSL carried out the semi-quantitative image analyses, statistical evaluation and drafted the manuscript. SM carried out and evaluated the staining’s of the TGF-β pathway members and was involved in the drafting of the manuscript. UH and OB were responsible for the surgical part and clinical data. WS and RS provided the surgical tissue material following routine pathology. MR has contributed in writing and finalizing the manuscript. EV was responsible for the histopathological aspects. TG conceived of the study and was involved in drafting the manuscript. All authors have read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
D.S. Lang and S. Marwitz contributed equally to this research.
Rights and permissions
About this article
Cite this article
Lang, D.S., Marwitz, S., Heilenkötter, U. et al. Transforming Growth Factor-Beta Signaling Leads to uPA/PAI-1 Activation and Metastasis: A Study on Human Breast Cancer Tissues. Pathol. Oncol. Res. 20, 727–732 (2014). https://doi.org/10.1007/s12253-014-9753-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9753-2