Abstract
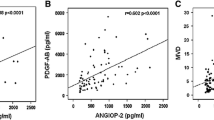
In multiple myeloma (MM), angiogenesis plays a substantial role in disease progression. Interleukin-8 (IL-8), a pro-inflammatory chemokine with potent pro-angiogenic properties, has been implicated in the pathophysiology of MM. The aim of the study is to measure serum levels of IL-8 in MM patients and to correlate them with markers of angiogenesis, such as circulating levels of platelet derived growth factor-AB (PDGF-AB) and angiogenin (Ang), and bone marrow microvascular density (MVD). Fifty-three newly diagnosed MM patients, 23 of them, who reached plateau phase after effective treatment and 20 healthy controls, were studied. Serum levels of PDGF-AB, Ang and IL-8 were measured by ELISA, whereas bone marrow MVD was estimated by immunohistochemical staining of vessels with anti-CD31. All measured parameters were higher in MM patients compared to controls and in increased disease stages. They all also significantly decreased in plateau phase. IL-8 correlated positively with Ang and PDGF-AB, but not with MVD. The circulating levels of IL-8, PDGF-AB and Ang are elevated in patients with MM. The lack of correlation between IL-8 with MVD suggests that its levels represent the inflammatory element of MM disease and the participation in angiogenesis process is rather complex with multifactorial mechanisms.


Similar content being viewed by others
References
Hideshima T, Mitsiades C, Tonon G, Richardson PG, Anderson KC (2007) Understanding multiple myeloma pathogenesis in the bone marrow to identify new therapeutic targets. Nat Rev Cancer 7:585–598
Sezer O, Niemoller K, Eucker J, Jacob C, Kaufmann O, Zavrski I, Dietel M, Possinger K (2000) Bone marrow microvessel density is a prognostic factor for survival in patients with multiple myeloma. Ann Hemal 79:574–577
Tsirakis G, Pappa C, Kanellou P, Stratinaki M, Xekalou A, Psarakis F, Sakellaris G, Alegakis A, Stathopoulos E, Alexandrakis M (2012) Role of platelet-derived growth factor-AB in tumour growth and angiogenesis in relation with other angiogenic cytokines in multiple myeloma. Hematol Oncol 30:131–136
Kastritis E, Roussou M, Michalis M, Gavriatopoulou M, Michalis E, Migkou M, Delimpasi S, Kyrtsonis MC, Gogos D, Liapis K, Charitaki E, Repousis P, Terpos E, Dimopoulos A (2010) On behalf of the Greek Myeloma Study Group. High levels of serum angiogenic growth factors in patients with AL amyloidosis: comparisons with normal individuals and multiple myeloma patients. Br J Haematol 150:587–591
Tsirakis G, Pappa C, Kaparou M, Katsomitrou V, Xatzivasili A, Alegakis T, Xekalou A, Stathopoulos E, Alexandrakis M (2011) Assessment of proliferating cell nuclear antigen and its relationship with proinflammatory cytokines and parameters of disease activity in multiple myeloma patients. Eur J Histochem 55(e21):113–116
Singh R, Varney M (2000) Il-8 expression in malignant melanoma: implications in growth and metastasis. Histol Histopathol 15:843–849
Kline M, Donovan K, Wellik L, Jin W, Moon-Tasson L, Xiong Y, Witsig TE, Kumar S, Rajkumar S, Lust J (2007) Cytokine and chemokine profiles in multiple myeloma; significance of stromal interaction and correlation of IL-8 production with disease progression. Leuk Res 31:591–598
Pappa C, Tsirakis G, Kanellou P, Kaparou M, Stratinaki M, Xekalou A, Alegakis A, Boula A, Stathopoulos E, Alexandrakis M (2011) Monitoring serum levels ELR + CXC chemokines and the relationship between microvessel density and angiogenic growth factors in multiple myeloma. Cytokine 56:616–620
Alexandrakis MG, Passam FH, Pappa CA, Dambaki C, Sfakiotaki G, Alegakis AK, Kyriakou DS, Stathopoulos E (2004) Expression of proliferating cell nuclear antigen (PCNA) in multiple myeloma: its relationship to bone marrow microvessel density and other factors of disease activity. Int J Immunopathol Pharmacol 17:49–56
Wright D, Bowman E, Wagers A, Butcher E, Weissman I (2002) Hematopoietic stem cells are uniquely selective in the migratory response to chemokines. J Exp Med 195:1145–1154
Qazi BS, Tang K, Qazi A (2011) Recent advances in underlying pathologies provide insight into interleukin-8 expression-mediated inflammation and angiogenesis. Int J Inflamm 2011:908468
Dobreva I, Waeber G, James RW, Widmann C (2006) Interleukin-8 secretion by fibroblasts induced by low density lipoproteins is p38 MAPK-dependent and leads to cell spreading and wound closure. J Biol Chem 281:199–205
Aggarwal R, Ghobrial IM, Roodman GD (2006) Chemokines in multiple myeloma. Exp Hematol 34:1289–1295
Benelli R, Lorusso G, Albini A, Noonan DM (2006) Cytokines and chemokines as regulators of angiogenesis in health and disease. Curr Pharm Des 12:3101–3115
Markovina S, Callander NS, O'Connor SL, Xu G, Shi Y, Leith CP, Kim K, Trivedi P, Kim J, Hematti P, Miyamoto S (2010) Bone marrow stromal cells from multiple myeloma patients uniquely induce bortezomib resistant NF-kappaB activity in myeloma cells. Mol Canc 9:176
Benelli R, Morini M, Carrozzino F, Ferrari N, Minghelli S, Santi L, Cassatella M, Noonan DM, Albini A (2002) Neutrophils as a key cellular target for angiostatin: implications for regulation of angiogenesis and inflammation. FASEB J 16:267–269
Benelli R, Albini A, Noonan D (2003) Neutrophils and angiogenesis: potential initiators of the angiogenic cascade. Chem Immunol Allergy 83:167–181
Scapini P, Morini M, Tecchio C, Minghelli S, Di Carlo E, Tanghetti E, Albini A, Lowell C, Berton G, Noonan DM, Cassatella MA (2004) CXCL1/macrophage inflammatory protein-2-induced angiogenesis in vivo is mediated by neutrophil-derived vascular endothelial growth factor-A. J Immunol 172:5034–5040
Li A, Varney ML, Valasek J, Godfrey M, Dave BJ, Singh RK (2005) Autocrine role of interleukin-8 in induction of endothelial cell proliferation, survival, migration and MMP-2 production and angiogenesis. Angiogenesis 8:63–71
Huang S, Mills L, Mian B, Tellez C, McCarty M, Yang XD, Gudas JM, Bar-Eli M (2002) Fully humanized neutralizing antibodies to interleukin-8 (ABX-IL8) inhibit angiogenesis, tumor growth, and metastasis of human melanoma. Am J Pathol 161:125–134
Dong X, Han ZC, Yang R (2007) Angiogenesis and antiangiogenic therapy in hematologic malignancies. Crit Rev Oncol Hematol 62:105–118
Negaard HF, Iversen N, Bowitz-Lothe IM, Sandset PM, Steinsvik B, Ostenstad B, Iversen PO (2009) Increased bone marrow microvascular density in haematological malignancies is associated with differential regulation of angiogenic factors. Leukemia 23:162–169
Rakjumar V, Mesa R, Fonseca R, Schroeder G, Plevak M, Dispenzieri A, Lacy M, Lust J, Witzig T, Gertz M, Kule R, Russell S, Greipp P (2002) Bone marrow angiogenesis in 400 patients with monoclonal gammopathy of undetermined significance, multiple myeloma and primary amyloidosis. Clin Canc Res 8:2210–2216
Zhang J, Cao R, Zhang Y, Jia T, Cao Y, Wahlberg E (2009) Differential roles of PDGFR-alpha and PDGFR-beta in angiogenesis and vessel stability. FASEB J 23:153–163
Miyagaki T, Sugaya M, Suga H, Akamata K, Ohmatsu H, Fujita H, Asano Y, Tada Y, Kadono T, Sato S (2012) Angiogenin levels are increased in lesional skin and sera in patients with erythrodermic cutaneous T cell lymphoma. Arch Dermatol Res 304:401–406
Hu GF, Riordan JF (1993) Angiogenin enhances actin acceleration of plasminogen activation. Biochem Biophys Res Commun 197:682–687
Hu GF, Chang SI, Riordan JF, Vallee BL (1991) An angiogenin-binding protein from endothelial cells. Proc Natl Acad Sci USA 88:2227–2231
Dungwa J, Uparkar U, May M, Ramani P (2012) Angiogenin up-regulation correlates with adverse clinicopathological and biological factors, increased microvascular density and poor patient outcome in neuroblastomas. Histopathology 60:911–923
Li S, Ibaragi S, Hu GF (2011) Angiogenin as a molecular target for the treatment of prostate cancer. Curr Canc Ther Rev 7:83–90
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pappa, C.A., Tsirakis, G., Boula, A. et al. The Significance of non Correlation Between Interleukin-8 Serum Levels with Bone Marrow Microvascular Density in Patients with Myeloma Multiple. Pathol. Oncol. Res. 19, 539–543 (2013). https://doi.org/10.1007/s12253-013-9614-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-013-9614-4