Abstract
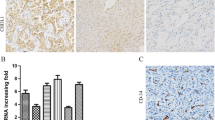
N-myc downstream regulated gene 1 (NDRG1) is a member of the N-myc downstream regulated gene family which belongs to the alpha/beta hydrolase superfamily. Earlier studies have shown its association with inhibition of tumor metastasis. However, its function in malignant tumors is not fully enunciated. Recently there was increasing evidence that NDRG1 is involved in stress responses. In the current study, we examined the expression of NDRG1 and its correlation with clinicopathological factors and microvessel density (MVD) in non-small cell lung cancer (NSCLC) using immunohistochemistry (IHC). NDRG1 expression in NSCLC (71/115, 61.7%) was higher than that in normal lung tissues (32/115, 27.8%) (p < 0.05). NDRG1 expression in NSCLC cells was found in cytoplasm (63/115, 54.8%), nuclear (24/115, 20.9%) and cell membrane (13/115, 11.3%). NDRG1 expression in NSCLC with advanced T stages (T2-4) (63/84, 75.0%) was significantly higher than that with T1 stage (8/31, 25.8%) (P < 0.05). No other clinicopathological factors including lymph node metastasis were found to be associated with NDRG1 expression (p > 0.05). Moreover increased NDRG1 expression was associated with lower MVD in NSCLC (P < 0.05). MVD in adenocarcinoma (33.4 ± 8.4/HP) was significantly higher than that in squamous cell carcinoma (SCC) (19.3 ± 8.1/HP) (P < 0.05). No other clinicopathological factors were associated with MVD in NSCLC (p > 0.05). The present findings indicate an increase of NDRG1 expression with the progress of tumour extent which may be due to unbalanced tumor oxygenation on account of poor vascularization in NSCLC.




Similar content being viewed by others
Abbreviations
- NDRG1:
-
N-myc downstream regulated gene 1
- MVD:
-
Microvessel density
- NSCLC:
-
Non-small cell lung cancer
- IHC:
-
Immunohistochemistry
- UICC:
-
International Union Against Cancer
- WHO:
-
World Health Organization
- SCC:
-
Squamous cell carcinoma
References
Lachat P, Shaw P, Gebhard S et al (2002) Expression of NDRG1, a differentiation-related gene, in human tissues. Histochem Cell Biol 118:399–408
Taketomi Y, Sugiki T, Saito T et al (2003) Identification of NDRG1 as an early inducible gene during in vitro maturation of cultured mast cells. Biochem Biophys Res Commun 306:339–346
Wakisaka Y, Furuta A, Masuda K et al (2003) Cellular distribution of NDRG1 protein in the rat kidney and brain during normal postnatal development. J Histochem Cytochem 51:1515–1525
Rutherford MN, Bayly GR, Matthews BP et al (2001) The leukemogenic transcription factor E2a-Pbx1 induces expression of the putative N-myc and p53 target gene NDRG1 in Ba/F3 cells. Leukemia 15:362–370
Angst E, Sibold S, Tiffon C et al (2006) Cellular differentiation determines the expression of the hypoxia-inducible protein NDRG1 in pancreatic cancer. Br J Cancer 95:307–313
Echaniz-Laguna A, Degos B, Bonnet C et al (2007) NDRG1-linked Charcot-Marie-Tooth disease (CMT4D) with central nervous system involvement. Neuromuscul Disord 17:163–168
Zhou D, Salnikow K, Costa M (1998) Cap43, a novel gene specially induced by Ni2+ compounds. Cancer Res 58:2182–2189
Salnikow K, Blagosklonny MV, Ryan H et al (2000) Carcinogenic nickel induces genes involved with hypoxic stress. Cancer Res 60:38–41
Hakan C (2004) Hypoxia upregulates the expression of the NDRG1 gene leading to its overexpression in various human cancers. BMC Genet 5:27
Joseph TC, Hung-Ming W, Ku-Wei C et al (2005) Identification of differentially expressed genes in oral squamous cell carcinoma (OSCC): Overexpression of NPM, CDK1 and NDRG1 and underexpression of CHES1. Int J Cancer 114:942–949
Sonja S, Vincent R, Adrian K et al (2007) Hypoxia increases cytoplasmic expression of NDRG1, but is insufficient for its membrane localization in human hepatocellular carcinoma. FEBS Lett 581:989–994
Mei-Sze C, Hongbo S, Siu TC et al (2007) Overexpression of NDRG1 is an indicator of poor prognosis in hepatocellular carcinoma. Mod Pathol 20:76–83
Sobin DHWC (2002) International Union Against Cancer (UICC): TNM Classification of Malignant Tumors, 6. Wiley-Liss, New York
Travis WDBE, Muller-Hermelink HK, Harris CC (2004) World health organization classification of tumors: pathology and genetics of tumors of the lung, pleura, thymus and heart. IARC, Lyon
Weidner N, Semple JP, Welch WR et al (1991) Tumor angiogenesis and metastasis-correlation in invasive breast carcinoma. N Engl J Med 324:1–8
Bandyopadhyay S, Pai SK, Gross SC et al (2003) The Drg-1 gene suppresses tumor metastasis in prostate cancer. Cancer Res 63:1731–1736
Bandyopadhyay S, Pai SK, Hirota S et al (2004) Role of the putative tumor metastasis suppressor gene Drg-1 in breast cancer progression. Oncogene 23:5675–5681
Ulrix W, Swinnen JV, Heyns W et al (1999) The differentiation-related gene 1, Drg1, is markedly upregulated by androgens in LNCaP prostatic adenocarcinoma cells. FEBS Lett 455:23–26
Kokame K, Kato H, Miyata T (1996) Homocysteine-respondent genes in vascular endothelial cells identified by differential display analysis. GRP78/BiP and novel genes. J Biol Chem 271:29659–29665
Shin N, Kimio U, Naotake T et al (2008) Cap43/NDRG1/Drg-1 is a molecular target for angiogenesis and a prognostic indicator in cervical adenocarcinoma. Cancer Letters 264:36–43
Maruyama Y, Ono M, Kawahara A et al (2006) Tumor growth suppression in pancreatic cancer by a putative metastasis suppressor gene Cap43/NDRG1/Drg-1 through modulation of angiogenesis. Cancer Res 66:6233–6242
Guan RJ, Ford HL, Fu Y et al (2000) Drg-1 as a differentiation-related, putative metastatic suppressor gene in human colon cancer. Cancer Res 60:749–755
Macchiarini P, Fontanini G, Hardin MJ et al (1992) Relation of neovascularisation to metastasis of non-small cell lung cancer. Lancet 340:145–146
Fontanini G, Bigini D, Vignati S et al (1995) Microvessel count predicts metastatic disease and survival in non-small cell lung cancer. J Pathol 177:57–63
Maeda K, Chung YS, Takatsuka S et al (1995) Tumor angiogenesis as a predictor of recurrence in gastric carcinoma. J Clin Oncol 13:477–481
Yamazaki K, Abe S, Takekawa H et al (1994) Tumor angiogenesis in human lung adenocarcinoma. Cancer 74:2245–2250
Anne E, Sebastian K, Valentin G et al (2000) Heterogeneity of angiogenesis and blood vessel maturation in human tumors: implications for antiangiogenic tumor therapies. Cancer Res 60:1388–1393
Fumihiro T, Hiroki O, Kazumasa T et al (2003) Glomeruloid microvascular proliferation is superior to intratumoral microvessel density as a prognostic marker in non-small cell lung cancer. Cancer Res 63:6791–6794
Guo J, Higashi K, Ueda Y et al (2006) Microvessel density: correlation with 18F-FDG uptake and prognostic impact in lung adenocarcinomas. J Nucl Med 47:419–425
Trivella M, Pezzella F, Pastorino U et al (2007) Microvessel density as a prognostic factor in non-small-cell lung carcinoma: a meta-analysis of individual patient data. Lancet Oncol 8:488–499
Sardari NP, Stessels F, Pezzella F et al (2003) Growth index is independent of microvessel density in non-small cell lung carcinomas. Hum Pathol 34:959–960
Zheng Y, Wang LS, Xia L et al (2009) NDRG1 is down-regulated in the early apoptotic event induced by camptothecin analogs: the potential role in proteolytic activation of PKC delta and apoptosis. Proteomics 9:2064–2075
Kurdistani SK, Arizti P, Reimer CL et al (1998) Inhibition of tumor cell growth by RTP/rit42 and its responsiveness to p53 and DNA damage. Cancer Res 58:4439–4444
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, C., Yu, J., Liu, Y. et al. Increased NDRG1 Expression is Associated with Advanced T Stages and Poor Vascularization in Non-small Cell Lung Cancer. Pathol. Oncol. Res. 18, 549–556 (2012). https://doi.org/10.1007/s12253-010-9294-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-010-9294-2