Abstract
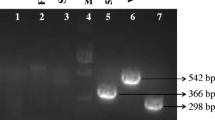
Seneca Valley virus (SVV), a newly determined etiological agent of vesicular disease in swine, causes porcine idiopathic disease and occasional acute death in piglets. Recently, an increased number of SVV infection cases have been reported in the United States (US) and China, resulting in significant economic losses to the swine industry. The first identification of SVV in China was reported in Guangdong Province, a major swine producing province. The cases of SVV were continuously reported in Guangdong in 2015 and 2016. However, the spread of SVV in Guangdong in 2017 remains unknown. In this study, we determined two new SVV strains, CH-GD-2017-1 and CH-GD-2017-2, from Guangdong. The genetic analysis suggested that the two Guangdong strains showed different characteristics to previous Guangdong strains. They showed lower nucleotide similarity with strains isolated in 2015 and 2016, and were more similar to the US strains. Phylogenetic analyses indicated that the new strains were clustered in a different clade with previous Guangdong strains. We found 28 mutated amino acids in the new strains, compared with the first Guangdong strain, SVV CH-01-2015. In the geographic analysis, we found that the US and China reported more SVV cases than other countries, and most of the SVV cases were reported in east and central China—of which, Guangdong Province is one of the major epidemic regions. In conclusion, our findings indicate that SVV continued to spread in Guangdong Province in 2017, and two different clades of SVVs have emerged in this region.



Similar content being viewed by others
References
Bao HF, Li D, Guo JH, Lu ZJ, Chen YL, Liu ZX, Liu XT, Xie QG (2008) A highly sensitive and specific multiplex RT-PCR to detect foot-and-mouth disease virus in tissue and food samples. Arch Virol 153:205–209
Bracht AJ, O’Hearn ES, Fabian AW, Barrette RW, Sayed A (2016) Real-time reverse transcription pcr assay for detection of Senecavirus A in swine vesicular diagnostic specimens. PLoS ONE 11:e0146211
Canning P, Canon A, Bates JL, Gerardy K, Linhares DC, Piñeyro PE, Schwartz KJ, Yoon KJ, Rademacher CJ, Holtkamp D (2016) Neonatal mortality, vesicular lesions and lameness associated with Senecavirus A in a U.S. Sow Farm. Transbound Emerg Dis 63:373
Chen Z, Yuan F, Li Y, Shang P, Schroeder R, Lechtenberg K, Henningson J, Hause B, Bai J, Rowland RR, Clavijo A, Fang Y (2016) Construction and characterization of a full-length cDNA infectious clone of emerging porcine Senecavirus A. Virology 497:111–124
Domingo E, Holland JJ (1997) RNA virus mutations and fitness for survival. Annu Rev Microbiol 51:151–178
Hales LM, Knowles NJ, Reddy PS, Xu L, Hay C, Hallenbeck PL (2008) Complete genome sequence analysis of Seneca Valley virus-001, a novel oncolytic picornavirus. J Gen Virol 89:1265–1275
Joshi LR, Mohr KA, Clement T, Hain KS, Myers B, Yaros J, Nelson EA, Christopher-Hennings J, Gava D, Schaefer R, Caron L, Dee S, Diel DG (2016) Detection of the emerging picornavirus Senecavirus A in pigs, mice, and houseflies. J Clin Microbiol 54:1536–1545
Knowles N, Hallenbeck P (2005) A new picornavirus is most closely related to cardioviruses. In: Abstract of the EUROPIC 2005—13th meeting of the European Study Group on the molecular biology of picornaviruses, Lunteren, the Netherlands, pp 23–29
Leme RA, Oliveira TE, Alcantara BK, Headley SA, Alfieri AF, Yang M, Alfieri AA (2016) Clinical manifestations of Senecavirus A infection in neonatal pigs, Brazil, 2015. Emerg Infect Dis 22:1238–1241
Leme RA, Alfieri AF, Alfieri AA (2017) Update on Senecavirus infection in pigs. Viruses. https://doi.org/10.3390/v9070170
Maggioli MF, Lawson S, de Lima M, Joshi LR, Faccin TC, Bauermann FV, Diel DG (2017) Adaptive immune responses following Senecavirus A infection in pigs. J Virol. https://doi.org/10.1128/JVI.01717-17
Montiel N, Buckley A, Guo B, Kulshreshtha V, VanGeelen A, Hoang H, Rademacher C, Yoon KJ, Lager K (2016) Vesicular disease in 9-week-old pigs experimentally infected with Senecavirus A. Emerg Infect Dis 22:1246–1248
Pasma T, Davidson S, Shaw SL (2008) Idiopathic vesicular disease in swine in Manitoba. Can Vet J 49:84–85
Qian SH, Fan WC, Qian P, Chen HC, Li XM (2016) Isolation and full-genome sequencing of Seneca Valley virus in piglets from China, 2016. Virol J 13:173
Reddy PS, Burroughs KD, Hales LM, Ganesh S, Jones BH, Idamakanti N, Hay C, Li SS, Skele KL, Vasko AJ, Yang J, Watkins DN, Rudin CM, Hallenbeck PL (2007) Seneca Valley virus, a systemically deliverable oncolytic picornavirus, and the treatment of neuroendocrine cancers. J Natl Cancer Inst 99:1623–1633
Saeng-Chuto K, Rodtian P, Temeeyasen G, Wegner M, Nilubol D (2017) The first detection of Senecavirus A in pigs in Thailand, 2016. Transbound Emerg Dis. https://doi.org/10.1111/tbed.12654
Sun D, Vannucci F, Knutson TP, Corzo C, Marthaler DG (2017) Emergence and whole-genome sequence of Senecavirus A in Colombia. Transbound Emerg Dis. https://doi.org/10.1111/tbed.12669
Wang H, Li C, Zhao B, Yuan T, Yang D, Zhou G, Yu L (2017) Complete genome sequence and phylogenetic analysis of Senecavirus A isolated in Northeast China in 2016. Arch Virol 65:614–617
Wu Q, Zhao X, Bai Y, Sun B, Xie Q, Ma J (2016) The first identification and complete genome of Senecavirus A affecting pig with idiopathic vesicular disease in China. Transbound Emerg Dis. https://doi.org/10.1111/tbed.12557
Zhao X, Wu Q, Bai Y, Chen G, Zhou L, Wu Z, Li Y, Zhou W, Yang H, Ma J (2017) Phylogenetic and genome analysis of seven Senecavirus A isolates in China. Transbound Emerg Dis. https://doi.org/10.1111/tbed.12619
Zheng H, Lian K, Yang F, Jin Y, Zhu Z, Guo J, Cao W, Liu H, He J, Zhang K, Li D, Liu X (2015) Cross-protective efficacy of engineering serotype A foot-and-mouth disease virus vaccine against the two pandemic strains in swine. Vaccine 33:5772–5778
Zhu Z, Yang F, Chen P, Liu H, Cao W, Zhang K, Liu X, Zheng H (2017) Emergence of novel Seneca Valley virus strains in China, 2017. Transbound Emerg Dis 64:1024–1029
Acknowledgements
This work was supported by grants from the National Natural Sciences Foundation of China (No. U1501213), the Key Development and Research Foundation of Yunnan (2018BB004) and the Project Supported by National Science and Technology Ministry (2015BAD12B04).
Author information
Authors and Affiliations
Contributions
PC, FY, ZZ and HZ designed the research, PC, FY, ZZ, WC and HL performed the experiments. KZ and XL provided experiment support. PC, ZZ and HZ wrote the manuscript. All authors have read and approved the final manuscript for submission.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and Human Rights Statement
This article does not contain any studies with human or animal subjects performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, P., Yang, F., Cao, W. et al. The Distribution of Different Clades of Seneca Valley Viruses in Guangdong Province, China. Virol. Sin. 33, 394–401 (2018). https://doi.org/10.1007/s12250-018-0056-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-018-0056-8