Abstract
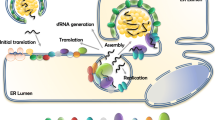
Flaviviruses are positive-sense RNA viruses, and many are important human pathogens. Nonstructural protein 2B and 3 of the flaviviruses (NS2BNS3) form an endoplasmic reticulum (ER) membrane-associated hetero-dimeric complex through the NS2B transmembrane region. The NS2BNS3 complex is multifunctional. The N-terminal region of NS3, and its cofactor NS2B fold into a protease that is responsible for viral polyprotein processing, and the C-terminal domain of NS3 possesses NTPase/RNA helicase activities and is involved in viral RNA replication and virus particle formation. In addition, NS2BNS3 complex has also been shown to modulate viral pathogenesis and the host immune response. Because of the essential functions that the NS2BNS3 complex plays in the flavivirus life cycle, it is an attractive target for antiviral development. This review focuses on the recent biochemical and structural advances of NS2BNS3 and provides a brief update on the current status of drug development targeting this viral protein complex.
Article PDF
Similar content being viewed by others
References
Aguirre S, Maestre A M, Pagni S, Patel J R, Savage T, Gutman D, Maringer K, Bernal-Rubio D, Shabman R S, Simon V, Rodriguez-Madoz J R, Mulder L C, Barber G N, Fernandez-Sesma A. 2012. DENV inhibits type I IFN production in infected cells by cleaving human STING. PLoS Pathog, 8: e1002934.
Aleshin A E, Shiryaev S A, Strongin A Y, Liddington R C. 2007. Structural evidence for regulation and specificity of flaviviral proteases and evolution of the Flaviviridae fold. Protein Sci, 16: 795–806.
Alvarez D E, Lodeiro M F, Filomatori C V, Fucito S, Mondotte J A, Gamarnik A V. 2006. Structural and functional analysis of dengue virus RNA. Novartis Found Symp, 277: 120–132; discussion 132–125, 251–123.
Appleby T C, Anderson R, Fedorova O, Pyle A M, Wang R, Liu X, Brendza K M, Somoza J R. 2010. Visualizing ATP-dependent RNA translocation by the NS3 helicase from HCV. J Mol Biol, 405: 1139–1153.
Arakaki T L, Fang N X, Fairlie D P, Young P R, Martin J L. 2002. Catalytically active Dengue virus NS3 protease forms aggregates that are separable by size exclusion chromatography. Protein Expr Purif, 25: 241–247.
Assenberg R, Mastrangelo E, Walter T S, Verma A, Milani M, Owens R J, Stuart D I, Grimes J M, Mancini E J. 2009. Crystal structure of a novel conformational state of the flavivirus NS3 protein: implications for polyprotein processing and viral replication. J Virol, 83: 12895–12906.
Balistreri G, Caldentey J, Kaariainen L, Ahola T. 2007. Enzymatic defects of the nsP2 proteins of Semliki Forest virus temperature-sensitive mutants. J Virol, 81: 2849–2860.
Benarroch D, Selisko B, Locatelli G A, Maga G, Romette J L, Canard B. 2004. The RNA helicase, nucleotide 5′-triphosphatase, and RNA 5′-triphosphatase activities of Dengue virus protein NS3 are Mg2+-dependent and require a functional Walker B motif in the helicase catalytic core. Virology, 328: 208–218.
Bollati M, Alvarez K, Assenberg R, Baronti C, Canard B, Cook S, Coutard B, Decroly E, de Lamballerie X, Gould E A, Grard G, Grimes J M, Hilgenfeld R, Jansson A M, Malet H, Mancini E J, Mastrangelo E, Mattevi A, Milani M, Moureau G, Neyts J, Owens R J, Ren J, Selisko B, Speroni S, Steuber H, Stuart D I, Unge T, Bolognesi M. 2010. Structure and functionality in flavivirus NS-proteins: perspectives for drug design. Antiviral Res, 87: 125–148.
Brooks A J, Johansson M, John A V, Xu Y, Jans D A, Vasudevan S G. 2002. The interdomain region of dengue NS5 protein that binds to the viral helicase NS3 contains independently functional importin beta 1 and importin alpha/beta-recognized nuclear localization signals. J Biol Chem, 277: 36399–36407.
Buttner K, Nehring S, Hopfner K P. 2007. Structural basis for DNA duplex separation by a superfamily-2 helicase. Nat Struct Mol Biol, 14: 647–652.
Byrd C M, Grosenbach D W, Berhanu A, Dai D, Jones K F, Cardwell K B, Schneider C, Yang G, Tyavanagimatt S, Harver C, Wineinger K A, Page J, Stavale E, Stone M A, Fuller K P, Lovejoy C, Leeds J M, Hruby D E, Jordan R. 2013. Novel benzoxazole inhibitor of dengue virus replication that targets the NS3 helicase. Antimicrob Agents Chemother, 57: 1902–1912.
Chambers T J, Hahn C S, Galler R, Rice C M. 1990. Flavivirus genome organization, expression, and replication. Annu Rev Microbiol, 44: 649–688.
Chandramouli S, Joseph J S, Daudenarde S, Gatchalian J, Cornillez-Ty C, Kuhn P. 2010. Serotype-specific structural differences in the protease-cofactor complexes of the dengue virus family. J Virol, 84: 3059–3067.
Chappell K J, Stoermer M J, Fairlie D P, Young P R. 2008. Mutagenesis of the West Nile virus NS2B cofactor domain reveals two regions essential for protease activity. J Gen Virol, 89: 1010–1014.
Chernov A V, Shiryaev S A, Aleshin A E, Ratnikov B I, Smith J W, Liddington R C, Strongin A Y. 2008. The two-component NS2B-NS3 proteinase represses DNA unwinding activity of the West Nile virus NS3 helicase. J Biol Chem, 283: 17270–17278.
Choksupmanee O, Hodge K, Katzenmeier G, Chimnaronk S. 2012. Structural platform for the autolytic activity of an intact NS2B-NS3 protease complex from dengue virus. Biochemistry, 51: 2840–2851.
Clum S, Ebner K E, Padmanabhan R. 1997. Cotranslational membrane insertion of the serine proteinase precursor NS2B-NS3(Pro) of dengue virus type 2 is required for efficient in vitro processing and is mediated through the hydrophobic regions of NS2B. J Biol Chem, 272: 30715–30723.
Decroly E, Ferron F, Lescar J, Canard B. 2012. Conventional and unconventional mechanisms for capping viral mRNA. Nat Rev Microbiol, 10: 51–65.
Dumont S, Cheng W, Serebrov V, Beran R K, Tinoco I, Jr., Pyle A M, Bustamante C. 2006. RNA translocation and unwinding mechanism of HCV NS3 helicase and its coordination by ATP. Nature, 439: 105–108.
Egloff M P, Benarroch D, Selisko B, Romette J L, Canard B. 2002. An RNA cap (nucleoside-2′-O-)-methyltransferase in the flavivirus RNA polymerase NS5: crystal structure and functional characterization. EMBO J, 21: 2757–2768.
Erbel P, Schiering N, D’Arcy A, Renatus M, Kroemer M, Lim S P, Yin Z, Keller T H, Vasudevan S G, Hommel U. 2006. Structural basis for the activation of flaviviral NS3 proteases from dengue and West Nile virus. Nat Struct Mol Biol, 13: 372–373.
Fairman-Williams M E, Guenther U P, Jankowsky E. 2010. SF1 and SF2 helicases: family matters. Curr Opin Struct Biol, 20: 313–324.
Falgout B, Pethel M, Zhang Y M, Lai C J. 1991. Both nonstructural proteins NS2B and NS3 are required for the proteolytic processing of dengue virus nonstructural proteins. J Virol, 65: 2467–2475.
Filomatori C V, Lodeiro M F, Alvarez D E, Samsa M M, Pietrasanta L, Gamarnik A V. 2006. A 5′ RNA element promotes dengue virus RNA synthesis on a circular genome. Genes Dev, 20: 2238–2249.
Frick D N, Rypma R S, Lam A M, Gu B. 2004. The nonstructural protein 3 protease/helicase requires an intact protease domain to unwind duplex RNA efficiently. J Biol Chem, 279: 1269–1280.
Gebhard L G, Kaufman S B, Gamarnik A V. 2012. Novel ATP-independent RNA annealing activity of the dengue virus NS3 helicase. PLoS One, 7: e36244.
Gorbalenya A E, Koonin E V. 1993. Helicases: amino acid sequence comparisons and structure-function relationships. Curr Opin Struc Biol, 3: 419–429.
Gorbalenya A E, Donchenko A P, Koonin E V, Blinov V M. 1989. N-terminal domains of putative helicases of flavi- and pestiviruses may be serine proteases. Nucleic Acids Res, 17: 3889–3897.
Gouvea I E, Izidoro M A, Judice W A, Cezari M H, Caliendo G, Santagada V, dos Santos C N, Queiroz M H, Juliano M A, Young P R, Fairlie D P, Juliano L. 2007. Substrate specificity of recombinant dengue 2 virus NS2B-NS3 protease: influence of natural and unnatural basic amino acids on hydrolysis of synthetic fluorescent substrates. Arch Biochem Biophys, 457: 187–196.
Gu M, Rice C M. 2010. Three conformational snapshots of the hepatitis C virus NS3 helicase reveal a ratchet translocation mechanism. Proc Natl Acad Sci USA, 107: 521–528.
Huang Q, Li Q, Joy J, Chen A S, Ruiz-Carrillo D, Hill J, Lescar J, Kang C. 2013. Lyso-myristoyl phosphatidylcholine micelles sustain the activity of Dengue non-structural (NS) protein 3 protease domain fused with the full-length NS2B. Protein Expr Purif, 92: 156–162.
Ishikawa H, Barber G N. 2008. STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature, 455: 674–678.
Jan L R, Yang C S, Trent D W, Falgout B, Lai C J. 1995. Processing of Japanese encephalitis virus non-structural proteins: NS2B-NS3 complex and heterologous proteases. J Gen Virol, 76: 573–580.
Keller T H, Chen Y L, Knox J E, Lim S P, Ma N L, Patel S J, Sampath A, Wang Q Y, Yin Z, Vasudevan S G. 2006. Finding new medicines for flaviviral targets. Novartis Found Symp, 277: 102–114; discussion 114–109, 251–103.
Khadka S, Vangeloff A D, Zhang C, Siddavatam P, Heaton N S, Wang L, Sengupta R, Sahasrabudhe S, Randall G, Gribskov M, Kuhn R J, Perera R, LaCount D J. 2011. A physical interaction network of dengue virus and human proteins. Mol Cell Proteomics, 10: M111 012187.
Kim J L, Morgenstern K A, Lin C, Fox T, Dwyer M D, Landro J A, Chambers S P, Markland W, Lepre C A, O’Malley E T, Harbeson S L, Rice C M, Murcko M A, Caron P R, Thomson J A. 1996. Crystal structure of the hepatitis C virus NS3 protease domain complexed with a synthetic NS4A cofactor peptide. Cell, 87: 343–355.
Krishnan M N, Garcia-Blanco M A. 2014. Targeting host factors to treat west nile and dengue viral infections. Viruses, 6: 683–708.
Le Breton M, Meyniel-Schicklin L, Deloire A, Coutard B, Canard B, de Lamballerie X, Andre P, Rabourdin-Combe C, Lotteau V, Davoust N. 2011. Flavivirus NS3 and NS5 proteins interaction network: a high-throughput yeast two-hybrid screen. BMC Microbiol, 11: 234.
Lescar J, Luo D, Xu T, Sampath A, Lim S P, Canard B, Vasudevan S G. 2008. Towards the design of antiviral inhibitors against flaviviruses: the case for the multifunctional NS3 protein from Dengue virus as a target. Antiviral Res, 80: 94–101.
Leung D, Schroder K, White H, Fang N X, Stoermer M J, Abbenante G, Martin J L, Young P R, Fairlie D P. 2001. Activity of recombinant dengue 2 virus NS3 protease in the presence of a truncated NS2B co-factor, small peptide substrates, and inhibitors. J Biol Chem, 276: 45762–45771.
Li H, Clum S, You S, Ebner K E, Padmanabhan R. 1999. The serine protease and RNA-stimulated nucleoside triphosphatase and RNA helicase functional domains of dengue virus type 2 NS3 converge within a region of 20 amino acids. J Virol, 73: 3108–3116.
Li J, Lim S P, Beer D, Patel V, Wen D, Tumanut C, Tully D C, Williams J A, Jiricek J, Priestle J P, Harris J L, Vasudevan S G. 2005. Functional profiling of recombinant NS3 proteases from all four serotypes of dengue virus using tetrapeptide and octapeptide substrate libraries. J Biol Chem, 280: 28766–28774.
Li K, Frankowski K J, Hanson A M, Ndjomou J, Shanahan M A, Mukherjee S, Kolli R, Shadrick W R, Sweeney N L, Belon C A, Neuenswander B, Ferguson J, Aubé J, Schoenen F J, Blagg B S J, Frick D N. 2010. Hepatitis C Virus NS3 Helicase Inhibitor Discovery. In: Probe Reports from the NIH Molecular Libraries Program. National Center for Biotechnology Information, Bethesda (MD).
Lim S P, Wang Q Y, Noble C G, Chen Y L, Dong H, Zou B, Yokokawa F, Nilar S, Smith P, Beer D, Lescar J, Shi P Y. 2013. Ten years of dengue drug discovery: progress and prospects. Antiviral Res, 100: 500–519.
Lin C W, Lin K H, Lyu P C, Chen W J. 2006. Japanese encephalitis virus NS2B-NS3 protease binding to phage-displayed human brain proteins with the domain of trypsin inhibitor and basic region leucine zipper. Virus Res, 116: 106–113.
Lindenbach B D, Thiel H J, Rice C M. 2007. Flaviviridae: the viruses and their replication. In: Knipe D M, Howley P M. Eds. Lippincott-Raven Publishers, Philadelphia. pp1101–1152.
Linder P, Jankowsky E. 2011. From unwinding to clamping — the DEAD box RNA helicase family. Nat Rev Mol Cell Biol, 12: 505–516.
Luo D, Lim S P, Lescar J. 2012. The Flavivirus NS3 Protein: Structure and Functions. In: Shi P Y. Eds. Molecular Virology and Control of Flaviviruses. Caister Academic Press, Norfolk. pp77–100.
Luo D, Xu T, Hunke C, Gruber G, Vasudevan S G, Lescar J. 2008. Crystal structure of the NS3 protease-helicase from dengue virus. J Virol, 82: 173–183.
Luo D, Wei N, Doan D N, Paradkar P N, Chong Y, Davidson A D, Kotaka M, Lescar J, Vasudevan S G. 2010. Flexibility between the protease and helicase domains of the dengue virus NS3 protein conferred by the linker region and its functional implications. J Biol Chem, 285: 18817–18827.
Luo D, Xu T, Watson R P, Scherer-Becker D, Sampath A, Jahnke W, Yeong S S, Wang C H, Lim S P, Strongin A, Vasudevan S G, Lescar J. 2008. Insights into RNA unwinding and ATP hydrolysis by the flavivirus NS3 protein. EMBO J, 27: 3209–3219.
Mackenzie J. 2005. Wrapping things up about virus RNA replication. Traffic, 6: 967–977.
Maga G, Gemma S, Fattorusso C, Locatelli G A, Butini S, Persico M, Kukreja G, Romano M P, Chiasserini L, Savini L, Novellino E, Nacci V, Spadari S, Campiani G. 2005. Specific targeting of hepatitis C virus NS3 RNA helicase. Discovery of the potent and selective competitive nucleotide-mimicking inhibitor QU663. Biochemistry, 44: 9637–9644.
Malet H, Masse N, Selisko B, Romette J L, Alvarez K, Guillemot J C, Tolou H, Yap T L, Vasudevan S, Lescar J, Canard B. 2008. The flavivirus polymerase as a target for drug discovery. Antiviral Res, 80: 23–35.
Malet H, Egloff M P, Selisko B, Butcher R E, Wright P J, Roberts M, Gruez A, Sulzenbacher G, Vonrhein C, Bricogne G, Mackenzie J M, Khromykh A A, Davidson A D, Canard B. 2007. Crystal structure of the RNA polymerase domain of the West Nile virus non-structural protein 5. J Biol Chem, 282: 10678–10689.
Mancini E J, Assenberg R, Verma A, Walter T S, Tuma R, Grimes J M, Owens R J, Stuart D I. 2007. Structure of the Murray Valley encephalitis virus RNA helicase at 1.9 Angstrom resolution. Protein Sci, 16: 2294–2300.
Markoff L. 2003. 5′- and 3′-noncoding regions in flavivirus RNA. Adv Virus Res, 59: 177–228.
Mastrangelo E, Pezzullo M, De Burghgraeve T, Kaptein S, Pastorino B, Dallmeier K, de Lamballerie X, Neyts J, Hanson A M, Frick D N, Bolognesi M, Milani M. 2012. Ivermectin is a potent inhibitor of flavivirus replication specifically targeting NS3 helicase activity: new prospects for an old drug. J Antimicrob Chemother, 67: 1884–1894.
Mastrangelo E, Milani M, Bollati M, Selisko B, Peyrane F, Pandini V, Sorrentino G, Canard B, Konarev P V, Svergun D I, de Lamballerie X, Coutard B, Khromykh A A, Bolognesi M. 2007. Crystal structure and activity of Kunjin virus NS3 helicase; protease and helicase domain assembly in the full length NS3 protein. J Mol Biol, 372: 444–455.
Matusan A E, Pryor M J, Davidson A D, Wright P J. 2001. Mutagenesis of the Dengue virus type 2 NS3 protein within and outside helicase motifs: effects on enzyme activity and virus replication. J Virol, 75: 9633–9643.
Miorin L, Romero-Brey I, Maiuri P, Hoppe S, Krijnse-Locker J, Bartenschlager R, Marcello A. 2013. Three-dimensional architecture of tick-borne encephalitis virus replication sites and trafficking of the replicated RNA. J Virol, 87: 6469–6481.
Murray C L, Jones C T, Rice C M. 2008. Architects of assembly: roles of Flaviviridae non-structural proteins in virion morphogenesis. Nat Rev Microbiol, 6: 699–708.
Myong S, Bruno M M, Pyle A M, Ha T. 2007. Spring-loaded mechanism of DNA unwinding by hepatitis C virus NS3 helicase. Science, 317: 513–516.
Nall T A, Chappell K J, Stoermer M J, Fang N X, Tyndall J D, Young P R, Fairlie D P. 2004. Enzymatic characterization and homology model of a catalytically active recombinant West Nile virus NS3 protease. J Biol Chem, 279: 48535–48542.
Niyomrattanakit P, Yahorava S, Mutule I, Mutulis F, Petrovska R, Prusis P, Katzenmeier G, Wikberg J E. 2006. Probing the substrate specificity of the dengue virus type 2 NS3 serine protease by using internally quenched fluorescent peptides. Biochem J, 397: 203–211.
Noble C G, Seh C C, Chao A T, Shi P Y. 2012. Ligand-bound structures of the dengue virus protease reveal the active conformation. J Virol, 86: 438–446.
Patkar C G, Kuhn R J. 2008. Yellow Fever virus NS3 plays an essential role in virus assembly independent of its known enzymatic functions. J Virol, 82: 3342–3352.
Paul D, Bartenschlager R. 2013. Architecture and biogenesis of plus-strand RNA virus replication factories. World J Virol, 2: 32–48.
Polacek C, Foley J E, Harris E. 2009. Conformational changes in the solution structure of the dengue virus 5′ end in the presence and absence of the 3′ untranslated region. J Virol, 83: 1161–1166.
Pyle A M. 2008. Translocation and unwinding mechanisms of RNA and DNA helicases. Annu Rev Biophys, 37: 317–336.
Ramanathan M P, Chambers J A, Pankhong P, Chattergoon M, Attatippaholkun W, Dang K, Shah N, Weiner D B. 2006. Host cell killing by the West Nile Virus NS2B-NS3 proteolytic complex: NS3 alone is sufficient to recruit caspase-8-based apoptotic pathway. Virology, 345: 56–72.
Rao S T, Rossmann M G. 1973. Comparison of super-secondary structures in proteins. J Mol Biol, 76: 241–256.
Ray D, Shah A, Tilgner M, Guo Y, Zhao Y, Dong H, Deas T S, Zhou Y, Li H, Shi P Y. 2006. West Nile virus 5′-cap structure is formed by sequential guanine N-7 and ribose 2′-O methylations by nonstructural protein 5. J Virol, 80: 8362–8370.
Rendi-Wagner P. 2008. Advances in vaccination against tick-borne encephalitis. Expert Rev Vaccines, 7: 589–596.
Robin G, Chappell K, Stoermer M J, Hu S H, Young P R, Fairlie D P, Martin J L. 2009. Structure of West Nile virus NS3 protease: ligand stabilization of the catalytic conformation. J Mol Biol, 385: 1568–1577.
Saalau-Bethell S M, Woodhead A J, Chessari G, Carr M G, Coyle J, Graham B, Hiscock S D, Murray C W, Pathuri P, Rich S J, Richardson C J, Williams P A, Jhoti H. 2012. Discovery of an allosteric mechanism for the regulation of HCV NS3 protein function. Nat Chem Biol, 8: 920–925.
Sabchareon A, Wallace D, Sirivichayakul C, Limkittikul K, Chanthavanich P, Suvannadabba S, Jiwariyavej V, Dulyachai W, Pengsaa K, Wartel T A, Moureau A, Saville M, Bouckenooghe A, Viviani S, Tornieporth N G, Lang J. 2012. Protective efficacy of the recombinant, live-attenuated, CYD tetravalent dengue vaccine in Thai schoolchildren: a randomised, controlled phase 2b trial. Lancet, 380: 1559–1567.
Salam K A, Akimitsu N. 2013. Hepatitis C virus NS3 inhibitors: current and future perspectives. Biomed Res Int, 2013: 467869.
Sampath A, Padmanabhan R. 2009. Molecular targets for flavivirus drug discovery. Antiviral Res, 81: 6–15.
Schiering N, D’Arcy A, Villard F, Simic O, Kamke M, Monnet G, Hassiepen U, Svergun D I, Pulfer R, Eder J, Raman P, Bodendorf U. 2011. A macrocyclic HCV NS3/4A protease inhibitor interacts with protease and helicase residues in the complex with its full-length target. Proc Natl Acad Sci USA, 108: 21052–21056.
Shadrick W R, Ndjomou J, Kolli R, Mukherjee S, Hanson A M, Frick D N. 2013. Discovering new medicines targeting helicases: challenges and recent progress. J Biomol Screen, 18: 761–781.
Shiryaev S A, Kozlov I A, Ratnikov B I, Smith J W, Lebl M, Strongin A Y. 2007. Cleavage preference distinguishes the two-component NS2B-NS3 serine proteinases of Dengue and West Nile viruses. Biochem J, 401: 743–752.
Speroni S, De Colibus L, Mastrangelo E, Gould E, Coutard B, Forrester N L, Blanc S, Canard B, Mattevi A. 2008. Structure and biochemical analysis of Kokobera virus helicase. Proteins, 70: 1120–1123.
Steimer L, Klostermeier D. 2012. RNA helicases in infection and disease. RNA Biol, 9.
Story R M, Steitz T A. 1992. Structure of the recA protein-ADP complex. Nature, 355: 374–376.
Suthar M S, Aguirre S, Fernandez-Sesma A. 2013. Innate immune sensing of flaviviruses. PLoS Pathog, 9: e1003541.
Tomei L, Failla C, Vitale R L, Bianchi E, De Francesco R. 1996. A central hydrophobic domain of the hepatitis C virus NS4A protein is necessary and sufficient for the activation of the NS3 protease. J Gen Virol, 77: 1065–1070.
Urbani A, Bazzo R, Nardi M C, Cicero D O, De Francesco R, Steinkuhler C, Barbato G. 1998. The metal binding site of the hepatitis C virus NS3 protease. A spectroscopic investigation. J Biol Chem, 273: 18760–18769.
Verma R, Khanna P, Chawla S. 2013. Yellow fever vaccine: An effective vaccine for travelers. Hum Vaccin Immunother, 10: 126–128.
Villordo S M, Gamarnik A V. 2009. Genome cyclization as strategy for flavivirus RNA replication. Virus Res, 139: 230–239.
Welsch S, Miller S, Romero-Brey I, Merz A, Bleck C K, Walther P, Fuller S D, Antony C, Krijnse-Locker J, Bartenschlager R. 2009. Composition and three-dimensional architecture of the dengue virus replication and assembly sites. Cell Host Microbe, 5: 365–375.
WHO. 2009. Fact sheet: Dengue and dengue haemorrhagic fever. http://www.who.int/csr/disease/dengue/en/
Wu J, Bera A K, Kuhn R J, Smith J L. 2005. Structure of the Flavivirus helicase: implications for catalytic activity, protein interactions, and proteolytic processing. J Virol, 79: 10268–10277.
Xu T, Sampath A, Chao A, Wen D, Nanao M, Chene P, Vasudevan S G, Lescar J. 2005. Structure of the Dengue virus helicase/nucleoside triphosphatase catalytic domain at a resolution of 2.4 A. J Virol, 79: 10278–10288.
Xu T, Sampath A, Chao A, Wen D, Nanao M, Luo D, Chene P, Vasudevan S G, Lescar J. 2006. Towards the design of flavivirus helicase/NTPase inhibitors: crystallographic and mutagenesis studies of the dengue virus NS3 helicase catalytic domain. Novartis Found Symp, 277: 87–97; discussion 97–101, 251–103.
Yamashita T, Unno H, Mori Y, Tani H, Moriishi K, Takamizawa A, Agoh M, Tsukihara T, Matsuura Y. 2008. Crystal structure of the catalytic domain of Japanese encephalitis virus NS3 helicase/nucleoside triphosphatase at a resolution of 1.8 A. Virology, 373: 426–436.
Yao N, Reichert P, Taremi S S, Prosise W W, Weber P C. 1999. Molecular views of viral polyprotein processing revealed by the crystal structure of the hepatitis C virus bifunctional protease-helicase. Structure, 7: 1353–1363.
Yon C, Teramoto T, Mueller N, Phelan J, Ganesh V K, Murthy K H, Padmanabhan R. 2005. Modulation of the nucleoside triphosphatase/RNA helicase and 5′-RNA triphosphatase activities of Dengue virus type 2 nonstructural protein 3 (NS3) by interaction with NS5, the RNA-dependent RNA polymerase. J Biol Chem, 280: 27412–27419.
Yu I M, Zhang W, Holdaway H A, Li L, Kostyuchenko V A, Chipman P R, Kuhn R J, Rossmann M G, Chen J. 2008. Structure of the immature dengue virus at low pH primes proteolytic maturation. Science, 319: 1834–1837.
Yu L, Nomaguchi M, Padmanabhan R, Markoff L. 2008. Specific requirements for elements of the 5′ and 3′ terminal regions in flavivirus RNA synthesis and viral replication. Virology, 374: 170–185.
Yun S I, and Lee Y M. 2013. Japanese Encephalitis: The virus and vaccines. Hum Vaccin Immunother, 10.
Yusof R, Clum S, Wetzel M, Murthy H M, Padmanabhan R. 2000. Purified NS2B/NS3 serine protease of dengue virus type 2 exhibits cofactor NS2B dependence for cleavage of substrates with dibasic amino acids in vitro. J Biol Chem, 275: 9963–9969.
Zhang L, Mohan P M, Padmanabhan R. 1992. Processing and localization of Dengue virus type 2 polyprotein precursor NS3-NS4A-NS4B-NS5. J Virol, 66: 7549–7554.
Zhong B, Yang Y, Li S, Wang Y Y, Li Y, Diao F, Lei C, He X, Zhang L, Tien P, Shu H B. 2008. The adaptor protein MITA links virus-sensing receptors to IRF3 transcription factor activation. Immunity, 29: 538–550.
Zou G, Chen Y L, Dong H, Lim C C, Yap L J, Yau Y H, Shochat S G, Lescar J, Shi P Y. 2011. Functional analysis of two cavities in flavivirus NS5 polymerase. J Biol Chem, 286: 14362–14372.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, K., Phoo, W.W. & Luo, D. Functional interplay among the flavivirus NS3 protease, helicase, and cofactors. Virol. Sin. 29, 74–85 (2014). https://doi.org/10.1007/s12250-014-3438-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12250-014-3438-6