Abstract
Purpose
In this work, we investigate the effect of adding polyvinylpyrrolidone (PVP K30) to the binary solid dispersion (indomethacin/kaolin) on the formation of physically stable amorphous drug. This aims to profit more effectively from the therapeutic effect of kaolin in the solid dosage forms of indomethacin.
Methods
Binary mixtures (indomethacin/kaolin) were ball milled at room temperature (≈ 25 °C) in presence of PVP K30 at different weight ratios (w/w). The characterization of the obtained materials was carried out using X-ray diffraction (XRD), infrared spectroscopy (FTIR), scanning electronic microscopy (SEM), high-performance liquid chromatography (HPLC), differential scanning calorimetry (DSC), and 13C MAS NMR spectroscopy.
Results
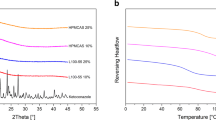
Results have shown that indomethacin (IND) interacted with kaolin and PVP K30 in solid state via hydrogen bonds without any polymorphic transformations or chemical degradation. The PVP seems to play a role of linker between drug and kaolin leading to physical stability enhancement of amorphous IND even under high stress conditions (RH = 75% and T = 40 °C for 3 months). Such ternary system (IND/kaolin/PVP) has shown a considerable improvement of drug solubility at T = 37 ± 0.5 °C and pH = 7.0 compared to the binary solid dispersion (IND/kaolin).
Conclusions
The addition of PVP to the solid dispersion (IND/kaolin) was advantageous not only in terms of physical stabilization of the amorphous IND but also made it possible to overcome the solubility challenges associated with the presence of kaolin.











Similar content being viewed by others
References
Knapik-Kowalczuk J, Tu W, Chmiel K, Rams-Baron M, Paluch M. Co-Stabilization of amorphous pharmaceuticals—the case of nifedipine and nimodipine. Mol Pharm. 2018;15(6):2455–65.
Karagianni A, Kachrimanis K, Nikolakakis I. Co-amorphous solid dispersions for solubility and absorption improvement of drugs: composition, preparation, characterization and formulations for oral delivery. Pharmaceutics. 2018;10(3):98.
Lucas S. The pharmacology of indomethacin. J Headache Pain. 2016;56(2):436–46.
El-Badry M, Fetih G, Fathy M. Improvement of solubility and dissolution rate of indomethacin by solid dispersions in Gelucire 50/13 and PEG4000. Saudi Pharm J. 2009;17(3):217–25.
Takeuchi H, Nagira S, Yamamoto H, Kawashima Y. Solid dispersion particles of amorphous indomethacin with fine porous silica particles by using spray-drying method. Int J Pharm. 2005;293(1–2):155–64.
Fini A, Cavallari C, Ospitali F. Raman and thermal analysis of indomethacin/PVP solid dispersion enteric microparticles. Eur J Pharm Biopharm. 2008;70(1):409–20.
Awad ME, López-Galindo A, Setti M, El-Rahmany MM, Iborra CV. Kaolinite in pharmaceutics and biomedicine. Int J Pharm. 2017;533(1):34–48.
Carretero MI. Clay minerals and their beneficial effects upon human health. A review Appl Clay Sci. 2002;21(3–4):155–63.
Mallick S, Pattnaik S, Swain K, De PK, Saha A, Ghoshal G, Mondal A. Formation of physically stable amorphous phase of ibuprofen by solid state milling with kaolin. Eur J Pharm Biopharm. 2008;68(2):346–51.
Bahl D, Hudak J, Bogner RH. Comparison of the ability of various pharmaceutical silicates to amorphize and enhance dissolution of indomethacin upon co-grinding. Pharm Dev Technol. 2008;13(3):255–69.
Onyekweli AO, Usifoh CO, Okunrobo LO, Zuofa JD. Adsorptive property of kaolin in some drug formulations. Trop J Pharm Res. 2003;2(1):155–9.
Hassan S, Ibrahim J. Adsorption of some drugs onto surface of Iraqi kaolin clay. Pak J Chem. 2011;1(3):132–7.
Aleanizy FS, Alqahtani F, Al Gohary O, El Tahir E, Al SR. Determination and characterization of metronidazole–kaolin interaction. Saudi Pharm J. 2015;23(2):167–76.
Mura P, Maestrelli F, Cirri M. Ternary systems of naproxen with hydroxypropyl-β-cyclodextrin and amino acids. Int J Pharm. 2003;260(2):293–302.
Viseras C, Aguzzi C, Cerezo P, Bedmar MC. Biopolymer–clay nanocomposites for controlled drug delivery. Mater Sci Technol. 2008;24(9):1020–6.
Giri TK, Badwaik H, Alexander A, Tripathi DK. Solubility enhancement of ibuprofen in the presence of hydrophilic polymer and surfactant. Int J Appl Biol Pharm Tech. 2010;1(2):793–800.
Bejaoui M, Galai H, Amara AB, Rhaiem HB. Formation of water soluble and stable amorphous ternary system: ibuprofen/β-cyclodextrin/PVP. Glass Phys Chem. 2019;45(6):580–8.
Watanabe T, Ohno I, Wakiyama N, Kusai A, Senna M. Stabilization of amorphous indomethacin by co-grinding in a ternary mixture. Int J Pharm. 2002;241(1):103–11.
Prasad D, Chauhan H, Atef E. Amorphous stabilization and dissolution enhancement of amorphous ternary solid dispersions: combination of polymers showing drug–polymer interaction for synergistic effects. J Pharm Sci. 2014;103(11):3511–23.
O’brien M, McCauley J, Cohen E. Analytical profiles of drug substances. Indomethacin. San Diego: Academic Press Inc. 1984:211–38.
Desprez S, Descamps M. Transformations of glassy indomethacin induced by ball-milling. J Non Cryst Solids. 2006;352(42–49):4480–5.
Gupta MK, Vanwert A, Bogner RH. Formation of physically stable amorphous drugs by milling with Neusilin. J Pharm Sci. 2003;92(3):536–51.
Otsuka M, Matsumoto T, Kaneniwa N. Effect of environmental temperature on polymorphic solid-state transformation of indomethacin during grinding. Chem Pharm Bull. 1986;34(4):1784–93.
Bhugra C, Shmeis R, Krill SL, Pikal MJ. Prediction of onset of crystallization from experimental relaxation times. II. Comparison between predicted and experimental onset times. J Pharm Sci. 2008; 97(1):455–72.
Imaizumi H, Nambu N, Nagai T. Stability and several physical properties of amorphous and crystalline forms of indomethacin. Chem Pharm Bull. 1980;28(9):2565–9.
Tong P, Taylor LS, Zografi G. Influence of alkali metal counterions on the glass transition temperature of amorphous indomethacin salts. Pharm Res. 2002;19(5):649–54.
Adrjanowicz K, Kaminski K, Grzybowska K, Hawelek L, Paluch M, Gruszka I, Zakowiecki D, Sawicki W, Lepek P, Kamysz W, Guzik L. Effect of cryogrinding on chemical stability of the sparingly water-soluble drug furosemide. Pharm Res. 2011;28(12):3220–36.
Masuda K, Tabata S, Kono H, Sakata Y, Hayase T, Yonemochi E, Terada K. Solid-state 13C NMR study of indomethacin polymorphism. Int J Pharm. 2006;318(1–2):146–53.
Watanabe T, Wakiyama N, Usui F, Ikeda M, Isobe T, Senna M. Stability of amorphous indomethacin compounded with silica. Int J Pharm. 2001;226(1–2):81–91.
Yuan X, Xiang TX, Anderson BD, Munson EJ. Hydrogen bonding interactions in amorphous indomethacin and its amorphous solid dispersions with poly (vinylpyrrolidone) and poly (vinylpyrrolidone-co-vinyl acetate) studied using 13C solid-state NMR. Mol Pharm. 2015;12(12):4518–28.
Gupta P, Thilagavathi R, Chakraborti AK, Bansal AK. Role of molecular interaction in stability of celecoxib− PVP amorphous systems. Mol Pharm. 2005;2(5):384–91.
Khougaz K, Clas SD. Crystallization inhibition in solid dispersions of MK-0591 and poly (vinylpyrrolidone) polymers. J Pharm Sci. 2000;89(10):1325–34.
Hancock BC, Zografi G. The relationship between the glass transition temperature and the water content of amorphous pharmaceutical solids. Pharm Res. 1994;11(4):471–7.
Xie T, Taylor LS. Effect of temperature and moisture on the physical stability of binary and ternary amorphous solid dispersions of celecoxib. J Pharm Sci. 2017;106(1):100–10.
Li Y, Rantanen J, Yang M, Bohr A. Molecular structure and impact of amorphization strategies on intrinsic dissolution of spray dried indomethacin. Eur J Pharm Sci. 2019;129:1–9.
Gupta P, Kakumanu VK, Bansal AK. Stability and solubility of celecoxib-PVP amorphous dispersions: a molecular perspective. Pharm Res. 2004;21(10):1762–9.
Zhang M, Suo Z, Peng X, Gan N, Zhao L, Tang P, Wei X, Li H. Microcrystalline cellulose as an effective crystal growth inhibitor for the ternary Ibrutinib formulation. Carbohydr Polym. 2020;229:115476.
Ding X, Zheng M, Lu J, Zhu X. Preparation and evaluation of binary and ternary inclusion complexes of fenofibrate/hydroxypropyl-β-cyclodextrin. J Incl Phenom Macrocycl Chem. 2018;91:17–24.
Maggi L, Canobbio A, Bruni G, Musitelli G, Conte U. Improvement of the dissolution behavior of gliclazide, a slightly soluble drug, using solid dispersions. J Drug Deliv Sci Technol. 2015;26:17–23.
Acknowledgements
The authors thank Pr. Abdessalem Ben Haj Amara (Faculty of sciences of Bizerte, Tunisia) for his considerable inputs and helpful discussions. On the other hand, the authors confirmed that this research work did not receive any specific funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bejaoui, M., Kalfat, R. & Galai, H. The Effect of Adding PVP to the Binary Solid Dispersion (Indomethacin: Kaolin) on the Formation of Physically Stable Amorphous Drug. J Pharm Innov 17, 736–746 (2022). https://doi.org/10.1007/s12247-021-09553-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-021-09553-6