Abstract
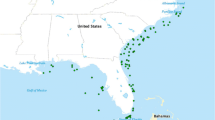
Fish diets play a critical role in our understanding of aquatic trophic dynamics and are an important component in developing ecosystem-based approaches to fisheries management. Although large nektonic fishes exert top-down predator effects on the food web and typically support viable commercial and recreational fisheries, little is known about the diet of this guild. We evaluated the diets (6327 stomachs) of four nektonic predatory fishes (Pomatomus saltatrix [78–395 mm], Cynoscion regalis [91–520 mm], Morone americana [156–361 mm], and Morone saxatilis [82–785 mm]) in Delaware Bay and in the adjacent ocean. To assess ontogenetic, geographic, and interspecific variation in diets, observations from individual fish stomachs were clustered into species-size class groups, and dietary overlap was estimated using multivariate analyses. A shift in diet composition, as well as diversity, occurred along the estuarine gradient and into the adjacent ocean. Some prey were shared by most predators, including some crustaceans (dominated by Callinectes sapidus, mysids, and Palaemonetes spp.), fundulids (dominated by Fundulus heteroclitus), engraulids (dominated by Anchoa mitchilli), and clupeids (dominated by Brevoortia tyrannus). However, inter- and intra-specific variation in diet was observed as well. In particular, M. americana consumed fewer engraulids and clupeids, and many more and diverse types of invertebrates, while P. saltatrix consumed more clupeids and less invertebrates. The lack of overlap in diet between the four predators evaluated, and between size groups for each predator, supports previous evidence that these groups feed in trophic guilds defined by species and by size within a species. The highly variable diets for these predators suggest high resolution spatial data are necessary in order to quantify their most important prey and their role in coastal ecosystems.





Similar content being viewed by others
References
Able, K.W., and M.P. Fahay. 2010. Ecology of estuarine fishes: Temperate waters of the western North Atlantic. Baltimore: Johns Hopkins University Press.
Able, K.W., D.M. Nemerson, R.O. Bush, and P. Light. 2001. Spatial variation in Delaware Bay (U.S.A.) marsh creek fish assemblages. Estuaries 24: 441–452.
Able, K.W., S.M. Hagan, K. Kovitvongsa, S.A. Brown, and J.C. Lamonaca. 2007. Piscivory by the mummichog Fundulus heteroclitus: Evidence from the laboratory and salt marshes. Journal of Experimental Marine Biology and Ecology 345: 26–37.
Able, K.W., K.M.M. Jones, and D.A. Fox. 2009. Large nektonic fishes in marsh creek habitats in the Delaware Bay estuary. Northeastern Naturalist 16: 27–44.
Able, K.W., T.M. Grothues, J.T. Turnure, M.A. Malone, and G.A. Henkes. 2014. Dynamics of residency and egress in selected estuarine fishes: Evidence from acoustic telemetry. Environmental Biology of Fishes. 97: 91–102.
Archambault, J.A., and R.J. Feller. 1991. Diel variations in gut fullness of juvenile spot, Leiostomus xanthurus (Pisces). Estuaries 14: 94–101.
Baker, R., and M. Sheaves. 2005. Redefining the piscivore assemblage of shallow estuarine nursery habitats. Marine Ecology Progress Series 291: 197–213.
Baker, R., and M. Sheaves. 2006. Visual surveys reveal high densities of large piscivores in shallow estuarine nurseries. Marine Ecology Progress Series 323: 75–82.
Beck, M.W., K.L. Heck, K.W. Able, D.L. Childers, D.B. Eggleston, B.M. Gillanders, B. Halpern, C.G. Hays, K. Hoshino, T.J. Minello, R.J. Orth, P.F. Sheridan, and M.P. Weinstein. 2001. The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. Bioscience 51: 633–641.
Biggs, R.B. 1978. Coastal bays. In Coastal sedimentary environments, ed. R.A. Davis, 69–99. New York: Springer-Verlag.
Bizzaro, J.J., M.M. Yoklavich, and W.W. Wakefield. 2016. Diet composition and foraging ecology of U. S. Pacific Coast groundfishes with applications for fisheries management. Environmental Biology of Fishes. doi:https://doi.org/10.1007/s10641-016-0529-2.
Buchheister, A., and R.J. Latour. 2015. Diets and trophic-guild structure of a diverse fish assemblage in Chesapeake Bay, U.S.A. Journal of Fish Biology 86: 967–992.
Buckel, J.A., and K.A. McKown. 2002. Competition between juvenile M. saxatilis and P. saltatrix: Resource partitioning and growth rate. Marine Ecology Progress Series 234: 191–204.
Buckel, J.A., M.J. Fogarty, and D.O. Conover. 1999. Foraging habits of bluefish, Pomatomus saltatrix, on the U.S. east coast continental shelf. Fishery Bulletin 97: 758–775.
Bundy, A., J.S. Link, B.E. Smith, and A.M. Cook. 2011. You are what you eat, whenever and wherever you eat it: An integrative analysis of fish food habits in Canadian and U.S.A. waters. Journal of Fish Biology 78: 514–539.
Carr, W.E., and C.A. Adams. 1972. Food habits of juvenile marine fishes: Evidence of the cleaning habit in the leatherjacket, Oligoplites saurus, and the spottail pinfish, Diplodus hoolbrooki. Fishery Bulletin 70: 1111–1120.
Cowan, J.H., J.C. Rice, C.J. Walters, R. Hilborn, T.E. Essington, J.W. Day, and K.M. Boswell. 2012. Challenges for implementing an ecosystem approach to fisheries management. Marine and Coastal Fisheries: Dynamics, Management, and Ecosystem Science 4: 496–510.
Daiber, F.C., and C.T. Roman. 1988. Tidal marshes. In The Delaware estuary: Rediscovery of a forgotten resource, ed. T.L. Bryant and J.R. Pennock, 95–113. Newark: University of Delaware Sea Grant College Program.
Dolbeth, M., F. Martinho, R. Leitao, H. Cabral, and M.A. Pardal. 2008. Feeding patterns of the dominant benthic and demersal fish community in a temperate estuary. Journal of Fish Biology 72: 2500–2517.
Eddy, T.D., H.K. Lotze, E.A. Fulton, M. Coll, C.H. Ainsworth, J.N. de Araujo, C.M. Bulman, A. Bundy, V. Christensen, J.C. Field, N.A. Gribble, M. Hasan, S. Mackinson, and H. Townsend. 2016. Ecosystem effects of invertebrate fisheries. Fish and Fisheries. doi:https://doi.org/10.1111/faf.12165.
Essington, T.E., and A.E. Punt. 2011. Implementing ecosystem-based fisheries management: Advances, challenges, and emerging tools. Fish and Fisheries 8: 241–268.
Essington, T.E., P.E. Moriarty, H.E. Froelich, E.E. Hodgson, L.E. Koehn, K.L. Oken, M. Siple, and C.C. Stawitz. 2015a. Fishing amplifies forage fish population collapses. Proceedings of the National Academy of Sciences of the United States 112: 6648–6652.
Essington, T.E., M. Siple, E.E. Hodgson, L.E. Koehn, P.E. Moriarty, K.L. Oken, and C.C. Stawitz. 2015b. Reply to Szuwalski and Hilborn: Forage fish require an ecosystem approach. Proceedings of the National Academy of Sciences of the United States 112: 3316.
Ferry, K.H., and M.E. Mather. 2012. Spatial and temporal diet patterns of subadult and small adult M. saxatilis in Massachusetts estuaries: Data, a synthesis, and trends across scales. Marine and Coastal Fisheries: Dynamics, Management, and Ecosystem Science 4: 30–45.
Friedland, K.D., G.C. Garman, A.J. Bejda, A.L. Studholme, and B. Olla. 1988. Interannual variation in diet and condition in juvenile bluefish during estuarine residency. Transactions of the American Fisheries Society 117: 474–479.
Gardinier, M.N., and J.B. Hoff. 1982. Diets of striped bass in the Hudson River estuary. New York Fish and Game Journal 29: 152–165.
Garrison, L.P., and J.S. Link. 2000. Dietary guild structure of the fish community in the Northeast United States continental shelf ecosystem. Marine Ecology Progress Series 202: 231–240.
Gartland, J., R.J. Latour, A.D. Halvorson, and H.M. Austin. 2006. Diet composition of young-of-the-year bluefish in the lower Chesapeake Bay and the coastal ocean of Virginia. Transactions of the American Fisheries Society 135: 371–378.
Griffin, J.C., and F.J. Margraf. 2003. The diet of Chesapeake Bay striped bass in the late 1950s. Fisheries Management and Ecology 10: 323–328.
Grothues, T.M., and K.W. Able. 2003a. Response of juvenile fish assemblages in tidal salt marsh creeks treated for Phragmites removal. Estuaries 26: 563–573.
Grothues, T.M., and K.W. Able. 2003b. Discerning vegetation and environmental correlates with subtidal marsh fish assemblage dynamics during Phragmites eradication efforts: Interannual trend measures. Estuaries 26: 574–586.
Hampel, H., A. Cattrijsse, and M. Elliott. 2005. Feeding habits of young predatory fishes in marsh creeks situated along the salinity gradient of the Schelde estuary, Belgium and The Netherlands. Helgoland Marine Research 59: 151–162.
Harding, J.M., and R. Mann. 2001. Diet and habitat use by P. saltatrix, Pomatomus saltatrix, in a Chesapeake Bay estuary. Environmental Biology of Fishes 60: 401–409.
Harrison, T.D., and A.K. Whitfield. 2012. Fish trophic structure in estuaries, with particular emphasis on estuarine typology and zoogeography. Journal of Fish Biology 81: 2005–2029.
Hartman, K.J., and S.B. Brandt. 1995a. Trophic resource partitioning, diets and growth of sympatric estuarine predators. Transactions of the American Fisheries Society 124: 520–537.
Hartman, K.J., and S.B. Brandt. 1995b. Predatory demand and impact of striped bass, bluefish, and weakfish in the Chesapeake Bay: Applications of bioenergetics models. Canadian Journal of Fisheries and Aquatic Sciences 52: 1667–1687.
Heck, K.L., K.W. Able, C.T. Roman, and M.P. Fahay. 1995. Composition, abundance, biomass, and production of macrofauna in a New England estuary: Comparisons among eelgrass meadows and other nursey habitats. Estuaries 18: 379–389.
Hilborn, R. 2011. Future directions in ecosystem based fisheries management: A personal perspective. Fisheries Research 108: 235–239.
Jaworski, A., and S.A. Ragnarsson. 2006. Feeding habits of demersal fish in Icelandic waters: A multivariate approach. ICES Journal of Marine Science 63: 1682–1694.
Jones, K.M.M., and K.W. Able. 2015. Abundance and diet of predatory fishes in Phragmites, treated Phragmites, and natural Spartina marshes in Delaware Bay. Estuaries and Coasts 38: 1350–1364.
Jones, K.M.M., P.E. McGrath, and K.W. Able. 2014. White perch Morone americana (Gmelin, 1789) habitat choice and movements: Comparisons between Phragmites-invaded and Spartina reference marsh creeks based on acoustic telemetry. Journal of Experimental Marine Biology and Ecology 455: 14–21.
Jost, L. 2006. Entropy and diversity. Oikos 113: 363–375.
Juanes, F., J.A. Buckel, and F.S. Scharf. 2002. Feeding ecology of piscivorous fishes. In Handbook of fish biology and fisheries volume 1: Fish biology, ed. P.J.B. Hart and J.D. Reynolds, 267–283. Malden: Blackwell Science.
Kleypas, J., and J.M. Dean. 1983. Migration and feeding of the predatory fish, Bairdiella chrysoura (Lacepede), in an intertidal creek. Journal of Experimental Marine Biology and Ecology 72: 199–209.
Lai, J. 2013. Canoco 5: A new version of an ecological multivariate data ordination program. Biodiversity Science 21: 765–768.
Link, J.S., A. Bundy, W.J. Overholtz, N. Shackell, J. Manderson, D. Duplisea, J. Hare, M. Koen-Alonso, and K.D. Friedland. 2011. Ecosystem-based fisheries management in the Northwest Atlantic. Fish and Fisheries 12: 152–170.
Manderson, J.P., L.L. Stehlik, J. Pessutti, J. Rosendale, and B. Phelan. 2014. Residence time and habitat duration for predators in a small mid-Atlantic estuary. Fishery Bulletin 112: 144–158.
Manooch, C.S., III. 1973. Food habits of yearling and adult striped bass, Morone saxatilis (Walbaum) from Albemarle Sound, North Carolina. Chesapeake Science 2: 142–205.
McFadden, K.W., and C. Barnes. 2009. The implementation of an ecosystem approach to management within a federal government agency. Marine Policy 33: 156–163.
Merriner, J.V. 1975. Food habits of the weakfish, Cynoscion regalis, in North Carolina waters. Chesapeake Science 16: 74–76.
Mollmann, C., M. Lindegren, T. Blenckner, L. Bergstrom, M. Casini, R. Diekmann, J. Flinkmanm, B. Muller-Karulis, S. Neuenfeldt, J.O. Schmidt, M. Tomczak, R. Voss, and A. Gardmark. 2014. Implementing ecosystem-based fisheries management: From single-species to integrated ecosystem assessment and advice for Baltic Sea fish stocks. ICES Journal of Marine Science 71: 1187–1197.
Murawski, S.A. 2007. Ten myths concerning ecosystem approaches to marine resource management. Marine Policy 31: 681–690.
Murtagh, F., and P. Legendre. 2014. Ward’s hierarchical agglomerative clustering method: Which algorithms implement Ward’s criterion? Journal of Classification 31: 274–295.
Musumeci, V.L., K.W. Able, M.C. Sullivan, and J.M. Smith. 2014. Estuarine predator—prey interactions in the early life history of two eels (Anguilla rostrata and Conger oceanicus). Environmental Biology of Fishes 97: 929–938.
Nagelkerken, I., G. van der Velde, and E. Cocheret de la Moriniere. 2001. Fish feeding guilds along a gradient of bay biotopes and coral reef depth zones. Aquatic Ecology 35: 73–86.
Nemerson, D.M., and K.W. Able. 2003. Spatial and temporal patterns in the distribution and feeding habits of Morone saxatilis, in marsh creeks of Delaware Bay, USA. Fisheries Management and Ecology 20: 337–348.
Nemerson, D.M., and K.W. Able. 2004. Spatial patterns in diet and distribution of juveniles of four fish species in Delaware Bay, USA marsh creeks: Factors influencing fish abundance. Marine Ecology Progress Series 276: 249–262.
Neuman, M.J., G. Ruess, and K.W. Able. 2004. Species composition and food habits of dominant fish predators in salt marshes of an urbanized estuary, the Hackensack Meadowlands, New Jersey. Urban Habitats 2: 62–82.
Overton, A.S., F.J. Margraf, and E.B. May. 2009. Spatial and temporal patterns in the diet of striped bass in Chesapeake Bay. Transactions of the American Fisheries Society 138: 915–926.
Patrick, W.S., and J.S. Link. 2015. Myths that continue to impede progress in ecosystem-based fisheries management. Fisheries 40: 155–160.
Pikitch, E.K., P.D. Boersma, I.L. Boyd, D.O. Conover, P. Cury, T.E. Essington, S.S. Heppell, E.D. Houde, M. Mangel, D. Pauly, E. Plaganyi, K. Sainsbury, and R. Steneck. 2012. Little fish, big impact: Managing a crucial link in ocean food webs. Washington D. C: Lenfest Ocean Program.
Pikitch, E. K., K. J. Rountos, T. E. Essington, C. Santora, D. Pauly, R. Watson, U. Sumaila, P. D. Boersma, I. L. Boyd, D. O. Conover, P. Cury, S. S. Heppell, E. D. Houde, M. Mangel, , E. Plaganyi, K. Sainsbury, R. Steneck, and T. M. Gears. 2014. The global contribution of forage fish to marine fisheries and ecosystems. Fish and Fisheries 15: 43–64.
R Development Core Team. 2016. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing.
Ralph, G.M., R.D. Seitz, R.J. Orth, K.E. Knick, and R.N. Lipcius. 2013. Broad-scale association between seagrass cover and juvenile blue crab density in Chesapeake Bay. Marine Ecology Progress Series 488: 51–63.
Ross, S.T. 1986. Resource partitioning in fish assemblages: A review of field studies. Copeia 1986: 352–388.
Rountree, R.A., and K.W. Able. 1997. Nocturnal fish use of New Jersey marsh creek and adjacent bay shoal habitats. Estuarine Coastal and Shelf Science 44: 703–711.
Ryer, C.H., J. Van Montfrans, and R.J. Orth. 1990. Utilization of sea grass meadow and tidal marsh creek by blue crabs Callinectes sapidus. II. Spatial and temporal patterns in molting. Bulletin of Maine Science 46: 95–104.
Scharf, F.S., J.A. Buckel, and F. Juanes. 2009. Contrasting patterns of resource utilization between juvenile estuarine predators: The influence of relative prey size and foraging ability on the ontogeny of piscivory. Canadian Journal of Fisheries and Aquatic Sciences 66: 790–801.
Schoener, T.W. 1968. Resource partitioning in ecological communities. Science 185: 27–39.
Sharp, J.H. 1988. Dynamics. In The Delaware estuary: Rediscovery of a forgotten resource, ed. T.L. Bryant and J.R. Pennock, 43–54. Newark: University of Delaware Sea Grant College Program.
Sheaves, M. 2001. Are there really few piscivorous fishes in shallow estuarine habitats? Marine Ecology Progress Series 222: 279–290.
Shelton, A.O., J.F. Samhouri, A.C. Stier, and P.S. Levin. 2014. Assessing trade-offs to inform ecosystem-based fisheries management of forage fish. Scientific Reports 4: 7110.
Smith, A.D.M., E.J. Fulton, A.J. Hobday, D.C. Smith, and P. Shoulder. 2007. Scientific tools to support the practical implementation of ecosystem-based fisheries management. ICES Journal of Marine Science 64: 633–639.
deSylva, D.P. 1985. Nektonic food webs in estuaries. In Fish community ecology in estuaries and coastal lagoons: Towards an ecosystem integration, ed. A. Yañez-Arancibia, 233–246. Mexico City: Universidad Nacional Autonoma de Mexico Press.
Taylor, E.T. 1987. Food habits of dominant piscivorous fishes in Delaware Bay, with special reference to predation on juvenile weakfish. M.S. Thesis, University of Delaware.
Toft, C.A. 1985. Resource partitioning in amphibians and reptiles. Copeia 1985: 1–21.
Tupper, M., and K.W. Able. 2000. Movements and food habits of striped bass (Morone saxatilis) in Delaware Bay (USA) salt marshes: Comparison of a restored and a reference marsh. Marine Biology 147: 1049–1058.
Turnure, J.T., T.M. Grothues, and K.W. Able. 2015. Seasonal residency of adult weakfish (Cynoscion regalis) in a small temperate estuary based on acoustic telemetry: A local perspective of a coast wide phenomenon. Environmental Biology of Fishes 98: 1207–1221.
Uphoff, J.H. 2003. Predator-prey analysis of striped bass and Atlantic menhaden in upper Chesapeake Bay. Fisheries Management and Ecology 10: 313–322.
Walter, J.F., III, and H.M. Austin. 2003. Diet composition of large striped bass (Morone saxatilis) in Chesapeake Bay. Fishery Bulletin 101: 414–423.
Walter, J.F., III, A.S. Overton, K.H. Ferry, and M.E. Mather. 2003. Atlantic coast feeding habits of striped bass: A synthesis supporting a coast-wide understanding of trophic biology. Fisheries Management and Ecology 10: 349–360.
Weinstein, M.P., J.H. Balletto, J.M. Teal, and D.F. Ludwig. 1997. Success criteria and adaptive management for a large-scale wetland restoration project. Wetlands Ecology Management 4: 111–127.
Weis, J.S. 2005. Diet and food web support of the white perch, Morone americana, in the Hackensack meadowlands of New Jersey. Environmental Biology of Fishes 74: 109–113.
Weisberg, S.B., R. Whalen, and V.A. Lotrich. 1981. Tidal and diurnal influence on food consumption of a salt marsh killifish Fundulus heteroclitus. Marine Biology 61: 243–246.
Winemiller, K.O. 1989. Ontogenetic diet shifts and resource partitioning among piscivorous fishes in Venezualan ilanos. Environmental Biology of Fishes 26: 177–199.
Wuenschel, M.J., K.W. Able, J.M. Vasslides, and D.M. Byrne. 2013. Habitat and diet overlap of four piscivorous fishes: Variation on the inner continental shelf off New Jersey. Fishery Bulletin 111: 352–369.
Acknowledgements
This project was funded by the collaborative Rutgers University-National Marine Fisheries Service Bluefish-Striped Bass Dynamics Research Program. We would like to thank all of the technicians and support staff at Rutgers University Marine Field Station (RUMFS) who assisted with this project, particularly R. Nichols, N. Salvi, J. Toth, R. Hagan, G. Petruzzelli, J. Lamonaca, and M. Greaney. We would especially like to thank the late Stacy Hagan for all of her help over the years with this research project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Matthew D. Taylor
Rights and permissions
About this article
Cite this article
Able, K.W., Morson, J.M. & Fox, D.A. Food Habits of Large Nektonic Fishes: Trophic Linkages in Delaware Bay and the Adjacent Ocean. Estuaries and Coasts 41, 866–883 (2018). https://doi.org/10.1007/s12237-017-0308-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-017-0308-0