Abstract
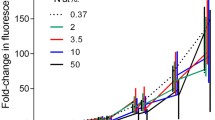
Leucine and thymidine incorporation were examined in size-fractionated estuarine communities and in cultures of phytoplankton known to use dissolved organic nitrogen (DON). Cultured phytoplankton species were used to establish that phytoplankton took up leucine and thymidine into protein and DNA, respectively. Subsequently, incorporation of leucine and thymidine was measured in size-fractionated populations collected from the Lafayette River, VA, a eutrophic estuary where resident populations contain bloom-forming phytoplankton known to take up DON, and the Gulf of Mexico during a bloom of the mixotrophic red tide dinoflagellate, Karenia brevis. We examined the efficacy of size fractionation for determining phytoplankton versus bacterial incorporation of leucine and thymidine under conditions employed during bacterial productivity bioassays, and antibiotics were used to distinguish between bacterial and phytoplankton incorporation in cultured and natural populations. Results suggest that cultures and natural assemblages of phytoplankton can take up both leucine and thymidine when supplied at low concentrations (10 and 12 nmol L−1, respectively) and during short incubations (15 min to 1 h). In natural populations, up to 95% of the leucine and thymidine incorporation during short bioassays was recovered in the >5.0-μm size fraction that contained ≤4.2% of the bacterial biomass.








Similar content being viewed by others
References
Alonso, C., and J. Pernthaler. 2006. Concentration-dependent patterns of leucine incorporation by coastal picoplankton. Applied and Environmental Microbiology 72: 2141–2147.
Alonso-Sáez, L., J. Arístegui, J. Pinhassi, L. Gómez-Consarnau, J.M. González, D. Vaqué, S. Agustí, and J.M. Gasol. 2006. Bacterial assemblage structure and carbon metabolism along a productivity gradient in the NE Atlantic Ocean. Aquatic Microbial Ecology 46: 43–53.
Antia, N.J., P.J. Harrison, and L. Oliveria. 1991. The role of dissolved organic nitrogen in phytoplankton nutrition, cell biology and ecology. Phycologia 30: 1–89.
Ast, M., A. Gruber, S. Schmitz-Esser, H.E. Neuhaus, P.G. Kroth, M. Horn, and I. Haferkamp. 2009. Diatom plastids depend on nucleotide import from the cytosol. Proceedings of the National Academy of Sciences 106: 3621–3626.
Azam, F., and R.E. Hodson. 1977. Size distribution and activity of marine microheterotrophs. Limnology and Oceanography 22: 492–501.
Azam, F., T. Fenchel, J.G. Field, J.S. Gray, L.A. Meyer-Reil, and F. Thingstad. 1983. The ecological role of water-column microbes in the sea. Marine Ecology Progress Series 10: 257–263.
Bell, R.T. 1993. Estimating production of heterotrophic bacterioplankton via incorporation of tritiated thymidine. In Handbook of methods in aquatic microbial ecology, ed. P.F. Kemp, B.F. Sherr, E.B. Sherr, and J.J. Cole, 495–504. Boca Raton: Lewis.
Berg, G.M., P.M. Glibert, M.W. Lomas, and M. Burford. 1997. Organic nitrogen uptake and growth by the chrysophyte Aureococcus anophagefferens during a brown tide event. Marine Biology 227: 377–387.
Berg, G.M., D.J. Repta, and J. LaRoche. 2003. The role of the picoeukaryote Aureococcus anophagefferens in cycling of marine high-molecular weight dissolved organic nitrogen. Limnology and Oceanography 48: 1825–1830.
3Berg, G.M., J. Shrager, G. Glöckner, K.R. Arrigo, and A.R. Grossman. 2008. Understanding nitrogen limitation in Aureococcus anophagefferens (Pelagophyceae) through cDNA and qRT-PCR analysis. Journal of Phycology 44: 1235–1249.
Berman, T., and D.A. Bronk. 2003. Dissolved organic nitrogen: A dynamic participant in aquatic ecosystems. Aquatic Microbial Ecology 31: 279–305.
Bern, L. 1985. Autoradiographic studies of (methyl-3H)-thymidine incorporation in a cyanobacterium (Microcystis wesenbergii)—bacterium association and in selected algae and bacteria. Applied and Environmental Microbiology 49: 232–233.
Bird, D.F., and J. Kalff. 1984. Empirical relationships between bacterial abundance and chlorophyll concentration in fresh and marine waters. Canadian Journal of Fisheries and Aquatic Sciences 41: 1015–1023.
Bouvier, T.C., and P.A. Del Giorgio. 2002. Compositional changes in free-living bacterial communities along a salinity gradient in two temperate estuaries. Limnology and Oceanography 47: 453–470.
Burdige, D.J., and J. Homstead. 1994. Fluxes of dissolved organic carbon from Chesapeake Bay sediments. Geochimica et Cosmochimica Acta 58: 3407–3424.
Burkholder, J.M., P.M. Glibert, and H.M. Skelton. 2008. Mixotrophy, a major mode of nutrition for harmful algal species in eutrophic waters. Harmful Algae 8(1): 77–93.
Carlson, C.A. 2002. Production and removal processes. In Biogeochemistry of dissolved organic matter, ed. D.A. Hansell and C.A. Carlson, 91–151. San Diego: Academic.
Chin-Leo, G., and D.L. Kirchman. 1988. Estimating bacterial production in marine waters from the simultaneous incorporation of thymidine and leucine. Applied and Environmental Microbiology 54: 1934–1939.
Chin-Leo, G., and D.L. Kirchman. 1990. Unbalanced growth in natural assemblages of marine bacterioplankton. Marine Ecology Progress Series 63: 1–8.
Cho, B.C., and F. Azam. 1990. Biogeochemical significance of bacterial biomass in the ocean’s euphotic zone. Marine Ecology Progress Series 63: 253–259.
Cole, J.J., S. Findlay, and M.L. Pace. 1988. Bacterial production in fresh and saltwater ecosystems: A cross-system overview. Marine Ecology Progress Series 43: 1–10.
Cottrell, M.T., and D.L. Kirchman. 2003. Contribution of major bacterial groups to bacterial biomass production (thymidine and leucine incorporation) in the Delaware estuary. Limnology and Oceanography 48: 167–178.
Cowie, G.L., and J.I. Hedges. 1992. Improved amino acid quantification in environmental samples: Charged-matched recovery standards and reduced analysis time. Marine Chemistry 37: 223–238.
Ducklow, H.W. 2000. Bacterioplankton stocks and production. In Microbial ecology of the oceans, ed. D.L. Kirchman, 85–120. New York: Wiley-Liss.
Ducklow, H.W., and F.-K. Shiah. 1993. Bacterila production in estuaries. In Aquatic Microbiology: an ecological approach, ed. T. E. Ford, 261–287. Oxford: Blackwell Scientific Publications.
Fan, C., and P.M. Glibert. 2005. Effects of light on nitrogen and carbon uptake during a Prorocentrum minimum bloom. Harmful Algae 4: 629–641.
Fan, C., P.M. Glibert, J. Alexander, and M.W. Lomas. 2003. Characterization of urease activity in three marine phytoplankton species, Aureococcus anophagefferens, Prorocentrum minimum, and Thalassiosira weissflogii. Marine Biology 142: 949–958.
Fuhrman, J.A., and F. Azam. 1980. Bacterioplankton secondary production estimates for coastal waters of British Columbia, Antarctica, and California. Applied and Environmental Microbiology 39: 1085–1095.
Fuhrman, J.A., and F. Azam. 1982. Thymidine incorporation as a measure of heterotrophic bacterioplankton production in marine surface waters: Evaluation and field results. Marine Biology 66: 109–120.
Glibert, P.M., C.A. Heil, D. Hollander, M. Revilla, A. Hoare, J. Alexander, and S. Murasko. 2004. Evidence for dissolved organic nitrogen and phosphorus uptake during a cyanobacterial bloom in Florida Bay. Marine Ecology Progress Series 280: 73–83.
Gobler, C.J., and S.A. Sañudo-Wilhelmy. 2001. Effects of organic carbon, organic nitrogen, inorganic nutrients, and iron additions on the growth of phytoplankton and bacteria during a brown tide bloom. Marine Ecology Progress Series 209: 19–34.
Goosen, N.K., P. van Rijswijk, J. Kromkamp, and J. Peene. 1997. Regulation of annual variation in heterotrophic bacterial production in the Schelde estuary (SW Netherlands). Aquatic Microbial Ecology 12: 223–232.
Graneli, E., P. Carlsson, and C. Legrand. 1999. The role of C, N, and P in dissolved and particulate organic matter as a nutrient source for phytoplankton growth, including toxic species. Aquatic Ecology 33: 17–27.
Griffith, P., F.-K. Shiah, K. Gloersen, H.W. Ducklow, and M. Fletcher. 1994. Activity and distribution of attached bacteria in Chesapeake Bay. Marine Ecology Progress Series 108: 1–10.
Hamdan, L.J., and R.B. Jonas. 2007. The use of antibiotics to reduce bacterioplankton uptake of phytoplankton extracellular organic carbon (EOC) in the Potomac River estuary. Journal of Experimental Marine Biology and Ecology 342: 242–252.
Heinänen, A., and J. Kuparinen. 1992. Response of bacterial thymidine and leucine incorporation to nutrient (NH4, PO4) and carbon (sucrose) enrichment. Archives of Hydrobiological Beih Ergebn Limnological 37: 241–251.
Heinänen, A., K. Kononen, H. Kuosa, J. Kuparinen, and K. Mäkelä. 1995. Bacterioplankton growth associated with physical fronts during a cyanobacterial bloom. Marine Ecology Progress Series 116: 233–245.
Hietanen, S., J.M. Lehtimaki, L. Tuominen, K. Sivonen, and J. Kuparinen. 2002. Nodularia spp. (Cyanobacteria) incorporate leucine but not thymidine: Importance for bacterial-production measurements. Aquatic Microbial Ecology 28: 99–104.
Jasti, S., M.E. Sieracki, N.J. Poulton, M.W. Giewat, and J.N. Rooney-Varga. 2005. Phylogenetic diversity and specificity of bacteria closely associated with Alexandrium spp. and other phytoplankton. Applied and Environmental Microbiology 71: 3483–3494.
Jeong, H.J., Y.D. Yoo, J.Y. Park, J.Y. Song, S.T. Kim, S.H. Lee, K.Y. Kim, and W.H. Yih. 2005a. Feeding by phototrophic red-tide dinoflagellates: Five species newly revealed and six species previously known to be mixotrophic. Aquatic Microbial Ecology 40: 133–150.
Jeong, H.J., J.Y. Park, J.H. Nho, M.O. Park, J.H. Ha, K.A. Seong, C. Jeng, C.N. Seong, K.Y. Lee, and W.H. Yih. 2005b. Feeding by red-tide dinoflagellates on the cyanobacterium Synechococcus. Aquatic Microbial Ecology 41: 131–143.
Kamjunke, N., and S. Jahnichen. 2000. Leucine incorporation by Microcystis aeruginosa. Limnology and Oceanography 45: 741–743.
Kirchman, D.L. 1993. Leucine incorporation as a measure of biomass production by heterotrophic bacteria. In Handbook of methods in aquatic microbial ecology, ed. P.F. Kemp, B.F. Sherr, E.B. Sherr, and J.J. Cole, 509–512. Boca Raton: Lewis.
Kirchman, D.L., and H.W. Ducklow. 1993. Estimating conversion factors for the thymicine and leucine methods for measuring bacterial production. In Handbook of methods in aquatic microbial ecology, ed. P.F. Kemp, B.F. Sherr, E.B. Sherr, and J.J. Cole, 513–517. Boca Raton: Lewis.
Kirchman, D.L., E. K’nees, and R. Hodson. 1985. Leucine incorporation and its potential as a measure of protein synthesis by bacteria in natural aquatic systems. Applied and Environmental Microbiology 49: 599–607.
Lee, S.-O., J. Kato, N. Takiguchi, A. Kuroda, T. Ikeda, A. Mitsutani, and H. Ohtake. 2000. Involvement of an extracellular protease in algicidal activity of the marine bacterium Pseudoalteromonas sp. strain A28. Applied and Environmental Microbiology 66: 4334–4339.
Lewitus, A.J. 2006. Osmotrophy in marine microalgae. In Algal cultures, analogues and blooms, ed. D.V. Subba Rao, 343–383. Enfield: Science.
Lewitus, A.J., B.M. Willis, K.C. Hayes, J.M. Burkholder, H.B. Glasgow Jr., P.M. Glibert, and M.K. Burke. 1999. Mixotrophy and nitrogen uptake by Pfiesteria piscicida (Dinophyceae). Journal of Phycology 35: 1430–1437.
Marshall, H.G., L. Burchardt, and R. Lacouture. 2005. A review of phytoplankton composition within Chesapeake Bay and its tidal estuaries. Journal of Plankton Research 27: 1083–1102.
Martinez, J., M. Riera, J. Lalucat, and J. Vives-Rego. 1989. Thymidine incorporation into algal DNA from axenic cultures of Synechococcus, Chlorella and Tetraselmis. Letters in Applied Microbiology 8: 135–138.
Michelou, V.K., M.T. Cottrell, and D.L. Kirchman. 2007. Light-stimulated bacterial production and amino acid assimilation by cyanobacteria and other microbes in the North Atlantic Ocean. Applied and Environmental Microbiology 73: 5539–5546.
Middelburg, J.J., and J. Nieuwenhuize. 2000. Nitrogen uptake by heterotrophic bacteria and phytoplankton in the nitrate-rich Thames estuary. Marine Ecology Progress Series 203: 13–21.
Minor, E.C., J.P. Simjouw, and M.R. Mulholland. 2006. Seasonal variations in dissolved organic carbon concentrations and characteristics in a shallow coastal bay. Marine Chemistry 101: 166–179.
Mulholland, M.R., and C. Lee. 2009. Peptide hydrolysis and dipeptide uptake in cultures and natural communities dominated by phytoplankton mixotrophs. Limnology and Oceanography 54: 856–868.
Mulholland, M.R., and M.W. Lomas. 2008. N uptake and assimilation. In Nitrogen in the marine environment, ed. D.G. Capone, D.A. Bronk, M.R. Mulholland, and E.J. Carpenter, 303–384. Amsterdam: Elsevier.
Mulholland, M.R., C.J. Gobler, and C. Lee. 2002. Peptide hydrolysis, amino acid oxidation, and nitrogen uptake in communities seasonally dominated by Aureococcus anophagefferens. Limnology and Oceanography 47: 1094–1108.
Mulholland, M.R., G. Boneillo, and E.C. Minor. 2004. A comparison of N and C uptake during brown tide (Aureococcus anophagefferens) blooms from two coastal bays on the east coast of the USA. Harmful Algae 3: 361–376.
Mulholland, M.R., G.E. Boneillo, P.W. Bernhardt, and E.C. Minor. 2009a. Comparison of nutrient and microbial dynamics over a seasonal cycle in a mid-Atlantic coastal lagoon prone to Aureococcus anophagefferens (brown tide) blooms. Estuaries and Coasts 32: 1176–1194.
Mulholland, M.R., R.E. Morse, G.E. Boneillo, P.W. Bernhardt, K.C. Filippino, L.A. Procise, J.L. Blanco-Garcia, H.G. Marshall, T.A. Egerton, W.S. Hunley, K.A. Moore, D.L. Berry, and C.J. Gobler. 2009b. Understanding causes and impacts of the dinoflagellate, Cochlodinium polykrikoides, blooms in the Chesapeake Bay. Estuaries and Coasts 32: 734–747.
Murrell, M.C. 2003. Bacterioplankton dynamics in a subtropical estuary: Evidence for substrate limitation. Aquatic Microbial Ecology 32: 239–250.
Murrell, M.C., J.T. Hollibaugh, M.W. Silver, and P.S. Wong. 1999. Bacterioplankton dynamics in northern San Francisco Bay: Role of particle association and seasonal freshwater flow. Limnology and Oceanography 44: 295–308.
North, B.B., and G.C. Stephens. 1972. Amino acid transport in Nitzschia ovalis Arnott. Journal of Phycology 8: 64–68.
Oremland, R.S., and D.G. Capone. 1988. Use of “specific” inhibitors in biogeochemistry and microbial ecology. Advances in Microbial Ecology 10: 285–383.
Paerl, H.W. 1991. Ecolophysiological and trophic implications of light-stimulated amino-acid utilization in marine picoplankton. Applied and Environmental Microbiology 57: 473–479.
Palenik, B.B., F.W. Larimer, M. Land, L. Hauser, P. Chain, J. Lamerdin, W. Regala, E.A. Allen, J. McCarren, I. Paulsen, A. Dufresne, F. Partensky, E.A. Webb, and J. Waterbury. 2003. The genome of a motile marine Synechococcus. Nature 424: 1037–1042.
Palenik, B., Q. Ren, C.L. Dupont, et al. 2006. Genome sequence of Synechococcus CC9311: Insights into adaptation to a coastal environment. Proceedings of the National Academy of Sciences 103: 13555–13559.
Parsons, T.R., Y. Maita, and C. Lalli. 1984. A manual of chemical and biological methods for seawater analysis. New York: Pergamon.
Phlips, E.J., S. Badylak, and T.C. Lynch. 1999. Blooms of the picoplanktonic cyanobacterium Synechococcus in Florida Bay, a subtropical inner-shelf lagoon. Limnology and Oceanography 44: 1166–1175.
Porter, K.G., and Y.S. Feig. 1980. The use of DAPI for identifying and counting aquatic microflora. Limnology and Oceanography 25: 943–948.
Price, N., and P.J. Harrison. 1987. Comparison of methods for the analysis of dissolved urea in seawater. Marine Biology 94: 307–317.
Rivkin, R.B. 1986. Incorporation of tritiated thymidine by eukaryotic microalgae. Journal of Phycology 22: 193–198.
Rivkin, R.B., and M.A. Voytek. 1986. Cell division rates of eukaryotic algae measured by tritiated thymidine incorporation into DNA: Coincident measurements of photosynthesis and cell division of individual species of phytoplankton isolated from natural populations. Journal of Phycology 22: 199–205.
Robarts, R.D., and T. Zohary. 1993. Fact or fiction—Bacterial growth rates and production as determined by [methyl-3H]-thymidine? Advances in Microbial Ecology 13: 371–425.
Schultz Jr., G.E., E.D. White III, and H.W. Ducklow. 2003. Bacterioplankton dynamics in the York River estuary: Primary influence of temperature and freshwater inputs. Aquat. Microbial Ecology 30: 135–148.
Shiah, F.-K., and H.W. Ducklow. 1994. Temperature regulation of heterotrophic bacterioplankton abundance, production, and specific growth rate in Chesapeake Bay. Limnology and Oceanography 39: 1243–1258.
Shiah, F.-K., and H.W. Ducklow. 1995. Multiscale variability in bacterioplankton abundance, production, and specific growth rate in a temperate salt-marsh tidal creek. Limnology and Oceanography 40: 55–66.
Steffensen, D.M., and W.F. Sheridan. 1965. Incorporation of H3-thymidine into chloroplast DNA of marine algae. The Journal of Cell Biology 25: 619–626.
Stoecker, D.K., A. Li, D.W. Coats, D.E. Gustafson, and M.K. Nannen. 1997. Mixotrophy in the dinoflagellate Prorocentrum minimum. Marine Ecology Progress Series 152: 1–12.
Tango, P.J., R. Magnien, W. Butler, C. Luckett, M. Luckenbach, R. Lacouture, and C. Pukish. 2005. Impacts and potential effects due to Prorocentrum minimum blooms in Chesapeake Bay. Harmful Algae 4: 525–531.
Valderrama, J.C. 1981. The simultaneous analysis of total nitrogen and total phosphorus in natural waters. Marine Chemistry 10: 109–122.
Veuger, B., J.J. Middelburg, H.T.S. Boschker, J. Nieuwenhuize, P. Van Rijswijk, E.J. R-Newall, and N. Navarro. 2004. Microbial uptake of dissolved organic and inorganic nitrogen in Randers Fjord. Estuarine, Coast and Shelf Science 61: 507–515.
Welschmeyer, N.A. 1994. Fluorometric analysis of chlorophyll a in the presence of chlorophyll b and pheopigments. Limnology and Oceanography 39: 1985–1992.
Wheeler, P.A., B.B. North, and G.C. Stephens. 1974. Amino acid uptake by marine phytoplankters. Limnology and Oceanography 19: 249–259.
Wheeler, P.A., B. North, M. Littler, and G. Stephens. 1977. Uptake of glycine by natural phytoplankton communities. Limnology and Oceanography 22: 900–909.
Wheeler, P.A., and D.L. Kirchman. 1986. Utilization of inorganic and organic nitrogen by bacteria in marine systems. Limnology and Oceanography 31: 998–1009.
Zubkov, M.V., and D.G.A. Tarran. 2005. Amino acid uptake of Prochlorococcus spp. in surface waters across the South Atlantic subtropical front. Aquatic Microbial Ecology 40: 241–249.
Zubkov, M.V., B.M. Fuchs, G.A. Tarran, P.H. Burkill, and R. Amann. 2003. High rate of uptake of organic nitrogen compounds by Prochlorococcus cyanobacteria as a key to their dominance in oligotrophic oceanic waters. Applied and Environmental Microbiology 69: 1299–1304.
Zubkov, M.V., D.G.A. Tarran, and B. Fuchs. 2004. Depth related amino acid uptake by Prochlorococcus cyanobacteria in the Southern Atlantic tropical gyre. FEMS Microbiology Ecology 20: 153–161.
Acknowledgments
We thank A. M. Watson and the captain and crew of the R/V Pelican for their help in collecting samples. A. Rocha was partially supported by a fellowship from the Hall-Bonner Program for Doctoral Scholars, established through a grant by the National Science Foundation to Ben Cuker (Hampton University), Greg Cutter (ODU), and Linda Schaffner (Virginia Institute of Marine Science); this program also provided support for laboratory supplies. This study was funded by grants from the National Science Foundation, the US ECOHAB program, the Virginia Environmental Endowment, and the Virginia Water Resources Research Center to MRM. The ECOHAB Program is sponsored by the National Oceanographic and Atmospheric Administration, Environmental Protection Agency, National Science Foundation, National Aeronautics and Space Administration, and Office of Naval Research. This is contribution number 636 from the US ECOHAB Program. Finally, we thank three anonymous reviewers and Carlos Duarte for their comments on the earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mulholland, M.R., Rocha, A.M. & Boneillo, G.E. Incorporation of Leucine and Thymidine by Estuarine Phytoplankton: Implications for Bacterial Productivity Estimates. Estuaries and Coasts 34, 310–325 (2011). https://doi.org/10.1007/s12237-010-9366-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-010-9366-2