Abstract
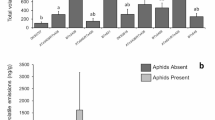
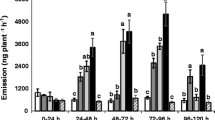
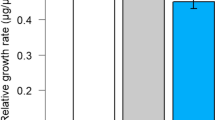
Green peach aphid Myzus persicae (Sulzer) is a serious pest of potato and causes severe yield loss to potato production worldwide. The volatiles emitted from potato plants influence host selection by M. persicae. A laboratory experiment was conducted to study how these volatile emissions vary among four potato varieties (IdaRose, Desiree, Chipeta and Russet Burbank) having different genetic backgrounds, and to compare the aphid responses to the volatiles emitted by these varieties. Results indicated that headspace volatiles detected, and aphid responses, differed significantly among the varieties. Myzus persicae arrestment was greater in response to Desiree and Chipeta than on Russet Burbank and arrestment on IdaRose was intermediate and did not differ significantly from any of the other varieties, but the arrestment was not related to the total concentration of volatiles released by these varieties. This indicates that the blend of volatiles was important for eliciting arrestment. Eigenvector weightings of principal component 1 were predominantly sesquiterpenes (especially caryophyllene, sesquiphellandrene, α-farnesene and longifolene), whereas those for principal component 2 were predominantly aldehydes and green leaf volatiles (n-hexana1, (Z)-3-hexenol, (E)-2-hexenal and nonanal. The pedigrees of the varieties are not associated with the volatile blends or aphid responses.
Resumen
El áfido verde del durazno Myzus persicae (Sulzer) es una plaga seria de la papa y causa severas pérdidas de rendimiento en la producción de papa en todo el mundo. Los compuestos volátiles emitidos de la planta de papa influencian la selección de hospedante por M. persicae. Se condujo un experimento de laboratorio para estudiar cómo éstas emisiones de volátiles varían entre cuatro variedades de papa (IdaRose, Desiree, Chipeta, y Russet Burbank) con contenidos de diferentes antecedentes genéticos, y para comparar las respuestas del áfido a los volátiles emitidos por estas variedades. Los resultados indicaron que los volátiles detectados en espacios libres y la respuesta de los áfidos, diferían significativamente entre las variedades. La permanencia de M. persicae era mayor en respuesta a Desiree y Chipeta que en Russet Burbank y la retención en IdaRose fue intermedia, y no se diferenció significativamente de cualquiera de las otras variedades, pero la retención no estuvo relacionada a la concentración total de volátiles liberados por estas variedades. Esto indica que la mezcla de volátiles era importante para inducir la permanencia. La ponderación del vector propio del componente principal 1 fue predominantemente sesquiterpenos (especialmente cariofileno, sesquifelandreno, α-farneseno y longifoleno), mientras que aquellos para el componente principal 2 fueron predominantemente aldehidos y volátiles de hoja verde (n-hexana1, (Z)-3-hexenol, (E)-2-hexenal y nonanal). No se asocia al pedigrí de las variedades con las mezclas de volátiles o con las respuestas de los áfidos.




Similar content being viewed by others
References
Agelopoulos, N.G., and J.A. Picket. 1998. Headspace analysis in chemical ecology: Effects of different sampling methods on ratios of volatile compounds present in headspace samples. Journal of Chemical Ecology 24: 1161–1172.
Baldwin, I.T., R. Halitschke, A. Paschold, C.C.V. Dahl, and C.A. Preston. 2006. Volatile signaling in plant-plant interactions: ‘talking trees’ in the genomics era. Science 311: 812–815.
Bernays, E.A., and R.F. Chapman. 1994. Host plant selection by phytophagous Insects. New York: Chapman and Hall.
Blackman, R.L., and V.F. Eastop. 1984. Aphids on the World’s crops: An identification and information guide. London: John Wiley and Sons.
Bolter, C.J., M. Dicke, J.J.A. Vanloon, J.H. Visser, and M.A. Posthumus. 1997. Attraction of Colorado potato beetle to herbivore-damaged plants during herbivory and after its termination. Journal of Chemical Ecology 23: 1003–1023.
Broadbent, L. 1953. Aphids and potato virus diseases. Biological Review 28: 350–380.
Bruce, T.J.A., L.J. Wadhams, and C.M. Woodcock. 2005. Insect host location: A volatile situation. Trends in Plant Science 10: 269–274.
Degan, T., C. Dillmann, F. Marian-Poll, and T.C.J. Turlings. 2004. High genetic variability of herbivore-induced volatile emission within a broad range of maize inbred lines. Plant Physiology 135: 1928–1938.
Dudareva, N., F. Negre, D.A. Nagegowda, and I. Orlova. 2006. Plant volatiles: Recent advances and future perspectives. Critical Reviews in Plant Sciences 25: 417–440.
Eigenbrode, S.D., H. Ding, P. Shiel, and P.H. Berger. 2002. Volatiles from potato plants infected with Potato leafroll virus attract and arrest the virus vector, Myzus persicae (Homoptera: Aphididae). Proceedings of the Royal Society of B: Biological Sciences 269: 455–460.
Emden, V.H.F., V.F. Eastop, R.D. Hughes, and M.J. Way. 1969. The ecology of Myzus persicae. Annual Review of Entomology 14: 197–270.
Fernandez, P.C., T. Meiners, C. Bjorkman, and M. Hilker. 2007. Electrophysiological responses of the blue willow leaf beetle, Phratora vulgatissima, to volatiles of different Salix viminalis genotypes. Entomologia Experimentalis et Applicata 125: 157–164.
Flanders, K.L., and E.B. Radcliffe. 1992. Host plant resistance in Solanum germplasm. Minnesota Agricultural Experiment Station, University of Minnesota, Station Bulletin, 599–1992.
Fritz, R.S., and E.L. Simms (eds.). 1992. Plant resistance to herbivores and pathogens. Chicago: University of Chicago Press.
Gouinguene, S., T. Degen, and T.C.J. Turlings. 2001. Variability in herbivore induced odour emissions among maize cultivars and their wild ancestors (teosinte). Chemoecology 11: 9–16.
Hoballah, M.E.F., C. Tamo, and T.C.J. Turlings. 2002. Differential attractiveness of induced odors emitted by eight maize varieties for the parasitoid Cotesia marginiventris: Is quality or quantity important? Journal of Chemical Ecology 28: 951–968.
Karlsson, M.F., G. Bigersson, A.M.C. Prado, F. Bosa, M. Bengtsson, and P. Witzgall. 2009. Plant odor analysis of potato: response of Guatemalan moth to above- and belowground potato volatiles. Journal of Agricultural and Food Chemistry 57: 5903–5909.
Kennedy, J.S., M.F. Day, and V.F. Eastop. 1962. A conspectus of aphids as vectors of plant viruses. London: Commonwealth Institute of Entomology.
Kleine, S., and C. Müller. 2011. Intraspecific plant chemical diversity and its relation to herbivory. Oecologia 166: 175–186.
Krips, O.E., P.E.L. Willems, R. Gols, M.A. Posthumus, G. Gort, and M. Dicke. 2001. Comparison of cultivars of ornamental crop Gerbera jamesonii on production of spider mite-induced volatiles, and their attractiveness to the predator Phytoseiulus persimilis. Journal of Chemical Ecology 27: 1355–1372.
Lou, Y., X. Hua, T.C.J. Turlings, J.A. Cheng, X. Chen, and G. Ye. 2006. Differences in induced volatile emissions among rice varieties result in differential attraction and parasitism of Nilaparvata lugens eggs by the parasitoid Anagrus nilaparvatae in the field. Journal of Chemical Ecology 32: 2375–2387.
Loughrin, J.H., A. Manukian, R.R. Heath, and J.H. Tumlinson. 1995. Volatiles emitted by different cotton varieties damaged by feeding beet armyworm larvae. Journal of Chemical Ecology 21: 1217–1227.
Love, S.L., D.L. Corsini, R. Novy, J.J. Pavek, A.R. Mosley, R.E. Thornton, S.R. James, D.C. Hane, and K.A. Rykbost. 2002. IdaRose: A potato variety with bright red skin, excellent culinary quality, and long tuber dormancy. American Journal of Potato Research 79: 79–84.
Martin, V., and G. Jander. 2010. Volatile communications in plant aphid interactions. Current Opinion in Plant Biology 13: 366–371.
Miller, J.R., and K.L. Strickler. 1984. Finding and accepting host plants. In Chemical ecology of insects, ed. W.J. Bell and R.T. Carde, 127–157. Boston: Sinauer Associates.
Najar-Rodriguez, A.J., C.G. Galizia, J. Stierle, and S. Dorn. 2010. Behavioral and neurophysiological responses of an insect to changing ratios of constituents in host plant-derived volatile mixtures. Journal of Experimental Biology 213: 3388–3397.
Ngumbi, E., S.D. Eigenbrode, N.A. Bosque-Pérez, H. Ding, and A. Rodriguez. 2007. Myzus persicae is arrested more by blends than by individual compounds elevated in headspace of PLRV-infected potato. Journal of Chemical Ecology 33: 1733–1747.
Nottingham, S.F., J. Hardie, G.W. Dawson, A.J. Hick, J.A. Pickett, L.J. Wadhams, and C.M. Woodcock. 1991. Behavioural and electrophysiological responses of aphids to host and non-host plant volatiles. Journal of Chemical Ecology 17: 1231–1242.
Painter, R.H. 1951. Insect resistance in crop plants. New York: Macmillan.
Petitt, F.L., and Z. Smilowitz. 1982. Green peach aphid feeding damage to potato in various plant growth stages. Journal of Economic Entomology 75: 431–435.
Pichersky, E., J.P. Noel, and N. Duderava. 2006. Biosynthesis of plant volatiles: Nature’s diversity and ingenuity. Science 311: 808–811.
Pickett, J.A., L.J. Wadhams, and C.M. Woodcock. 1992. The chemical ecology of aphids. Annual Review of Entomology 37: 67–90.
SAS Institute. 2010. SAS user’s guide statistics, version 9.2. Cary, NC.
Smith, C.M. 1989. Plant resistance to insects: A fundamental approach. New York: John Wiley and Sons.
Staudt, M., B. Jackson, H. El-Aouni, B. Buatois, J. Lacroze, J. Poësse, and M. Sauge. 2010. Volatile organic compound emissions induced by the aphid Myzus persicae differ among resistant and susceptible peach cultivars and a wild relative. Tree Physiology : 1–15.
Takabayashi, J., M. Dicke, and M.A. Posthumus. 1991. Variation in composition of predator-attracting allelochemicals emitted by herbivore-infested plants: Relative influence of plant and herbivore. Chemoecology 2: 1–6.
Tasin, M., A.C. Backman, M. Coracini, D. Casado, C. Ioriatti, and P. Witzgall. 2007. Synergism and redundancy in a plant volatile blend attracting grapevine moth females. Phytochemistry 68: 203–209.
Twomey, J.A., J.J. Pavek, D.G. Holm, M. Workman, and D.L. Corsini. 1982. Sangre: an oval, high quality red potato. American Potato Journal 59: 435–437.
Visser, J.H., P.G.M. Piron, and J. Hardie. 1996. The aphid’s peripheral perception of plant volatiles. Entomologia Experimentalis et Applicata 80: 35–38.
Wageningen potato pedigree database. 2012. http://www.plantbreeding.wur.nl/potatopedigree, accessed July 1.
Webster, B. 2012. The role of olfaction in host selection. Physiological Entomology 37: 10–18.
Webster, B., T. Bruce, J. Pickett, and J. Hardie. 2010. Volatiles functioning as host cues in a blend become non host cues when presented alone to the black bean aphid. Animal Behaviour 79: 451–457.
Webster, B., E. Qvarfordt, U. Olsson, and R. Glinwood. 2012. Different roles for innate and learnt behavioral responses to odors in insect host location. Behavioral Ecology 172. doi:10.1093/beheco/ars172.
Werner, B.J., T.M. Mowry, N.A. Bosque-Pérez, H. Ding, and S.D. Eigenbrode. 2009. Changes in green peach aphid responses to Potato leafroll virus-induced volatiles emitted during disease progression. Environmental Entomology 38: 1429–1438.
Acknowledgments
The research was supported in part by award number 2009-65104-05730 from the National Institute for Food and Agriculture. We thank William Price for assistance with statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajabaskar, D., Ding, H., Wu, Y. et al. Behavioral Responses of Green Peach Aphid, Myzus persicae (Sulzer), to the Volatile Organic Compound Emissions from Four Potato Varieties. Am. J. Potato Res. 90, 171–178 (2013). https://doi.org/10.1007/s12230-012-9282-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-012-9282-z