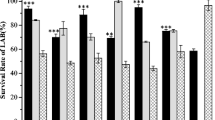
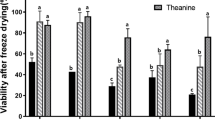
Abstract
Streptococcus thermophilus, the only Streptococcus species considered “Generally Recognized Safe”, has been used widely in the food industry. This bacterium is one of the most valuable industrial lactic acid bacterial species. Due to the importance of this bacterium in industrial applications, it should be stored for a long time without losing its metabolic properties. The present study aimed to investigate the cryoprotectant effect of three compatible solutes (ectoine, trehalose, and sucrose) on bacterial cells stored at different temperatures (frozen at -80 °C or freeze-dried and subsequently stored at +4, -20, and -80 °C) for three months. The bacterial cells were tested for cell viability, bile salt tolerance, and lactic acid production before and after processing. The highest cell viability, bile salt tolerance, and lactic acid production were obtained with ectoine and under frozen (storage at -80 °C) conditions. In freeze-dried and subsequently stored at various temperatures, the best preservation was obtained at -80 °C, followed by -20 °C and +4 °C. Moreover, when ectoine’s preservation potential was compared to other cryoprotectants, ectoine showed the highest preservation, followed by trehalose and sucrose. Although ectoine has a variety of qualities that have been proven, in the current work, we have shown for the first time that ectoine has cryoprotectant potential in yogurt starter cultures (S. thermophilus).



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abd El Ghany K, Hamouda R, Abd Elhafez E et al (2015) A potential role of Lactobacillus acidophilus LA1 and its exopolysaccharides on cancer cells in male albino mice. Biotechnol Biotechnol Equip 29:977–983. https://doi.org/10.1080/13102818.2015.1050455
Al-Fageeh MB, Smales CM (2006) Control and regulation of the cellular responses to cold shock: the responses in yeast and mammalian systems. Biochem J 397:247–259. https://doi.org/10.1042/BJ20060166
Amaretti A, Di Nunzio M, Pompei A et al (2013) Antioxidant properties of potentially probiotic bacteria: in vitro and in vivo activities. Appl Microbiol Biotechnol 97:809–817. https://doi.org/10.1007/s00253-012-4241-7
Andersen AB, Fog-Petersen MS, Larsen H, Skibsted LH (1999) Storage stability of freeze-dried starter cultures (Streptococcus thermophilus) as related to physical state of freezing matrix. LWT Food Sci Technol 32:540–547. https://doi.org/10.1006/fstl.1999.0594
Ayala-del-Río HL, Chain PS, Grzymski JJ et al (2010) The genome sequence of Psychrobacter arcticus 273-4, a psychroactive siberian permafrost bacterium, reveals mechanisms for adaptation to low-temperature growth. Appl Environ Microbiol 76:2304–2312. https://doi.org/10.1128/AEM.02101-09
Bachmann H, Kleerebezem M, Van Hylckama Vlieg JET (2008) High-throughput identification and validation of in situ-expressed genes of Lactococcus lactis. Appl Environ Microbiol 74:4727–4736. https://doi.org/10.1128/AEM.00297-08
Baindara P, Mandal SM (2020) Bacteria and bacterial anticancer agents as a promising alternative for cancer therapeutics. Biochimie 177:164–189. https://doi.org/10.1016/j.biochi.2020.07.020
Bonczar G, Walczycka MB, Domagała J et al (2016) Effect of dairy animal species and of the type of starter cultures on the cholesterol content of manufactured fermented milks. Small Rumin Res 136:22–26. https://doi.org/10.1016/j.smallrumres.2015.12.033
Bruno-Bárcena JM, Andrus JM, Libby SL et al (2004) Expression of a heterologous manganese superoxide dismutase gene in intestinal lactobacilli provides protection against hydrogen peroxide toxicity. Appl Environ Microbiol 70:4702–4710. https://doi.org/10.1128/AEM.70.8.4702-4710.2004
Cao R, Sogabe T, Mikajiri S, Kawai K (2022) Effects of sucrose, carnosine, and their mixture on the glass transition behavior and storage stability of freeze-dried lactic acid bacteria at various water activities. Cryobiology 106:131–138. https://doi.org/10.1016/j.cryobiol.2022.02.003
Cheng Z, Yan X, Wu J et al (2022) Effects of freeze drying in complex lyoprotectants on the survival, and membrane fatty acid composition of Lactobacillus plantarum L1 and Lactobacillus fermentum L2. Cryobiology 105:1–9. https://doi.org/10.1016/j.cryobiol.2022.01.003
Cui Y, Xu T, Qu X et al (2016) New insights into various production characteristics of streptococcus thermophilus strains. IJMS 17:1701. https://doi.org/10.3390/ijms17101701
Cukkemane A, Kumar P, Sathyamoorthy B (2020) A metabolomics footprint approach to understanding the benefits of synbiotics in functional foods and dietary therapeutics for health, communicable and non-communicable diseases. Food Res Int 128:108679. https://doi.org/10.1016/j.foodres.2019.108679
Czech L, Hermann L, Stöveken N et al (2018) Role of the extremolytes ectoine and hydroxyectoine as stress protectants and nutrients: genetics, phylogenomics, biochemistry, and structural analysis. Genes 9:177. https://doi.org/10.3390/genes9040177
da Costa MS, Santos H, Galinski EA (1998) An overview of the role and diversity of compatible solutes in Bacteria and Archaea. In: Antranikian G (ed) Biotechnology of Extremophiles. Springer, Berlin Heidelberg, Berlin, Heidelberg, pp 117–153
Dateoka S, Ohnishi Y, Kakudo K (2012) Effects of CRM197, a specific inhibitor of HB-EGF, in oral cancer. Med Mol Morphol 45:91–97. https://doi.org/10.1007/s00795-011-0543-6
De Vuyst L, Tsakalidou E (2008) Streptococcus macedonicus, a multi-functional and promising species for dairy fermentations. Int Dairy J 18:476–485. https://doi.org/10.1016/j.idairyj.2007.10.006
De Vuyst L, Weckx S, Ravyts F et al (2011) New insights into the exopolysaccharide production of Streptococcus thermophilus. Int Dairy J 21:586–591. https://doi.org/10.1016/j.idairyj.2011.03.016
Dwivedi M, Brinkkötter M, Harishchandra RK, Galla HJ (2014) Biophysical investigations of the structure and function of the tear fluid lipid layers and the effect of ectoine. Part B: Artificial lipid films. Biochim Biophys Acta (BBA) Biomembranes 1838:2716–2727. https://doi.org/10.1016/j.bbamem.2014.05.007
El Assal R, Guven S, Gurkan UA et al (2014) Bio-inspired cryo-ink preserves red blood cell phenotype and function during nanoliter vitrification. Adv Mater 26:5815–5822. https://doi.org/10.1002/adma.201400941
Fonseca F, Béal C, Corrieu G (2001) Operating conditions that affect the resistance of lactic acid bacteria to freezing and frozen storage. Cryobiology 43:189–198. https://doi.org/10.1006/cryo.2001.2343
Foschino R, Fiori E, Galli A (1996) Survival and residual activity of Lactobacillus acidophilus frozen cultures under different conditions. J Dairy Res 63:295–303. https://doi.org/10.1017/S0022029900031782
Gao J, Gu F, Ruan H et al (2013) Induction of apoptosis of gastric cancer cells SGC7901 in vitro by a cell-free fraction of Tibetan kefir. Int Dairy J 30:14–18. https://doi.org/10.1016/j.idairyj.2012.11.011
Gezginc Y, Akyol I, Kuley E, Özogul F (2013) Biogenic amines formation in Streptococcus thermophilus isolated from home-made natural yogurt. Food Chem 138:655–662. https://doi.org/10.1016/j.foodchem.2012.10.138
Gholamhosseinpour A, Hashemi SMB (2019) Ultrasound pretreatment of fermented milk containing probiotic Lactobacillus plantarum AF1: Carbohydrate metabolism and antioxidant activity. J Food Process Eng 42:e12930. https://doi.org/10.1111/jfpe.12930
Gilbert JA, Hill PJ, Dodd CER, Laybourn-Parry J (2004) Demonstration of antifreeze protein activity in Antarctic lake bacteria. Microbiology 150:171–180. https://doi.org/10.1099/mic.0.26610-0
Giulio BD, Orlando P, Barba G et al (2005) Use of alginate and cryo-protective sugars to improve the viability of lactic acid bacteria after freezing and freeze-drying. World J Microbiol Biotechnol 21:739–746. https://doi.org/10.1007/s11274-004-4735-2
Guo N, Song Y, Yan J, Jiang M, Xu Y, Li Z, Wei Q (2023) The effect of Cryopreservation on the survival of Nocardia farcinica and Yersinia pestis vaccine strains. Biopreserv Biobank 21(4):397–406
Hahn MB, Meyer S, Schröter MA et al (2017) DNA protection by ectoine from ionizing radiation: molecular mechanisms. Phys Chem Chem Phys 19:25717–25722. https://doi.org/10.1039/C7CP02860A
Harishchandra RK, Wulff S, Lentzen G et al (2010) The effect of compatible solute ectoines on the structural organization of lipid monolayer and bilayer membranes. Biophys Chem 150:37–46. https://doi.org/10.1016/j.bpc.2010.02.007
Haroun BM, Refaat BM, El-Menoufy HA et al (2013) Structure analysis and antitumor activity of the exopolysaccharide from probiotic Lactobacillus plantarum NRRL B- 4496 in vitro and in vivo. J Appl Sci Res 9:425–434
Held C, Neuhaus T, Sadowski G (2010) Compatible solutes: Thermodynamic properties and biological impact of ectoines and prolines. Biophys Chem 152:28–39. https://doi.org/10.1016/j.bpc.2010.07.003
Heylen K, Hoefman S, Vekeman B et al (2012) Safeguarding bacterial resources promotes biotechnological innovation. Appl Microbiol Biotechnol 94:565–574. https://doi.org/10.1007/s00253-011-3797-y
Hirayama K, Rafter J (1999) The role of lactic acid bacteria in colon cancer prevention: mechanistic considerations. In: Konings WN, Kuipers OP, In ’T Veld JHJH (eds) Lactic acid bacteria: Genetics, metabolism and applications. Springer Netherlands, Dordrecht, pp 391–394
Hols P, Hancy F, Fontaine L et al (2005) New insights in the molecular biology and physiology of Streptococcus thermophilus revealed by comparative genomics. FEMS Microbiol Rev 29:435–463. https://doi.org/10.1016/j.fmrre.2005.04.008
Huang Y, Wu F, Wang X et al (2013) Characterization of Lactobacillus plantarum Lp27 isolated from Tibetan kefir grains: A potential probiotic bacterium with cholesterol-lowering effects. J Dairy Sci 96:2816–2825. https://doi.org/10.3168/jds.2012-6371
Hüfner E, Markieton T, Chaillou S et al (2007) Identification of Lactobacillus sakei genes induced during meat fermentation and their role in survival and growth. Appl Environ Microbiol 73:2522–2531. https://doi.org/10.1128/AEM.02396-06
Iyer R, Tomar SK, Uma Maheswari T, Singh R (2010) Streptococcus thermophilus strains: Multifunctional lactic acid bacteria. Int Dairy J 20:133–141. https://doi.org/10.1016/j.idairyj.2009.10.005
Jana AM, Singh P (2020) Bacterial Preservation. Int J Life Sci Technol 13:1–29. https://doi.org/10.5281/ZENODO.4014767
Jawan R, Abbasiliasi S, Tan JS et al (2022) Influence of type and concentration of lyoprotectants, storage temperature and storage duration on cell viability and antibacterial activity of freeze-dried lactic acid bacterium, Lactococcus lactis Gh1. Drying Technol 40:1774–1790. https://doi.org/10.1080/07373937.2021.1874968
Jin UH, Cheng Y, Park H et al (2017) Short chain fatty acids enhance Aryl Hydrocarbon (Ah) responsiveness in Mouse Colonocytes and Caco-2 human colon cancer cells. Sci Rep 7:10163. https://doi.org/10.1038/s41598-017-10824-x
Kadoya S, Fujii K, Izutsu K et al (2010) Freeze-drying of proteins with glass-forming oligosaccharide-derived sugar alcohols. Int J Pharm 389:107–113. https://doi.org/10.1016/j.ijpharm.2010.01.027
Kandil S, El Soda M (2015) Influence of freezing and freeze drying on intracellular enzymatic activity and autolytic properties of some lactic acid bacterial strains. AIDS Patient Care STDs 05:371–382. https://doi.org/10.4236/aim.2015.56039
Klein G, Pack A, Bonaparte C, Reuter G (1998) Taxonomy and physiology of probiotic lactic acid bacteria. Int J Food Microbiol 41:103–125. https://doi.org/10.1016/S0168-1605(98)00049-X
Kuhn E (2012) Toward understanding life under subzero conditions: The significance of exploring psychrophilic “Cold-Shock” proteins. Astrobiology 12:1078–1086. https://doi.org/10.1089/ast.2012.0858
Lagos R, Tello M, Mercado G et al (2009) Antibacterial and antitumorigenic properties of Microcin E492, a pore- forming Bacteriocin. Curr Pharm Biotechnol 10:74–85. https://doi.org/10.2174/138920109787048643
Lee J, Hwang KT, Heo M-S et al (2005) Resistance of Lactobacillus plantarum KCTC 3099 from Kimchi to oxidative stress. J Med Food 8:299–304. https://doi.org/10.1089/jmf.2005.8.299
Lee SB, Kim DH, Park HD (2016) Effects of protectant and rehydration conditions on the survival rate and malolactic fermentation efficiency of freeze-dried Lactobacillus plantarum JH287. Appl Microbiol Biotechnol 100:7853–7863. https://doi.org/10.1007/s00253-016-7509-5
Lentzen G, Schwarz T (2006) Extremolytes: natural compounds from extremophiles for versatile applications. Appl Microbiol Biotechnol 72:623–634. https://doi.org/10.1007/s00253-006-0553-9
Leslie SB, Israeli E, Lighthart B et al (1995) Trehalose and sucrose protect both membranes and proteins in intact bacteria during drying. Appl Environ Microbiol 61:3592–3597. https://doi.org/10.1128/aem.61.10.3592-3597.1995
Li B, Tian F, Liu X et al (2011) Effects of cryoprotectants on viability of Lactobacillus reuteri CICC6226. Appl Microbiol Biotechnol 92:609–616. https://doi.org/10.1007/s00253-011-3269-4
Li H, Lu M, Guo H et al (2010) Protective effect of sucrose on the membrane properties of Lactobacillus casei Zhang subjected to freeze-drying. J Food Prot 73:715–719. https://doi.org/10.4315/0362-028X-73.4.715
Li S, Huang R, Shah NP et al (2014) Antioxidant and antibacterial activities of exopolysaccharides from Bifidobacterium bifidum WBIN03 and Lactobacillus plantarum R315. J Dairy Sci 97:7334–7343. https://doi.org/10.3168/jds.2014-7912
Liu JR, Chen MJ, Lin CW (2005) Antimutagenic and antioxidant properties of Milk−Kefir and Soymilk−Kefir. J Agric Food Chem 53:2467–2474. https://doi.org/10.1021/jf048934k
Luzardo MDC, Amalfa F, Nuñez AM et al (2000) Effect of trehalose and sucrose on the hydration and dipole potential of lipid bilayers. Biophys J 78:2452–2458. https://doi.org/10.1016/S0006-3495(00)76789-0
Mahler S, Desille M, Frémond B et al (2003) Hypothermic storage and cryopreservation of Hepatocytes: The protective effect of Alginate Gel against cell damages. Cell Transplant 12:579–592. https://doi.org/10.3727/000000003108747181
Marino M, Maifreni M, Rondinini G (2003) Microbiological characterization of artisanal Montasio cheese: analysis of its indigenous lactic acid bacteria. FEMS Microbiol Lett 229:133–140. https://doi.org/10.1016/S0378-1097(03)00816-4
Meyer S, Schröter MA, Hahn MB et al (2017) Ectoine can enhance structural changes in DNA in vitro. Sci Rep 7:7170. https://doi.org/10.1038/s41598-017-07441-z
Mohan CD, Rangappa S, Nayak SC et al (2022) Bacteria as a treasure house of secondary metabolites with anticancer potential. Semin Cancer Biol 86:998–1013. https://doi.org/10.1016/j.semcancer.2021.05.006
Nam SO, Yotsumoto F, Miyata K et al (2016) Anti-tumor effect of intravenous administration of CRM197 for triple-negative breast cancer Therapy. Anticancer Res 36:3651–3657
Nouri Z, Karami F (2016) Dual anti-metastatic and anti-proliferative activity assessment of two probiotics on HeLa and HT-29 cell lines. Cell J 18:127–134. https://doi.org/10.22074/cellj.2016.4307
Obata H, Ishigaki H, Kawahara H, Yamade K (1998) Purification and characterization of a novel cold-regulated protein from an ice-nucleating bacterium, Pseudomonas fluorescens KUIN-1. Biosci Biotechnol Biochem 62:2091–2097. https://doi.org/10.1271/bbb.62.2091
Olsson C, Swenson J (2020) Structural comparison between Sucrose and Trehalose in Aqueous solution. J Phys Chem B 124:3074–3082. https://doi.org/10.1021/acs.jpcb.9b09701
Orhan F, Ceyran E (2022) Identification of novel halophilic/halotolerant bacterial species producing compatible solutes. Int Microbiol. https://doi.org/10.1007/s10123-022-00289-y
Orhan F, Ceyran E, Akincioğlu A (2023a) Optimization of ectoine production from Nesterenkonia xinjiangensis and one-step ectoine purification. Biores Technol 371:128646. https://doi.org/10.1016/j.biortech.2023.128646
Orhan F, Parlak KU, Tabay D, Bozarı S (2023b) Alleviation of the cadmium toxicity by application of a microbial derived compound, Ectoine. Water Air Soil Pollut 234:534. https://doi.org/10.1007/s11270-023-06562-8
Ott A, Germond J-E, Chaintreau A (2000) Origin of acetaldehyde during milk fermentation using 13 C-labeled precursors. J Agric Food Chem 48:1512–1517. https://doi.org/10.1021/jf9904867
Panoff JM, Legrand S, Thammavongs B, Boutibonnes P (1994) The cold shock response inLactococcus lactis subsp.lactis. Curr Microbiol 29:213–216. https://doi.org/10.1007/BF01570156
Parwata IP, Maryam S, Tika IN (2022) Ectoine and Hydroxyectoine from Halophilic bacteria isolated from traditional solar saltern at Pejarakan Village, Buleleng Regency, Bali. HAYATI J Biosci 29:669–680. https://doi.org/10.4308/hjb.29.5.669-680
Parwata IP, Wahyuningrum D, Suhandono S, Hertadi R (2021) Ability of ectoine to stabilize Lipase against elevated temperatures and methanol concentrations. Indones J Chem 21:494. https://doi.org/10.22146/ijc.54931
Pasley S, Zylberberg C, Matosevic S (2017) Natural killer-92 cells maintain cytotoxic activity after long-term cryopreservation in novel DMSO-free media. Immunol Lett 192:35–41. https://doi.org/10.1016/j.imlet.2017.09.012
Pei R, Martin DA, DiMarco DM, Bolling BW (2017) Evidence for the effects of yogurt on gut health and obesity. Crit Rev Food Sci Nutr 57:1569–1583. https://doi.org/10.1080/10408398.2014.883356
Phadtare S (2004) Recent developments in bacterial cold-shock response. Curr Issues Mol Biol 6:125–136. https://doi.org/10.21775/cimb.006.125
Pitts FN (1969) The biochemistry of anxiety. Sci Am 220:69–75
Polo L, Mañes-Lázaro R, Olmeda I et al (2017) Influence of freezing temperatures prior to freeze-drying on viability of yeasts and lactic acid bacteria isolated from wine. J Appl Microbiol 122:1603–1614. https://doi.org/10.1111/jam.13465
Reddy KBPK, Awasthi SP, Madhu AN, Prapulla SG (2009) Role of cryoprotectants on the viability and functional properties of probiotic lactic acid bacteria during freeze drying. Food Biotechnol 23:243–265. https://doi.org/10.1080/08905430903106811
Rodrigues JP, Paraguassú-Braga FH, Carvalho L et al (2008) Evaluation of trehalose and sucrose as cryoprotectants for hematopoietic stem cells of umbilical cord blood. Cryobiology 56:144–151. https://doi.org/10.1016/j.cryobiol.2008.01.003
Sarmadi BH, Ismail A (2010) Antioxidative peptides from food proteins: A review. Peptides 31:1949–1956. https://doi.org/10.1016/j.peptides.2010.06.020
Schröter MA, Meyer S, Hahn MB et al (2017) Ectoine protects DNA from damage by ionizing radiation. Sci Rep 7:15272. https://doi.org/10.1038/s41598-017-15512-4
Schwibbert K, Marin-Sanguino A, Bagyan I et al (2011) A blueprint of ectoine metabolism from the genome of the industrial producer Halomonas elongata DSM 2581T: Ectoine metabolism from the Halomonas genome. Environ Microbiol 13:1973–1994. https://doi.org/10.1111/j.1462-2920.2010.02336.x
Shan B, Ai Z, Zeng S et al (2020) Gut microbiome-derived lactate promotes to anxiety-like behaviors through GPR81 receptor-mediated lipid metabolism pathway. Psychoneuroendocrinology 117:104699. https://doi.org/10.1016/j.psyneuen.2020.104699
Sharma B, Singh S, Kanwar SS (2014) L -Methionase: A therapeutic enzyme to treat malignancies. Biomed Res Int 2014:1–13. https://doi.org/10.1155/2014/506287
Shu G, Zhang B, Hui Y, et al (2017) Optimization of cryoprotectants for Streptococcus thermophilus during freeze-drying using Box-Behnken experimental design. Emir J Food Agric 1. https://doi.org/10.9755/ejfa.2016-07-960
Siaterlis A, Deepika G, Charalampopoulos D (2009) Effect of culture medium and cryoprotectants on the growth and survival of probiotic lactobacilli during freeze drying. Lett Appl Microbiol 48:295–301. https://doi.org/10.1111/j.1472-765X.2008.02529.x
Stanishevskaya O, Silyukova Y, Tereshina V et al (2023) Trehalose as a stabilizer of the lipid composition of membranes and the composition of the Cytosol of frozen/thawed rooster Spermatozoa. Agriculture 13:1387. https://doi.org/10.3390/agriculture13071387
Tang Y, Chen Y, Jiang H et al (2011) G-protein-coupled receptor for short-chain fatty acids suppresses colon cancer. Int J Cancer 128:847–856. https://doi.org/10.1002/ijc.25638
To BCS, Etzel MR (1997) Spray drying, freeze drying, or freezing of three different lactic acid bacteria species. J Food Science 62:576–578. https://doi.org/10.1111/j.1365-2621.1997.tb04434.x
Tsai YT, Cheng PC, Pan TM (2014) Anti-obesity effects of gut microbiota are associated with lactic acid bacteria. Appl Microbiol Biotechnol 98:1–10. https://doi.org/10.1007/s00253-013-5346-3
Uriot O, Denis S, Junjua M et al (2017) Streptococcus thermophilus: From yogurt starter to a new promising probiotic candidate? J Funct Foods 37:74–89. https://doi.org/10.1016/j.jff.2017.07.038
Vaningelgem F, Zamfir M, Mozzi F et al (2004) Biodiversity of Exopolysaccharides produced by Streptococcus thermophilus strains is reflected in their production and their molecular and functional characteristics. Appl Environ Microbiol 70:900–912. https://doi.org/10.1128/AEM.70.2.900-912.2004
Vinderola G, Perdigón G, Duarte J et al (2006) Effects of the oral administration of the exopolysaccharide produced by Lactobacillus kefiranofaciens on the gut mucosal immunity. Cytokine 36:254–260. https://doi.org/10.1016/j.cyto.2007.01.003
Von Wright A, Axelsson L (2019) Lactic acid bacteria: An introduction. In: Lactic acid bacteria, 5th edn. CRC Press, pp 1–16
Waditee-Sirisattha R, Kageyama H, Takabe T (2016) Halophilic microorganism resources and their applications in industrial and environmental biotechnology. AIMS Microbiology 2:42–54. https://doi.org/10.3934/microbiol.2016.1.42
WHO (2006) Probiotics in food: health and nutritional properties and guidelines for evaluation. Food and Agriculture Organization of the United Nations: World Health Organization, Rome
Yu G, Li R, Hubel A (2019) Interfacial interactions of sucrose during cryopreservation detected by Raman spectroscopy. Langmuir 35:7388–7395. https://doi.org/10.1021/acs.langmuir.8b01616
Funding
This research received no specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This article contains no studies with human participants or animals.
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Orhan, F., Demirci, A., Efe, D. et al. Usage of ectoine as a cryoprotectant for cryopreservation of lactic acid bacteria. Folia Microbiol 69, 133–144 (2024). https://doi.org/10.1007/s12223-023-01098-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-023-01098-0