Abstract
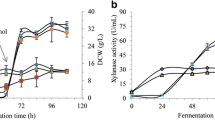
Pichia pastoris, a methylotrophic yeast, is known to be an efficient host for heterologous proteins production. In this study, a recombinant P. pastoris Y11430 was found better for β-glucosidase activity in comparison with a wild type P. pastoris Y11430 strain, and thereby, subjected to methanol intermittent feed profiling for β-glucosidase production. The results showed that at 72 h of cultivation time, the cultures with 16.67% and 33.33% methanol feeding with constant rate could produce the total dry cell weight of 52.23 and 118.55 g/L, respectively, while the total mutant β-glucosidase activities were 1001.59 and 3259.82 units, respectively. The methanol feeding profile was kept at 33% with three methanol feeding strategies such as constant feed rate, linear feed rate, and exponential feed rate which were used in fed-batch fermentation. At 60 h of cultivation, the highest total mutant β-glucosidase activity was 2971.85 units for exponential feed rate culture. On the other hand, total mutant β-glucosidase activity of the constant feed rate culture and linear feed rate culture were 1682.25 and 1975.43 units, respectively. The kinetic parameters of exponential feed rate culture were specific growth rate on glycerol 0.228/h, specific growth of methanol 0.061/h, maximum total dry cell weight 196.73 g, yield coefficient biomass per methanol (\({Y_{x/m}}\)) 0.57 gcell/gMeOH, methanol consumption rate (\({Q_m}\)) 5.76 gMeOH/h, and enzyme productivity (\({Q_p}\)) 75.96 units/h. In conclusion, higher cell mass and β- glucosidase activity were produced under exponential feed rate than constant and linear feed rates.








Similar content being viewed by others
Availability of data and materials
All the data has been declared.
References
Azadi S, Mahboubi A, Naghdi N, Solaimanian R, Mortazavi SA (2017) Evaluation of sorbitol-methanol co-feeding strategy on production of recombinant human growth hormone in Pichia pastoris. Iran J Pharm Res 16:1555–1564. PMID: 29552064
Beiroti A, Hosseini SN, Aghasadeghi MR, Norouzian D (2019) Comparative study of μ-stat methanol feeding control in fed-batch fermentation of Pichia pastoris producing HBsAg: an open-loop control versus recurrent artificial neural network-based feedback control. J Chem Technol Biotechnol 94:3924–3931. https://doi.org/10.1002/jctb.6192
Chang CH, Hsiung HA, Hong KL, Huang CT (2018) Enhancing the efficiency of the Pichia pastoris AOX1 promoter via the synthetic positive feedback circuit of transcription factor Mxr1. BMC Biotechnol 18:81. https://doi.org/10.1186/s12896-018-0492-4
Cos O, Ramon R, Montesinos JL, Valero F (2006) Operational strategies, monitoring and control of heterologous protein production in the methylotrophic yeast Pichia pastoris under different promoters: a review. Microb Cell Fact 5:17. https://doi.org/10.1186/1475-2859-5-17
Gmeiner C, Saadati A, Maresch D, Krasteva S, Frank M, Altmann F, Herwig C, Spadiut O (2015) Development of a fed-batch process for a recombinant Pichia pastoris Δoch1 strain expressing a plant peroxidase. Microb Cell Fact 14:1. https://doi.org/10.1186/s12934-014-0183-3
Gunes H, Boy E, Ata O, Zerze GH, Calik P, ve Ozdamar TH (2016) Methanol feeding strategy design enhances recombinant human growth hormone production by Pichia pastoris. J Chem Technol Biotechnol 91:664–671. https://doi.org/10.1002/jctb.4619
Jahic M, Rotticci-Mulder J, Martinelle M, Hult K, Enfors SO (2002) Modeling of growth and energy metabolism of Pichia pastoris producing a fusion protein. Bioprocess Biosyst Eng 24:385–393. https://doi.org/10.1007/s00449-001-0274-5
Jia L, Li T, Wu Y, Wu C, Li H, Huang A (2021) Enhanced human lysozyme production by Pichia pastoris via periodic glycerol and dissolved oxygen concentrations control. Appl Microbiol Biotechnol 105:1041–1050. https://doi.org/10.1007/s00253-021-11100-9
Karnišová Potocká E, Mastihubová M, Mastihuba V (2021) Apiose-Relevant Glycosidases Catalysts 11:1251. https://doi.org/10.3390/catal11101251
Kastilan R, Boes A, Spiegel H, Voepel N, Chudobová I, Hellwig S, Fischer R (2017) Improvement of a fermentation process for the production of two PfAMA1-DiCo-based malaria vaccine candidates in Pichia pastoris. Sci Rep 7:11991. https://doi.org/10.1038/s41598-017-11819-4
Kongsaeree PT, Ratananikom K, Choengpanya K, Tongtubtima N, Sujiwattanarat P, Porncharoennop C, Onpium A, Svasti J (2010) Substrate specificity in hydrolysis and transglucosylation by family 1 β-glucosidases from cassava and Thai rosewood. J Mol Catal B Enzym 67:257–265. https://doi.org/10.1016/j.molcatb.2010.09.003
Krause M, Neubauer A, Neubauer P (2016) The fed-batch principle for the molecular biology lab: controlled nutrient diets in ready-made media improve production of recombinant proteins in Escherichia coli. Microb Cell Fact 15:110. https://doi.org/10.1186/s12934-016-0513-8
Kytidou K, Artola KM, Overkleeft HS, Aerts JMFG (2020) Plant glycosides and glycosidases: a treasure-trove for therapeutics. Front Plant Sci 11:357. https://doi.org/10.3389/fpls.2020.00357
Li T, Zhang W, Hao J, Sun M, Lin SX (2018) Cold-active extracellular lipase: expression in Sf9 insect cells, purification, and catalysis. Biotechnol Rep (amst) 21:e00295. https://doi.org/10.1016/j.btre.2018.e00295
Liu W, Gong T, Wang QH, Liang X, Chen JJ, Zhu P (2016) Scaling-up fermentation of Pichia pastoris to demonstration-scale using new methanol-feeding strategy and increased air pressure instead of pure oxygen supplement. Sci Rep 6:18439. https://doi.org/10.1038/srep18439
Liu W, Xiang H, Zhang T, Pang X, Su J, Liu H, Ma B, Yu L (2020) Development of a new high-cell density fermentation strategy for enhanced production of a fungus β-glucosidase in Pichia pastoris. Front Microbiol 11:1988. https://doi.org/10.3389/fmicb.2020.0198
Liu W, Sarah I, Ting G, Ashish S, Li-Yan Y, Ping Z (2019) Fed-batch high-cell-density fermentation strategies for Pichia pastoris growth and production. Crit Rev Biotechnol 39:258–271. https://doi.org/10.1080/07388551.2018.1554620
Looser V, Bruhlmann B, Bumbak F, Stenger C, Costa M, Camattari A, Fotiadis D, Kovar K (2015) Cultivation strategies to enhance productivity of Pichia pastoris. A Review Biotechnol Adv 33:1177–1193. https://doi.org/10.1016/j.biotechadv.2015.05.008
Mastropietro G, Aw R, Polizzi KM (2021) Expression of proteins in Pichia pastoris. Methods Enzymol 660:53–80. https://doi.org/10.1016/bs.mie.2021.07.004
Nguyen S, Nguyen H, Truong K (2020) Comparative cytotoxic effects of methanol, ethanol and DMSO on human cancer cell lines. Biomed Res Ther 7:3855–3859. https://doi.org/10.15419/bmrat.v7i7.614
Nieto-Taype MA, Garcia-Ortega X, Albiol J, Montesinos-Seguí JL, Valero F (2020) Continuous cultivation as a tool toward the rational bioprocess development with Pichia pastoris cell factory. Front Bioeng Biotechnol 8:632. https://doi.org/10.3389/fbioe.2020.00632
Paul L, Mudogo C, Mtei K, Machunda R, Ntie-Kang F (2020) A computer-based approach for developing linamarase inhibitory agents. Phys Sci Rev 5:20190098. https://doi.org/10.1515/psr-2019-0098
Poontawee R, Limtong S (2020) Feeding strategies of two-stage fed-batch cultivation processes for microbial lipid production from sugarcane top hydrolysate and crude glycerol by the oleaginous red yeast Rhodosporidiobolus fluvialis. Microorganisms 8:151. https://doi.org/10.3390/microorganisms8020151
Ratananikom K, Choengpanya K, Tongtubtim N, Charoenrat T, Withers SG, Kongsaeree PT (2013) Mutational analysis in the glycone binding pocket of Dalbergia cochinchinensis β-glucosidase to increase catalytic efficiency toward mannosides. Carbohydr Res 373:35–41. https://doi.org/10.1016/j.carres.2012.10.018
Sultan IN, Keawsompong S, Kongsaeree P, Parakulsuksatid P (2020) Formulation of an efficient combinatorial cellulase cocktail by comparative analysis of gibson assembly and NEBuilder HiFi DNA assembly modus operandi. Int J Emerging Technol 11:490–495
Tissopi T, Kumar S, Sadhu A, Mutturi S (2022) Surface display of novel transglycosylating α-glucosidase from Aspergillus neoniger on Pichia pastoris for synthesis of isomaltooligosaccharides. Biochem Eng J 181:108400. https://doi.org/10.1016/j.bej.2022.108400
Toonkool P, Metheenukul P, Sugiwattanrat P, Paiboon P, Tongtubtim N, Ketudat-Cairns M, Ketudat-Cairsns J, Svasti J (2006) Expression and purification of dalcochinase, a β-glucosidase from Dalbergia cochinchinesis Pierre, in yeast and bacteria hosts. Protein Expr Purif 48:195–204. https://doi.org/10.1016/j.pep.2006.05.011
Torres P, Saa PA, Albiol J, Ferrer P, Agosin E (2019) Contextualized genome-scale model unveils high-order metabolic effects of the specific growth rate and oxygenation level in recombinant Pichia pastoris. Metab Eng Commun 9:e00103. https://doi.org/10.1016/j.mec.2019.e00103
Türkanoğlu Özçelik A, Yılmaz S, Inan M (2019) Pichia pastoris promoters. Methods Mol Biol 1923:97–112. https://doi.org/10.1007/978-1-4939-9024-5_3
Wang J, Wang X, Shi L, Qi F, Zhang P, Zhang Y, Zhou X, Song Z, Cai M (2017) Methanol-independent protein expression by AOX1 promoter with trans-acting elements engineering and glucose-glycerol-shift induction in Pichia pastoris. Sci Rep 7:41850. https://doi.org/10.1038/srep41850
Wollborn D, Müller RL, Munkler LP, Horstmann R, Germer A, Blank LM, Büchs J (2022) Auto-induction screening protocol for ranking clonal libraries of Pichia pastoris MutS strains. Biotechnol Bioproc E 27:572–585. https://doi.org/10.1007/s12257-022-0006-z
Acknowledgements
This research was supported by the Department of Biotechnology, Faculty of Agro-Industry, Kasetsart University, Thailand. The authors also acknowledge the support of Associate Professor Prachumporn Kongsaree, Department of Biochemistry, Kasetsart University, Bangkok, Thailand.
Author information
Authors and Affiliations
Contributions
Sirirat Changming: investigation and writing-original draft. Prachumporn Kongsaree: resource person and supervision. Imrana Niaz Sultan: validation and review and editing the draft. Afrasiab Khan Tareen: formal analysis, conceptualization, and review and editing draft. Wirat Vanichsriratana: project administration and resources. Sarote Sirisansaneeyakul: supervision and formal analysis. Pramuk Parakulsuksatid: conceptualization, supervision, project administration, and resources.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Changming, S., Kongsaree, P., Sultan, I.N. et al. β-glucosidase production by recombinant Pichia pastoris strain Y1433 under optimal feed profiles of fed-batch cultivation. Folia Microbiol 68, 245–256 (2023). https://doi.org/10.1007/s12223-022-01008-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-022-01008-w