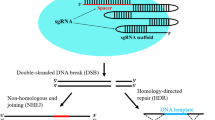
Abstract
Kojic acid is an industrially important secondary metabolite produced by Aspergillus oryzae. The construction of genetic materials for kojic acid related genes is important for understanding the mechanism of kojic acid synthesis in A. oryzae. However, multigene simultaneous knockout mutants for kojic acid synthesis genes remain limited because A. oryzae is multinuclear and good selectable markers are scarce. Here, we firstly successfully obtained single mutants of kojA, kojR, and kojT by our previously constructed CRISPR/Cas9 system in A. oryzae, which demonstrated the feasibility of the targeting sgRNAs for kojA, kojR, and kojT. Then, the AMA1-based genome-editing system for multiplex gene editing was developed in A. oryzae. In the multiplex gene-editing system, two guide RNA expression cassettes were ligated in tandem and driven by two U6 promoters in the AMA1-based autonomously replicating plasmid with the Cas9-expression cassette. Moreover, the multiplex gene-editing technique was applied to target the kojic acid synthesis genes kojA, kojR, and kojT, and the double and triple mutants within kojA, kojR, and kojT were obtained successfully. Additionally, the selectable marker pyrG was knocked out in the single and triple mutants of kojA, kojR, and kojT to obtain the auxotrophic strains, which can facilitate to introduce a target gene into the single and triple mutants of kojA, kojR, and kojT for investigating their relationship. The multiplex gene-editing system and release of these materials provide a foundation for further kojic acid research and utilization.





Similar content being viewed by others
Availability of data and material
All datasets obtained for this study are included in the manuscript/Supplementary data.
References
Ammar HAM, Ezzat SM, Houseny AM (2017) Improved production of kojic acid by mutagenesis of Aspergillus flavus HAk1 and Aspergillus oryzae HAk2 and their potential antioxidant activity. 3 Biotech 7(5):276. https://doi.org/10.1007/s13205-017-0905-4
Arakawa GY, Kudo H, Yanase A, Eguchi Y, Kodama H, Ogawa M, Koyama Y, Shindo H, Hosaka M, Tokuoka M (2019) A unique Zn(II)2-Cys6-type protein, KpeA, is involved in secondary metabolism and conidiation in Aspergillus oryzae. Fungal Genet Biol 127:35–44. https://doi.org/10.1016/j.fgb.2019.02.004
Bentley R (2006) From miso, sake and shoyu to cosmetics: a century of science for kojic acid. Nat Prod Rep 23:1046–1062. https://doi.org/10.1039/b603758p
Brtko J, Rondahl L, Fickova M, Hudecova D, Eybl V, Uher M (2004) Kojic acid and its derivatives: history and present state of art. Central Cent Eur J Public Health 12(Suppl):S16-18
Chaudhary J, Pathak AN, Lakhawat S (2014) Production technology and applications of kojic acid. Annu Res Rev Biol 4:3165–3196. https://doi.org/10.9734/ARRB/2014/10643
Chutrakul C, Panchanawaporn S, Jeennor S, Anantayanon J, Vorapreeda T, Vichai V, Laoteng K (2019) Functional characterization of novel U6 RNA polymerase III promoters: their implication for CRISPR-Cas9-mediated gene editing in Aspergillus oryzae. Curr Microbiol 76:1443–1451. https://doi.org/10.1007/s00284-019-01770-0
Donohoue PD, Barrangou R, May AP (2018) Advances in industrial biotechnology using CRISPR-Cas systems. Trends Biotechnol 36:134–146. https://doi.org/10.1016/j.tibtech.2017.07.007
Fan J, Zhang Z, Long C, He B, Hu Z, Jiang C, Zeng B (2020) Identification and functional characterization of glycerol dehydrogenase reveal the role in kojic acid synthesis in Aspergillus oryzae. World J Microb Biot 36:136. https://doi.org/10.1007/s11274-020-02912-4
Feng W, Liang J, Wang B, Chen J (2019) Improvement of kojic acid production in Aspergillus oryzae AR-47 mutant strain by combined mutagenesis. Bioprocess Biosyst Eng 42:753–761. https://doi.org/10.1007/s00449-019-02079-9
Jiang F, Doudna JA (2017) CRISPR-Cas9 structures and mechanisms. Annu Rev Biophys 46:505–529. https://doi.org/10.1146/annurev-biophys-062215010822
Kadooka C, Yamaguchi M, Okutsu K, Yoshizaki Y, Takamine K, Katayama T, Maruyama JI, Tamaki H, Futagami T (2020) A CRISPR/Cas9-mediated gene knockout system in Aspergillus luchuensis mut. kawachii. Biosci Biotechnol Biochem 84:2179–2183. https://doi.org/10.1080/09168451.2020.1792761
Katayama T, Nakamura H, Zhang Y, Pascal A, Fujii W, Maruyama JI (2019) Forced recycling of an AMA1-based genome-editing plasmid allows for efficient multiple gene deletion/integration in the industrial filamentous fungus Aspergillus oryzae. Appl Environ Microbiol 85:e01896-e1918. https://doi.org/10.1128/AEM.01896-18
Katayama T, Tanaka Y, Okabe T, Nakamura H, Fujii W, Kitamoto K, Maruyama J (2016) Development of a genome editing technique using the CRISPR/Cas9 system in the industrial filamentous fungus Aspergillus oryzae. Biotechnol Lett 38:637–642. https://doi.org/10.1007/s10529-015-2015-x
Kawauchi M, Nishiura M, Iwashita K (2013) Fungus-specific sirtuin HstD coordinates secondary metabolism and development through control of LaeA. Eukaryot Cell 12:1087–1096. https://doi.org/10.1128/EC.00003-13
Labuhn M, Adams FF, Ng M, Knoess S, Schambach A, Charpentier EM, Schwarzer A, Mateo JL, Klusmann JH, Heckl D (2018) Refined sgRNA efficacy prediction improves large- and small-scale CRISPR-Cas9 applications. Nucleic Acids Res 46:1375–1385. https://doi.org/10.1093/nar/gkx1268
Malina A, Cameron CJF, Robert F, Blanchette M, Dostie J, Pelletier J (2015) PAM multiplicity marks genomic target sites as inhibitory to CRISPR-Cas9 editing. Nat Commun 6:10124. https://doi.org/10.1038/ncomms10124
Marui J, Yamane N, Ohashi-Kunihiro S, Ando T, Terabayashi Y, Sano M, Ohashi S, Ohshima E, Tachibana K, Higa Y, Nishimura M, Koike H, Machida M (2011) Kojic acid biosynthesis in Aspergillus oryzae is regulated by a Zn(II)2Cys6 transcriptional activator and induced by kojic acid at the transcriptional level. J Biosci Bioeng 112:40–43. https://doi.org/10.1016/j.jbiosc.2011.03.010
Maruyama J, Kitamoto K (2011) Targeted gene disruption in Koji mold Aspergillus oryzae. Methods Mol Biol 765:447–456. https://doi.org/10.1007/978-1-61779-197-0_27
Nodvig CS, Nielsen JB, Kogle ME, Mortensen UH (2015) A CRISPR-Cas9 system for genetic engineering of filamentous fungi. PLoS One 10:e0133085. https://doi.org/10.1371/journal.pone.0133085
Oda K, Kobayashi A, Ohashi S, Sano M (2014) Aspergillus oryzae laeA regulates kojic acid synthesis genes. Biosci Biotech Bioch 75(9):1832–1834. https://doi.org/10.1271/bbb.110235
Saeedi M, Eslamifar M, Khezri K (2019) Kojic acid applications in cosmetic and pharmaceutical preparations. Biomed Pharmacother 110:582–593. https://doi.org/10.1016/j.biopha.2018.12.006
Sano M (2016) Aspergillus oryzae nrtA affects kojic acid production. Biosci Biotechnol Biochem 80:1776–1780. https://doi.org/10.1080/09168451.2016.1176517
Terabayashi Y, Sano M, Yamane N, Marui J, Tamano K, Sagara J, Dohmoto M, Oda K, Ohshima E, Tachibana K, Higa Y, Ohashi S, Koike H, Machida M (2010) Identification and characterization of genes responsible for biosynthesis of kojic acid, an industrially important compound from Aspergillus oryzae. Fungal Genet Biol 47:953–961. https://doi.org/10.1016/j.fgb.2010.08.014
Wan HM, Chen CC, Giridhar R, Chang TS, Wu WT (2005) Repeated-batch production of kojic acid in a cell-retention fermenter using Aspergillus oryzae M3B9. J Ind Microbiol Biotechnol 32:227–233. https://doi.org/10.1007/s10295-005-0230-5
Yamada R, Yoshie T, Wakai S, Asai-Nakashima N, Okazaki F, Ogino C, Hisada H, Tsutsumi H, Hata Y, Kondo A (2014) Aspergillus oryzae-based cell factory for direct kojic acid production from cellulose. Microb Cell Fact 13:71. https://doi.org/10.1186/1475-2859-13-71
Funding
This work was supported by Natural Science Foundation of China (32000049), Jiangxi Provincial Natural Science Foundation (20202BABL213042), Science and Technology Research Project of Jiangxi Provincial Department of Education (GJJ190611), and Doctoral Scientific Research Foundation of Jiangxi Science and Technology Normal University (2018BSQD030).
Author information
Authors and Affiliations
Contributions
BZ and ZZ designed the research and wrote the manuscript. YL, HZ, ZC, JF, and TC performed experiments. ZZ, YL, and HZ analyzed data. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals experiments.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Zhang, H., Chen, Z. et al. Construction of single, double, or triple mutants within kojic acid synthesis genes kojA, kojR, and kojT by the CRISPR/Cas9 tool in Aspergillus oryzae. Folia Microbiol 67, 459–468 (2022). https://doi.org/10.1007/s12223-022-00949-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-022-00949-6