Abstract
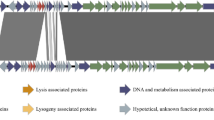
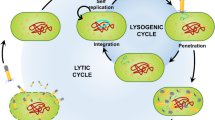
Antibiotic resistance is increasing among Staphylococcus saprophyticus strains isolated from urinary tract infection. This necessitates alternative therapies. For this, a lytic phage (vB_SsapS-104) against S. saprophyticus, which formed round and clear plaques on bacterial culture plates, was isolated from hospital wastewater and characterized. Microscopy analysis showed that it had a small head (about 50 nm), tail (about 80 nm), and a collar (about 22 nm in length and 19 nm in width) indicating to be a phage within Siphoviridae family. Phage vB_SsapS-104 showed a large latency period of about 40 min, rapid adsorption rate that was significantly enhanced by MgCl2 and CaCl2, and high stability to a wide range of temperatures and pH values. Restriction analyses demonstrated that phage consists of a double-stranded DNA with an approximate genome size of 40 Kb. BLAST results did not show high similarity (megablast) with other previously identified phages. But, in Blastn, similarity with Staphylococcus phages was observed. Phage vB_SsapS-104 represented high anti-bacterial activity against S. saprophyticus isolates in vitro as it was able to lyse 8 of the 9 clinical isolates (%88.8) obtained from a hospital in Gorgan, Iran. It was a S. saprophyticus-specific phage because no lytic activity was observed on some other pathogenic bacteria tested. Therefore, phage vB_SsapS-104 can be considered as a specific virulent phage against of S. saprophyitcus isolated from urinary tract infection. This study provided the partial genomic characterization of S. saprophyticus phage and its application against urinary tract infection associated with S. saprophyticus. This phage also can be considered as a good candidate for a therapeutic alternative in the future.







Similar content being viewed by others
References
Abedon ST, Kuhl SJ, Blasdel BG, Kutter EM (2011) Phage treatment of human infections. Bacteriophage 1:66–85. https://doi.org/10.4161/bact.1.2.15845
Ackermann HW (1998) Tailed bacteriophages: the order caudovirales. Adv Virus Res 51:135–201. https://doi.org/10.1016/S0065-3527(08)60785-X
Bes M (1994) Characterization of thirteen Staphylococcus epidermidis and S. saprophyticus bacteriophages. Res Virol 145:111–121. https://doi.org/10.1016/S0923-2516(07)80013-6
Bes M, Ackermann HW, Brun Y, Fleurette J (1990) Morphology of Staphylococcus saprophyticus bacteriophages
Carson L, Gorman SP, Gilmore BF (2010) The use of lytic bacteriophages in the prevention and eradication of biofilms of Proteus mirabilis and Escherichia coli. FEMS Immunol Med Microbiol 59:447–455. https://doi.org/10.1111/j.1574-695X.2010.00696.x
Cerca N, Oliveira R, Azeredo J (2007) Susceptibility of Staphylococcus epidermidis planktonic cells and biofilms to the lytic action of staphylococcus bacteriophage K. Lett Appl Microbiol 45:313–317. https://doi.org/10.1111/j.1472-765X.2007.02190.x
Curtin JJ, Donlan RM (2006) Using bacteriophages to reduce formation of catheter-associated biofilms by Staphylococcus epidermidis. Antimicrob Agents Chemother 50:1268–1275. https://doi.org/10.1128/AAC.50.4.1268-1275.2006
Deghorain M, Van Melderen L (2012) The staphylococci phages family: an overview. Viruses 4:3316–3335. https://doi.org/10.3390/v4123316
Estrella LA, Quinones J, Henry M, Hannah RM, Pope RK, Hamilton T, Teneza-mora N, Biswajit EH (2016) Characterization of novel Staphylococcus aureus lytic phage and defining their combinatorial virulence using the OmniLog® system. Bacteriophage 6(3):e1219440. https://doi.org/10.1080/21597081.2016.1219440
Foster RT Sr (2008) Uncomplicated urinary tract infections in women. Obstet Gynecol Clin N Am 35:235–248. https://doi.org/10.1016/j.ogc.2008.03.003
Fowler JE (1985) Staphylococcus saprophyticus as the cause of infected urinary calculus. Ann Intern Med 102:342–343. https://doi.org/10.7326/0003-4819-102-3-342
Ghasemi SM, Bouzari M, Baygloo NS, Chang HI (2014a) Insights into new bacteriophages of Lactococcus garvieae belonging to the family Podoviridae. Arch Virol 159:2909–2915. https://doi.org/10.1007/s00705-014-2142-z
Ghasemi SM, Bouzari M, Emtiazi G (2014b) Preliminary characterization of Lactococcus garvieae bacteriophage isolated from wastewater as a potential agent for biological control of lactococcosis in aquaculture. Aquac Int 22:1469–1480
Gutiérrez D, Martínez B, Rodríguez A, García P (2012) Genomic characterization of two Staphylococcus epidermidis bacteriophages with anti-biofilm potential. BMC Genomics 13:228. https://doi.org/10.1186/1471-2164-13-228
Hsieh SE, Lo HH, Chen ST, Lee MC, Tseng YH (2011) Wide host range and strong lytic activity of Staphylococcus aureus lytic phage Stau2. Appl Environ Microbiol 77:756–761. https://doi.org/10.1128/AEM.01848-10
Jamal M, Hussain T, Das CR, Andleeb S (2015) Characterization of Siphoviridae phage Z and studying its efficacy against multidrug-resistant Klebsiella pneumoniae planktonic cells and biofilm. J Med Microbiol 64:454–462. https://doi.org/10.1099/jmm.0.000040
Jhora ST, Paul S (2011) Urinary tract infections caused by Staphylococcus saprophyticus and their antimicrobial sensitivity pattern in young adult women. Bangladesh J Med Microbiol 5:21–25. https://doi.org/10.3329/bjmm.v5i1.15817
Komijani M, Bouzari M, Rahimi F (2016) Detection and characterization of a novel lytic bacteriophage (vB-KpneM-Isf48) against Klebsiella pneumoniae isolates from infected wounds carrying antibiotic-resistance genes (TEM, SHV, and CTX-M). Iran Red Crescent Med J 19:e34475. https://doi.org/10.5812/ircmj.34475
Kropinski AM, Prangishvili D, Lavigne R (2009) Position paper: the creation of a rational scheme for the nomenclature of viruses of bacteria and archaea. Environ Microbiol 11:2775–2777. https://doi.org/10.1111/j.1462-2920.2009.01970.x
Kutter E, De Vos D, Gvasalia G, Alavidze Z, Gogokhia L, Kuhl S, Abedon ST (2010) Phage therapy in clinical practice: treatment of human infections. Curr Pharm Biotechnol 11:69–86
Kwiatek M, Parasion S, Mizak L, Gryko R, Bartoszcze M, Kocik J (2012) Characterization of a bacteriophage, isolated from a cow with mastitis, that is lytic against Staphylococcus aureus strains. Arch Virol 157:225–234. https://doi.org/10.1007/s00705-011-1160-3
Li L, Zhang Z (2014) Isolation and characterization of a virulent bacteriophage SPW specific for Staphylococcus aureus isolated from bovine mastitis of lactating dairy cattle. Mol Biol Rep 41:5829–5838. https://doi.org/10.1007/s11033-014-3457-2
Łobocka M et al (2012) Genomics of staphylococcal Twort-like phages—potential therapeutics of the post-antibiotic era. Adv Virus Res 83:143–216. https://doi.org/10.1016/B978-0-12-394438-2.00005-0
Matsuzaki S et al (2005) Bacteriophage therapy: a revitalized therapy against bacterial infectious diseases. J Infect Chemother 11:211–219. https://doi.org/10.1007/s10156-005-0408-9
Nivas D, Ramesh N, Krishnakumar V, Rajesh P, Solomon EK, Kannan VR (2015) Distribution, isolation and characterization of lytic bacteriophages against multi-drug resistant and extended-spectrum of β-lactamase producing pathogens from hospital effluents. Asian J Pharm Clin Res 8:384–389
O’flaherty S, Ross R, Flynn J, Meaney W, Fitzgerald G, Coffey A (2005a) Isolation and characterization of two anti-staphylococcal bacteriophages specific for pathogenic Staphylococcus aureus associated with bovine infections. Lett Appl Microbiol 41:482–486. https://doi.org/10.1128/AEM.71.4.1836-1842.2005
O’flaherty S, Ross R, Meaney W, Fitzgerald G, Elbreki M, Coffey A (2005b) Potential of the polyvalent anti-Staphylococcus bacteriophage K for control of antibiotic-resistant staphylococci from hospitals. Appl Environ Microbiol 71:1836–1842. https://doi.org/10.1128/AEM.71.4.1836-1842.2005
O’flaherty S, Flynn J, Coffey A, Fitzgerald G, Meaney WJ, Ross RP (2006) Molecular characterisation of bacteriophage K towards applications for the biocontrol of pathogenic staphylococci. Doctoral thesis University College, Cork, Ireland
Patel J, Cockerill F, Alder J, Bradford P, Eliopoulos G, Hardy D (2014) Performance standards for antimicrobial susceptibility testing; twenty-fourth informational supplement. CLSI standards for antimicrobial susceptibility testing 34:1–226
Poppert S et al (2005) Rapid diagnosis of bacterial meningitis by real-time PCR and fluorescence in situ hybridization. J Clin Microbiol 43:3390–3397. https://doi.org/10.1128/JCM.43.7.3390-3397.2005
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning vol 2. Cold Spring Harbor Laboratory Press, New York
Schmelcher M, Donovan DM, Loessner MJ (2012) Bacteriophage endolysins as novel antimicrobials. Future Microbiol 7:1147–1171
Schneider PF, Riley TV (1996) Staphylococcus saprophyticus urinary tract infections: epidemiological data from Western Australia. Eur J Epidemiol 12:51–54. https://doi.org/10.1007/BF00144428
Shakeri F, Shojai A, Golalipour M, Rahimi Alang S, Vaez H, Ghaemi EA (2010) Spa diversity among MRSA and MSSA strains of Staphylococcus aureus in North of Iran. Int J Microbiol:1–5. https://doi.org/10.1155/2010/351397
Shen GH et al (2012) Isolation and characterization of φkm18p, a novel lytic phage with therapeutic potential against extensively drug resistant Acinetobacter baumannii. PLoS One 7:e46537. https://doi.org/10.1371/journal.pone.0046537
Sulakvelidze A, Alavidze Z, Morris JG Jr (2001) Bacteriophage therapy. Antimicrob Agents Chemother 45:649–659. https://doi.org/10.1128/AAC.45.3.649-659.2001
Synnott AJ, Kuang Y, Kurimoto M, Yamamichi K, Iwano H, Tanji Y (2009) Isolation from sewage influent and characterization of novel Staphylococcus aureus bacteriophages with wide host ranges and potent lytic capabilities. Appl Environ Microbiol 75:4483–4490. https://doi.org/10.1128/AEM.02641-08
Widerström M, Wiström J, Ferry S, Karlsson C, Monsen T (2007) Molecular epidemiology of Staphylococcus saprophyticus isolated from women with uncomplicated community-acquired urinary tract infection. J Clin Microbiol 45:1561–1564. https://doi.org/10.1128/JCM.02071-06
Wittebole X, De Roock S, Opal SM (2014) A historical overview of bacteriophage therapy as an alternative to antibiotics for the treatment of bacterial pathogens. Virulence 5:226–235. https://doi.org/10.4161/viru.25991
Yang H, Liang L, Lin S, Jia S (2010) Isolation and characterization of a virulent bacteriophage AB1 of Acinetobacter baumannii. BMC Microbiol 10:131. https://doi.org/10.1186/1471-2180-10-131
Zamani I, Bouzari M, Emtiazi G, Ghasemi SM, Chang HI (2014) Complete genome sequence of a novel phage, vB_MoxS-ISF9, infecting methylotrophic microbacterium: first report of a virulent microbacterium phage. Arch Virol 159:2537–2540. https://doi.org/10.1007/s00705-014-2092-5
Zhang Q et al (2017) Characterization and complete genome sequence analysis of a novel virulent Siphoviridae phage against Staphylococcus aureus isolated from bovine mastitis in Xinjiang, China. Virus Genes 53:464–476. https://doi.org/10.1007/s11262-017-1445-z
Funding
This study was financially supported by a joint grant from the University of Isfahan and Golestan University of Medical Sciences, Iran [35/219525].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
Formation of plaque on double layer agar plate. (a) Lower dilution of the virus. (b) Higher dilution of the virus. (JPG 724 kb)
Rights and permissions
About this article
Cite this article
Yazdi, M., Bouzari, M. & Ghaemi, E.A. Isolation and characterization of a potentially novel Siphoviridae phage (vB_SsapS-104) with lytic activity against Staphylococcus saprophyticus isolated from urinary tract infection. Folia Microbiol 64, 283–294 (2019). https://doi.org/10.1007/s12223-018-0653-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-018-0653-9