Abstract
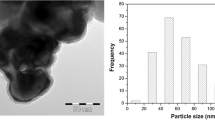
One of the main mechanisms of nanoparticle toxicity is known to be the generation of reactive oxygen species (ROS) which primarily damage cell membranes. However, very limited data on membrane effects in anaerobic environments (where ROS could not be the cause of membrane damage) are available. In the following study, rumen anaerobe Ruminococcus flavefaciens 007C was used as a bacterial model to assess the potential effects of Al2O3 and TiO2 nanoparticles on membranes in an anaerobic environment. Fatty acid profiles of cultures after exposure to Al2O3 or TiO2 nanoparticles were analyzed and compared with the profiles of non-exposed cultures or cultures exposed to bulk materials. Analysis revealed dose–effect changes in membrane composition exclusively when cells were exposed to Al2O3 nanoparticles in a concentration range of 3–5 g/L, but were not present in cultures exposed to bulk material. On the other hand, the tested concentrations of nano-TiO2 did not significantly affect the membrane profile of the exposed bacterium. The results suggest the possibility that Al2O3 induces changes in bacterial membranes by direct physical interaction, which was supported by TEM image analysis.


Similar content being viewed by others
Abbreviations
- DMA(s):
-
Dimethyl acetal(s)
- FA(s):
-
Fatty acid(s)
- ROS:
-
Reactive oxygen species
- TEM:
-
Transmission electron microscopy
References
Behra R, Krug H (2008) Nanoparticles at large. Nat Nanotechnol 3:253–254
Chattopadhyay MK, Jagannadham MV (2003) A branched chain fatty acid promotes cold adaptation in bacteria. J Biosci 28:363–364
Fiedler SL, Violi A (2010) Simulation of nanoparticle permeation through a lipid membrane. Biophys J 99:144–152
Giotis ES, McDowel DA, Blair IS, Wilkinson BJ (2007) Role of branched-chain fatty acids in pH stress tolerance in Listeria monocytogenes. Appl Environ Microbiol 73:997–1001
Ivancic T, Vodovnik M, Marinsek Logar R, Stopar D (2009) Conditioning of the membrane fatty acid profile of Escherichia coli during periodic temperature cycling. Microbiology 155:3461–3463
Kahru A, Dubourguier HC (2010) From ecotoxicology to nanoecotoxicology. Toxicology 269:105–119
Mjøs SA (2003) Identification of fatty acids in gas chromatography by application of different temperature and pressure programs on a single capillary column. J Chromatogr A 1015:151–161
Mortimer M, Kasemets K, Vodovnik M, Marinšek Logar R, Kahru A (2011) Exposure to CuO nanoparticles changes the fatty acid composition of protozoa Tetrahymena thermophila. Environ Sci Technol 45:6617–6624
Neal AL (2008) What can be inferred from bacterium–nanoparticle interactions about the potential consequences of environmental exposure to nanoparticles? Ecotoxicology 17:362–371
Rosa SM, Atunes-Madeira MC, Matos MJ, Jurado AS, Madeira VM (2000) Lipid composition and dynamics of cell membranes of Bacillus stearothermophilus adapted to amiodarone. Biochim Biophys Acta 1487:286–295
Saluzzi L, Flint HJ, Stewart CS (2001) Adaptation of Ruminococcus flavefaciens resulting in increased degradation of ryegrass cell walls. FEMS Microbiol Ecol 36:131–137
Singh AK, Zhang YM, Zhu K, Subramanian C, Li Z, Jayaswal RK, Gatto C, Rock CO, Wilkinson BJ (2009) FabH selectivity for anteiso branched-chain fatty acid precursors in low-temperature adaptation in Listeria monocytogenes. FEMS Microbiol Lett 30:188–192
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vodovnik, M., Kostanjšek, R., Zorec, M. et al. Exposure to Al2O3 nanoparticles changes the fatty acid profile of the anaerobe Ruminococcus flavefaciens . Folia Microbiol 57, 363–365 (2012). https://doi.org/10.1007/s12223-012-0143-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-012-0143-4